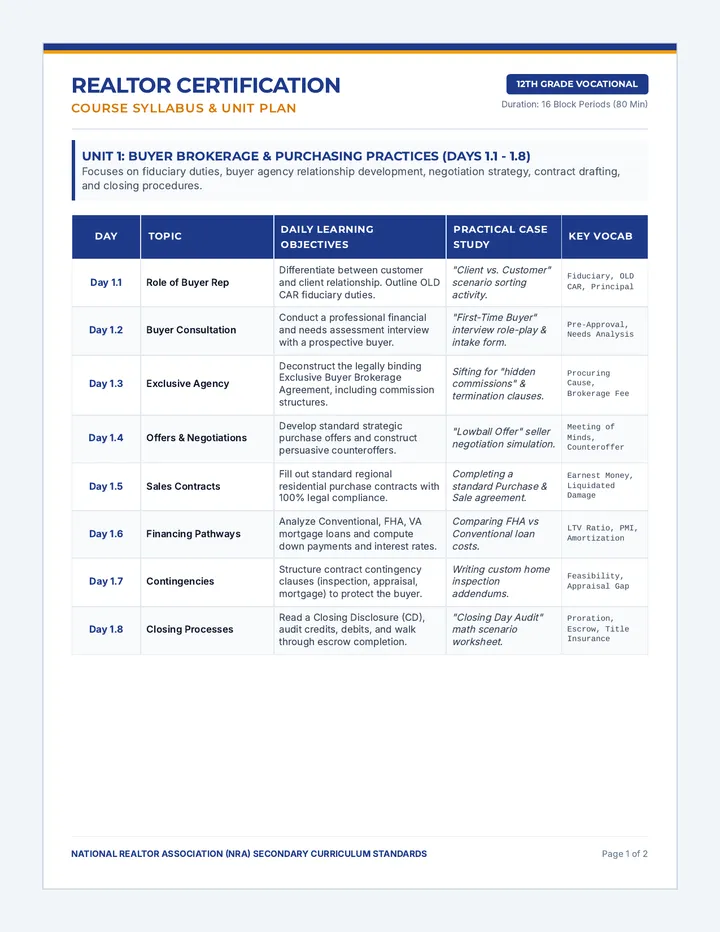
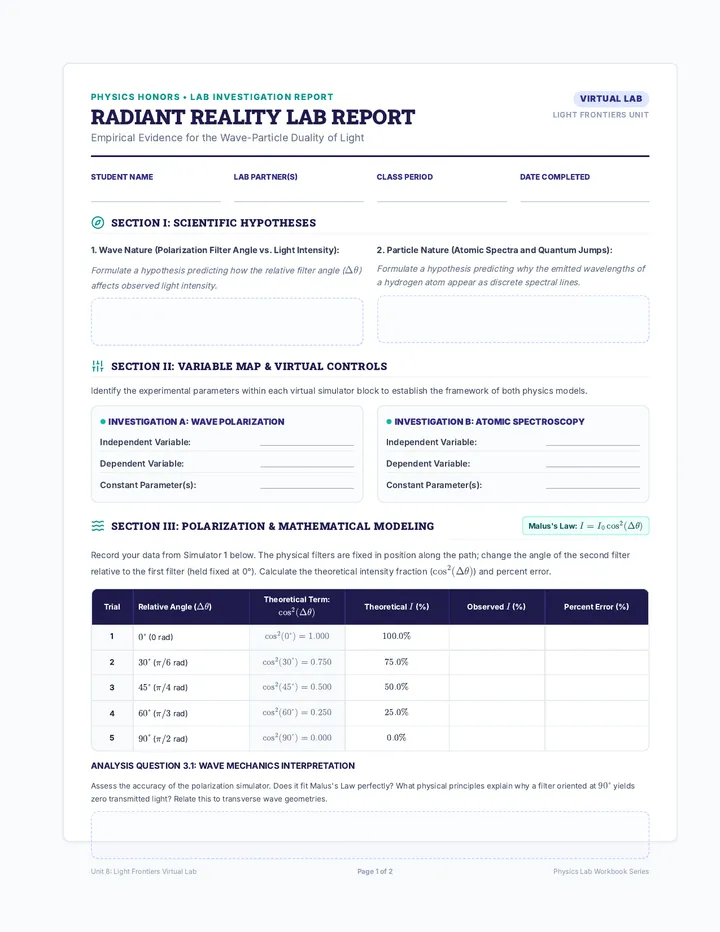
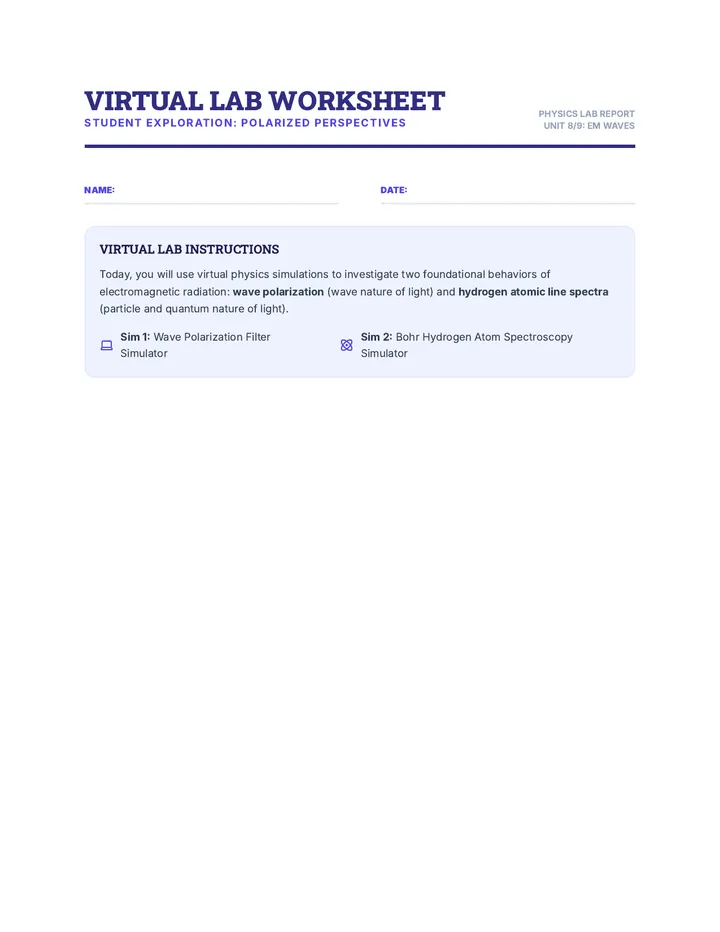
