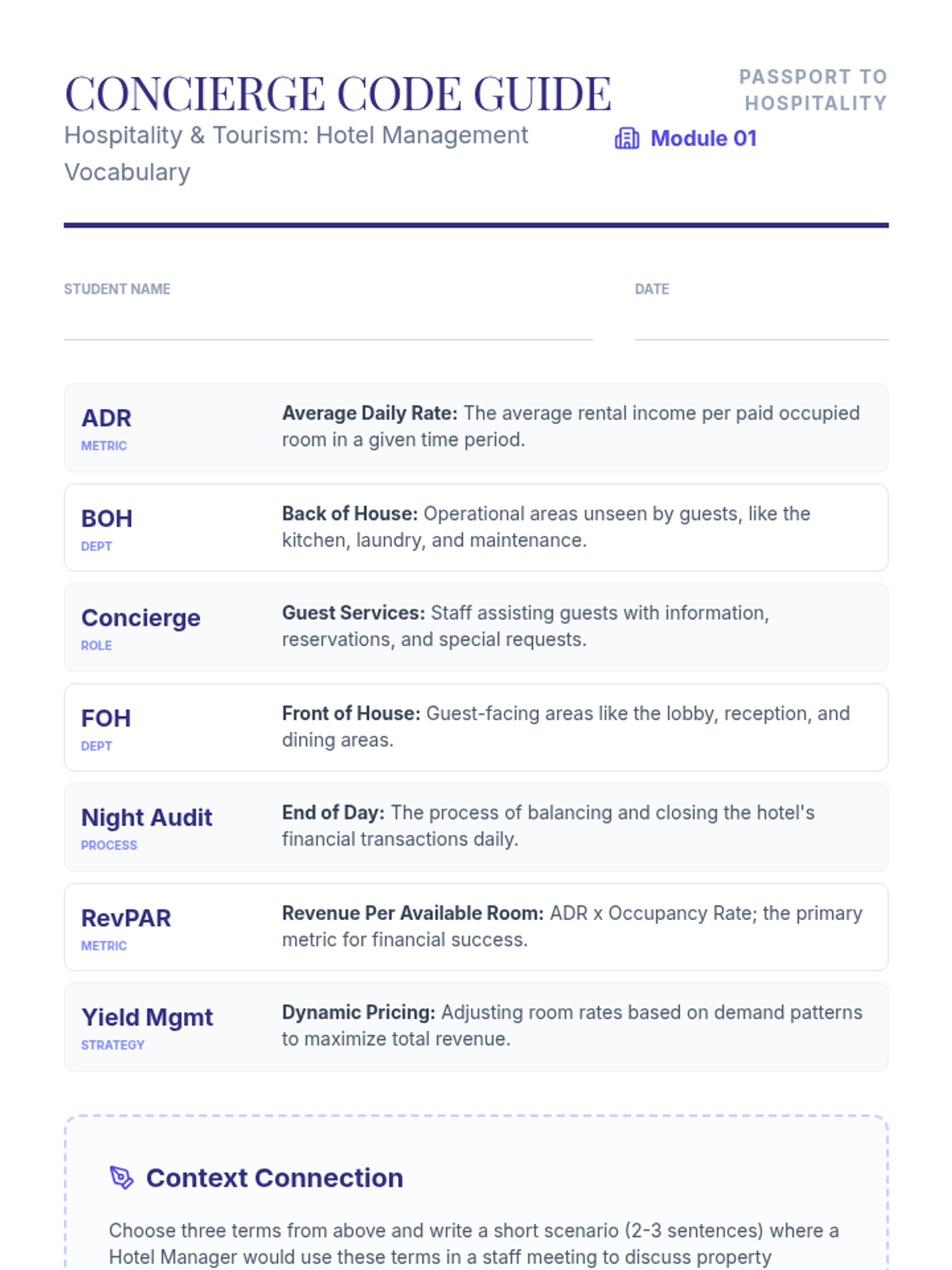