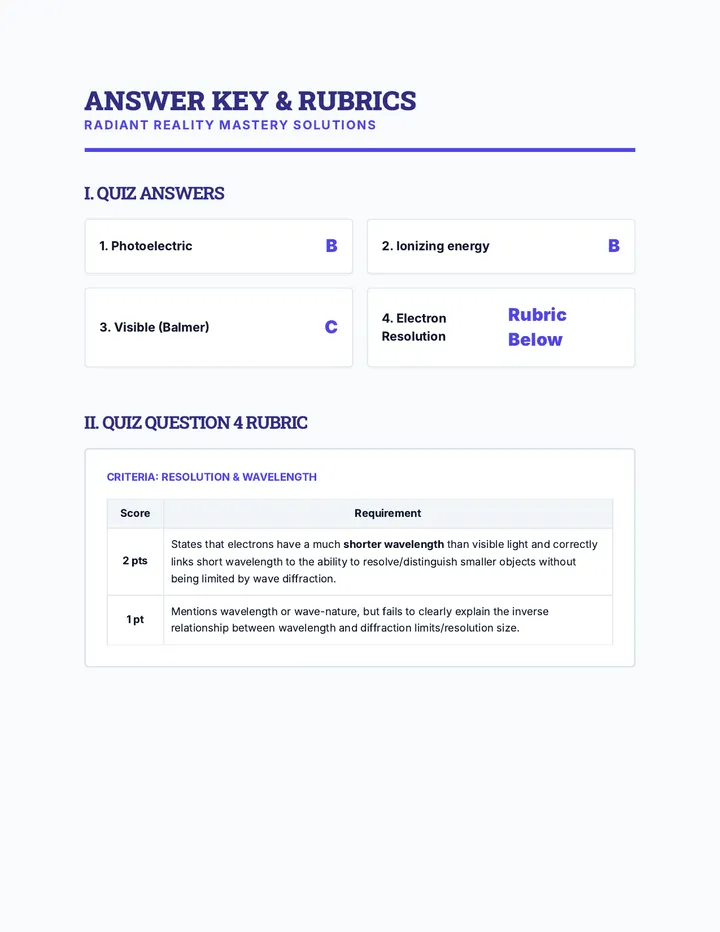
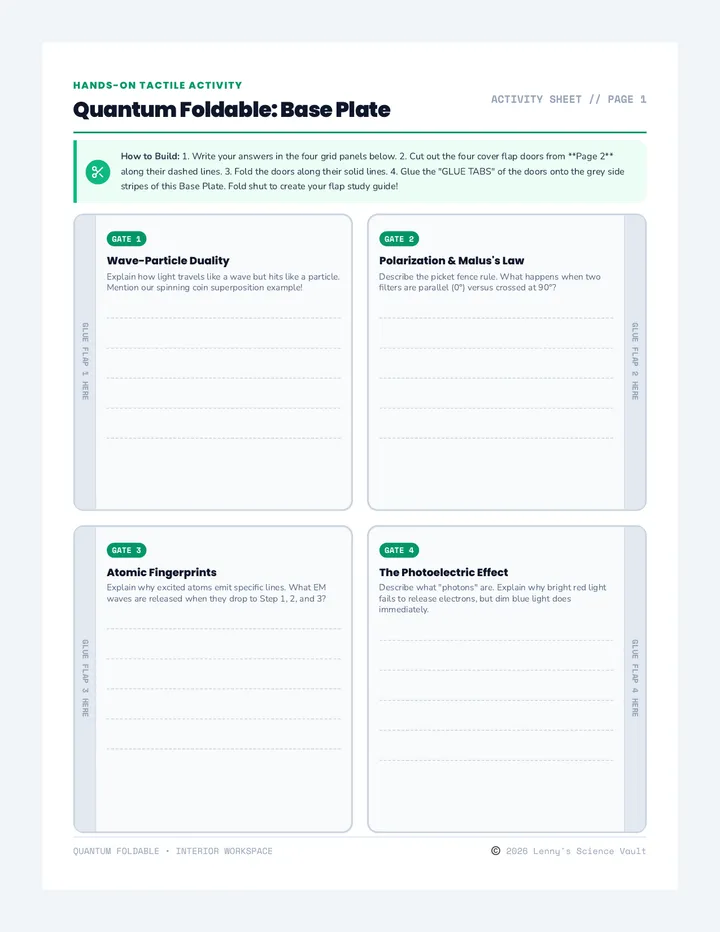
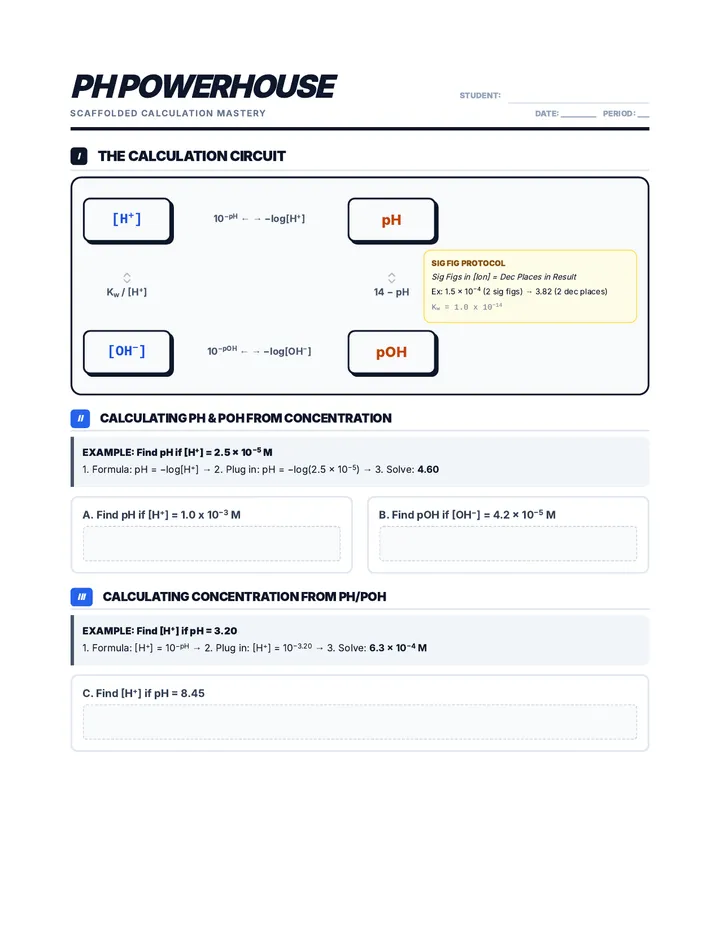
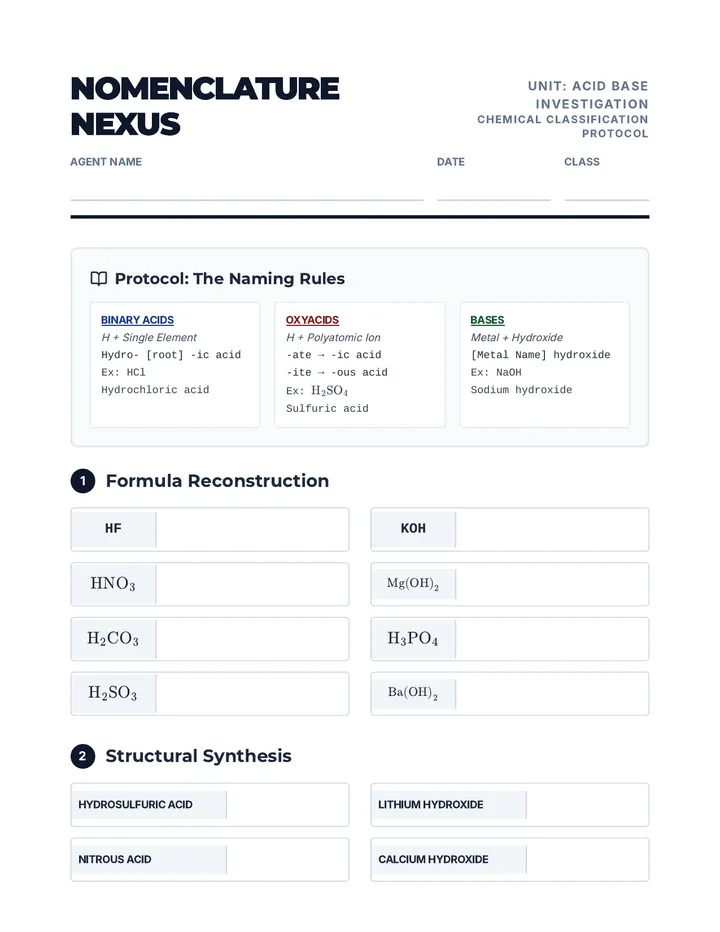
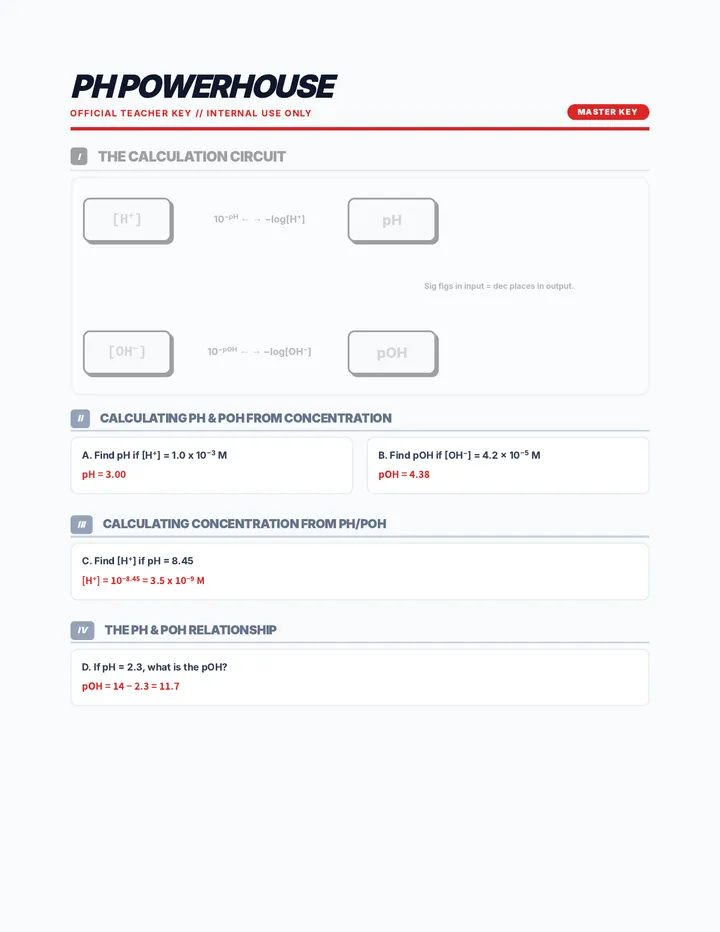