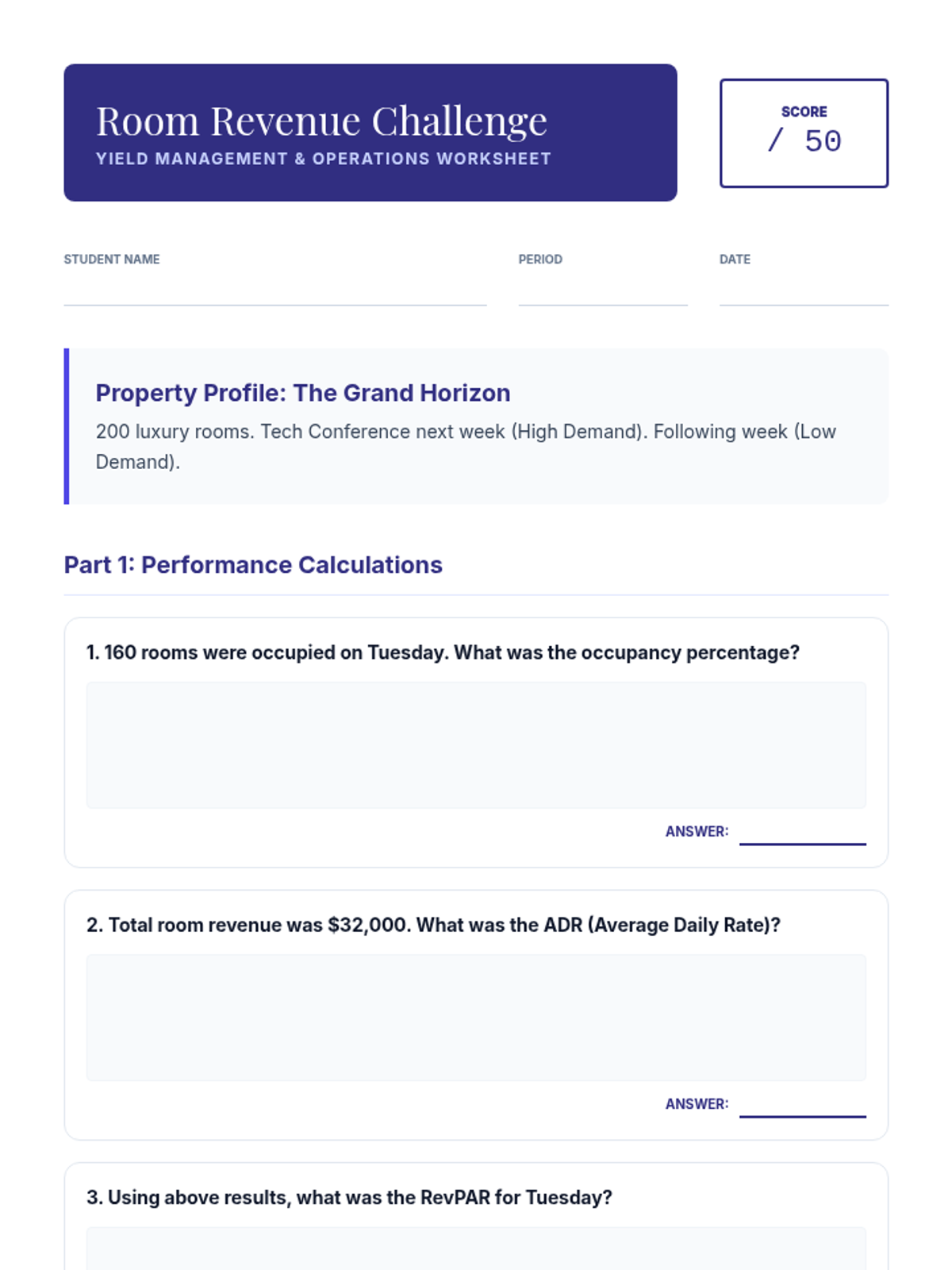