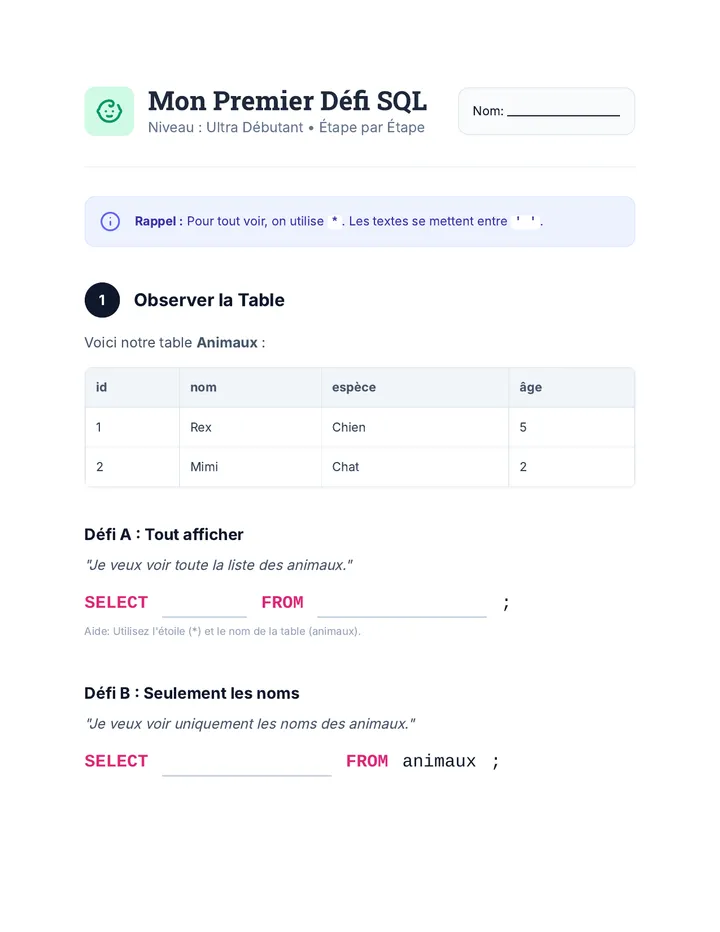
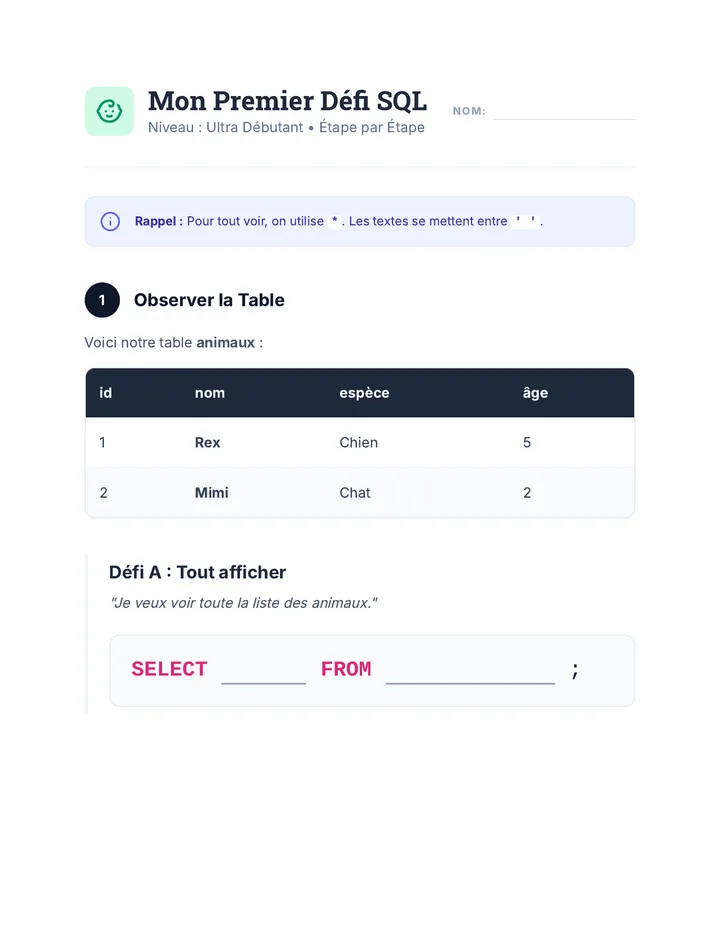
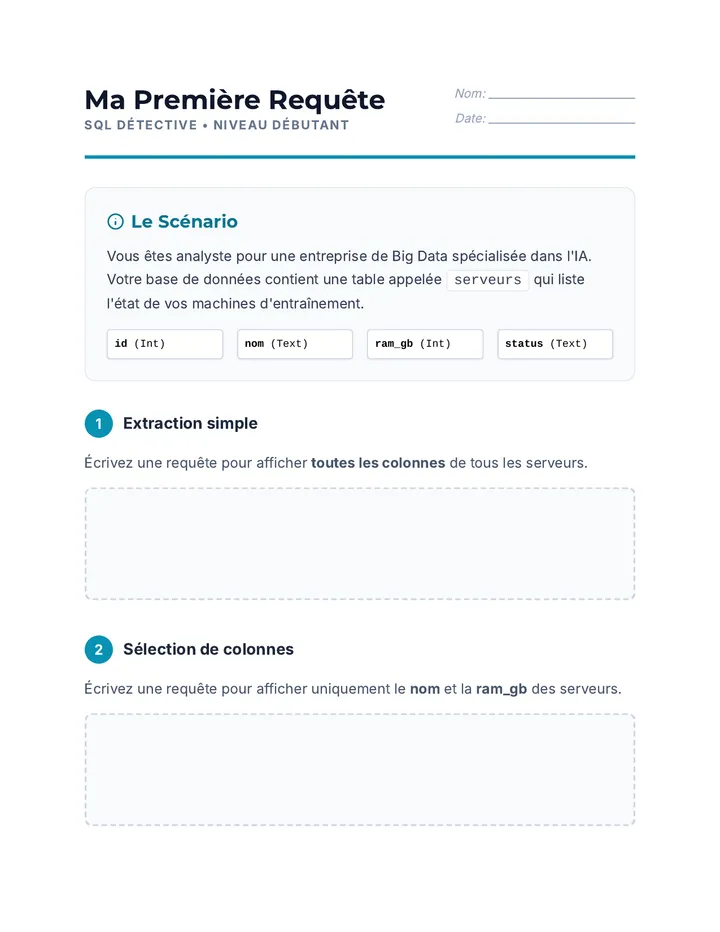
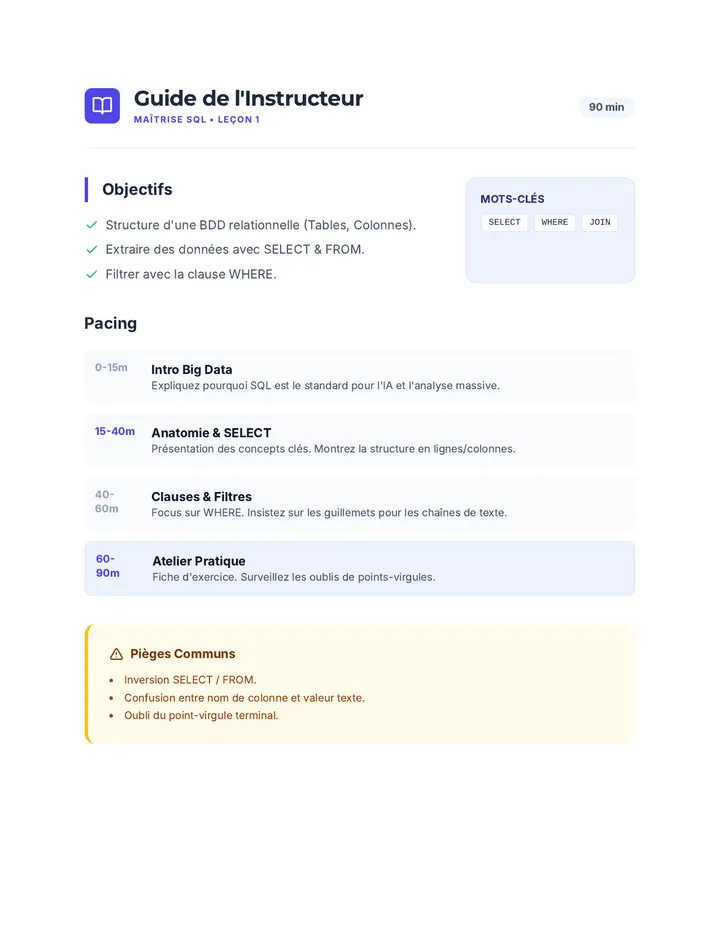
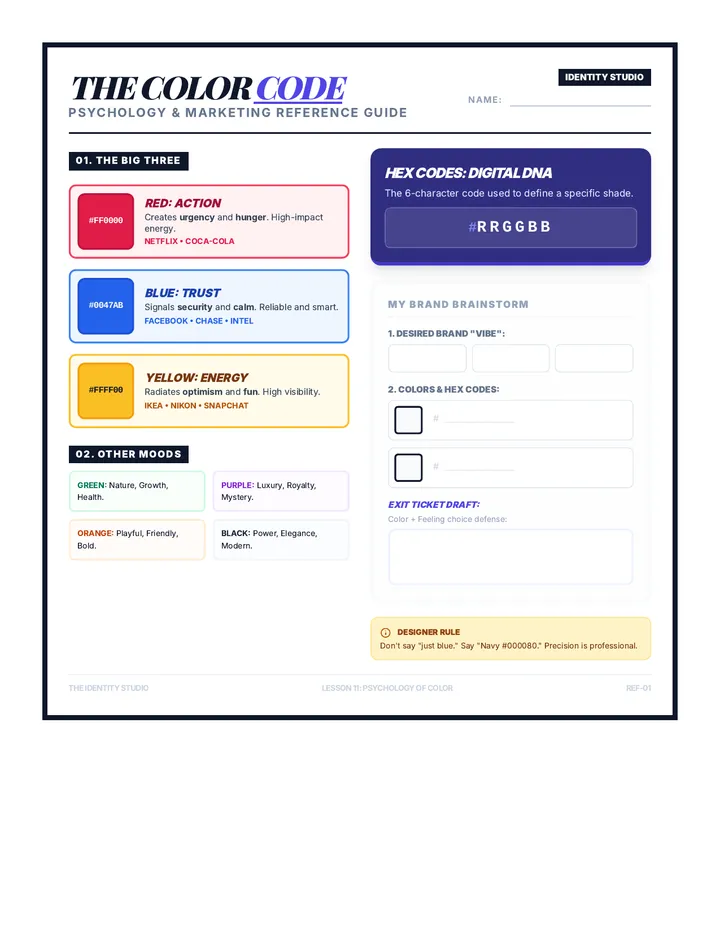