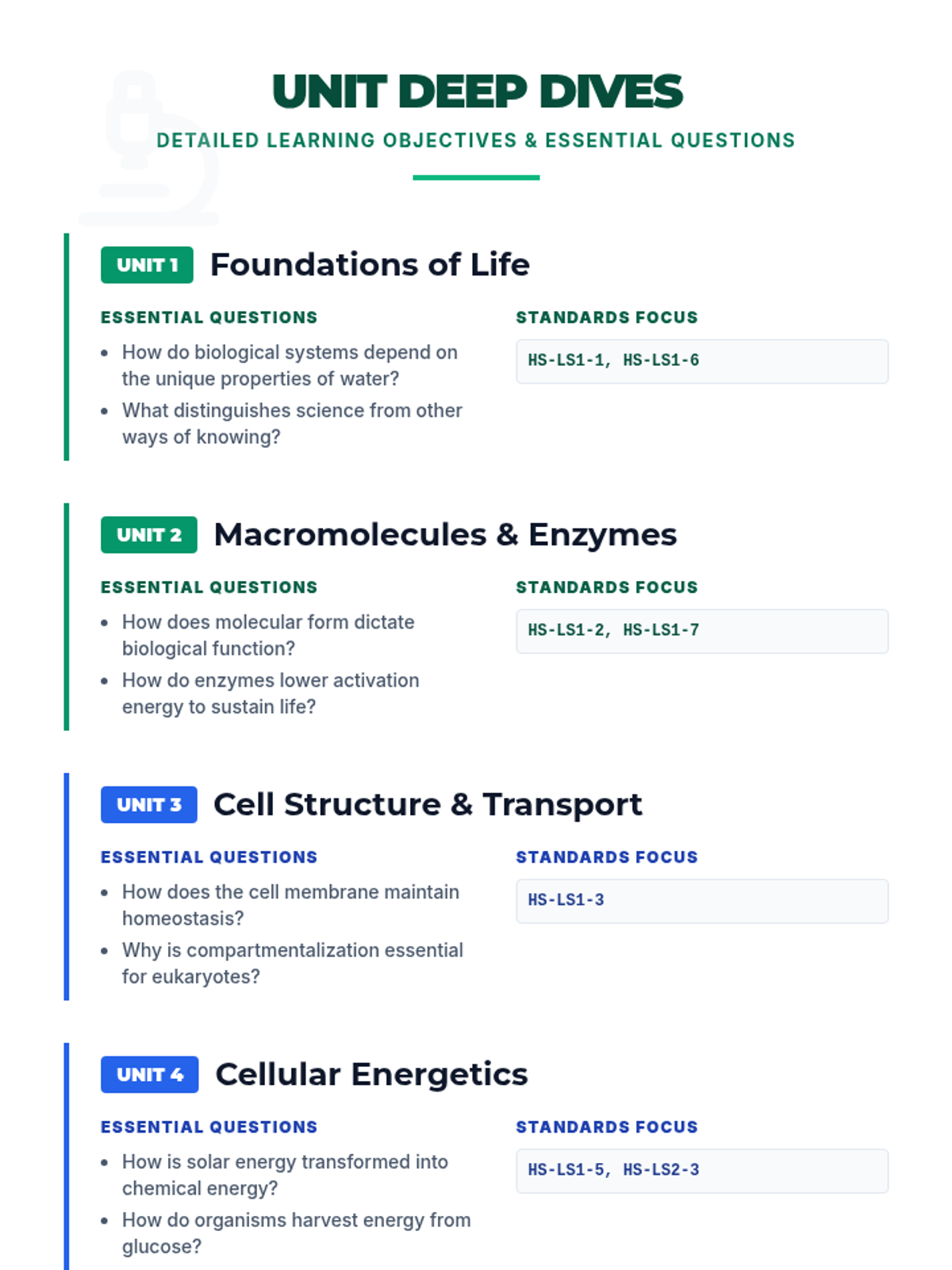