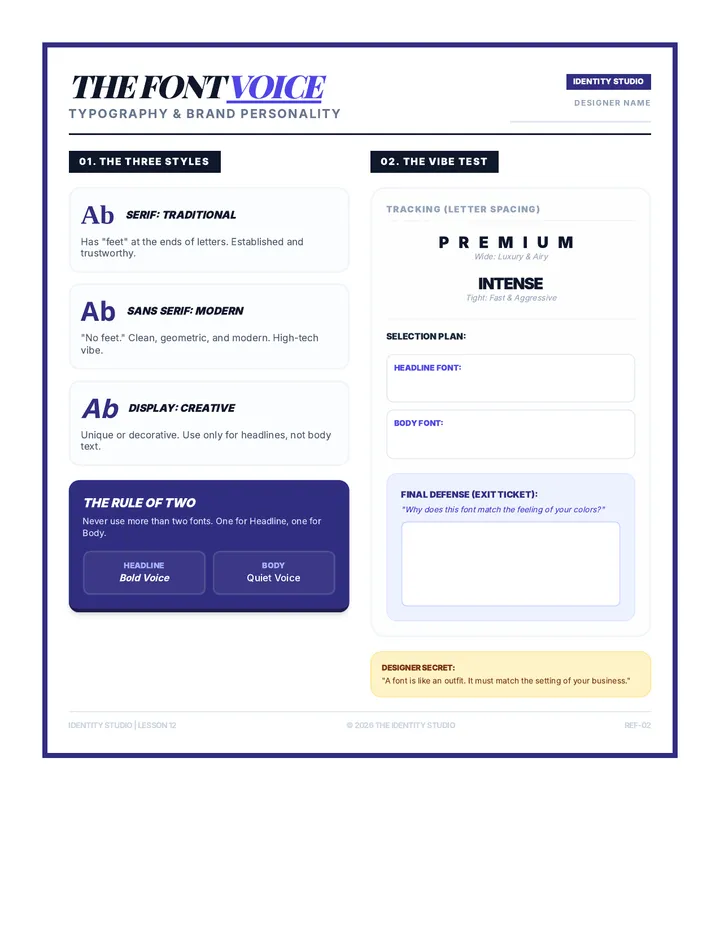
Real Gas Dynamics
This undergraduate chemistry sequence explores the mathematical and physical reasons why gases deviate from ideal behavior. Students use the compressibility factor and the Van der Waals equation to model real gas behavior, culminating in industrial engineering applications and phase analysis.