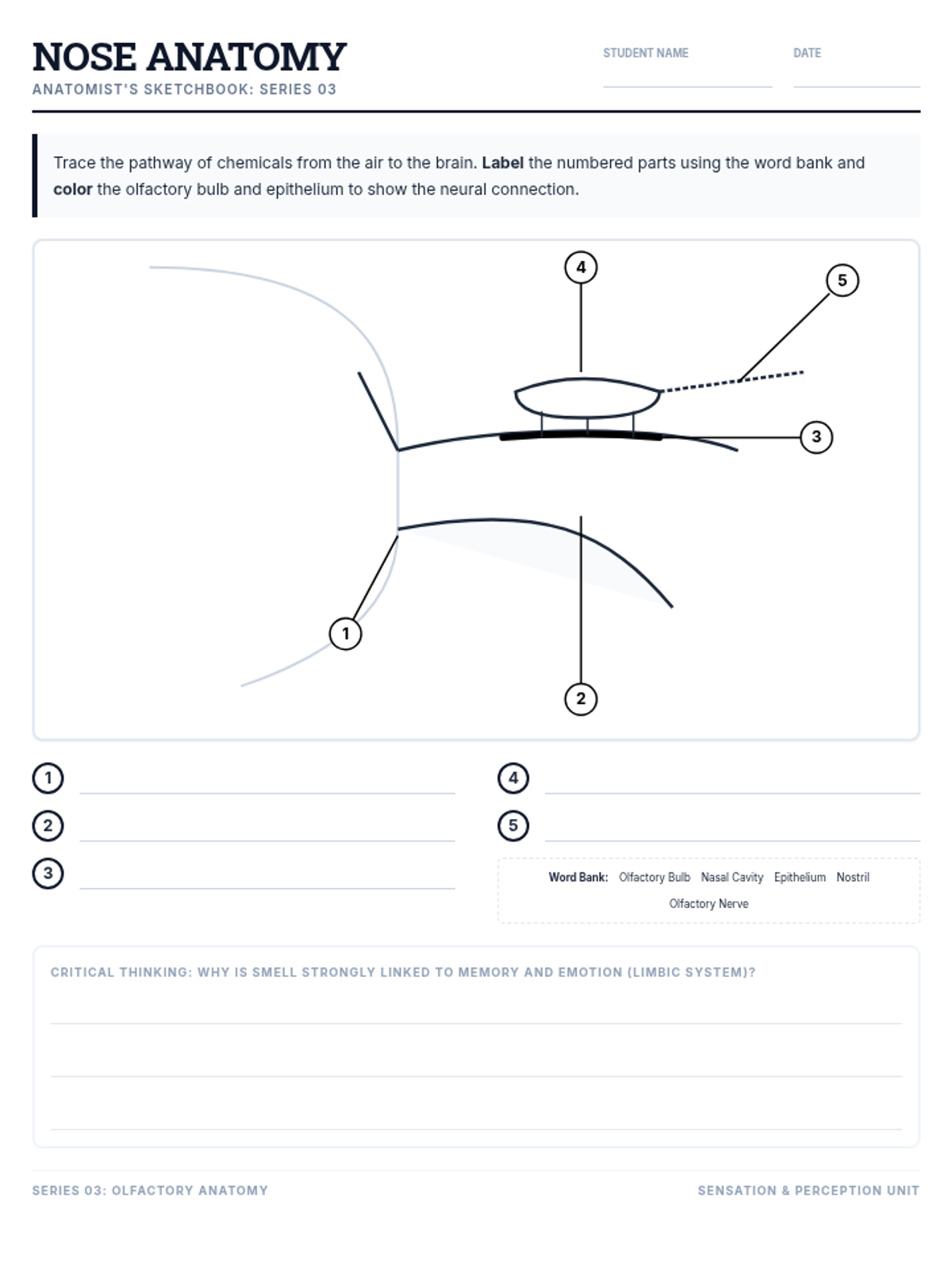
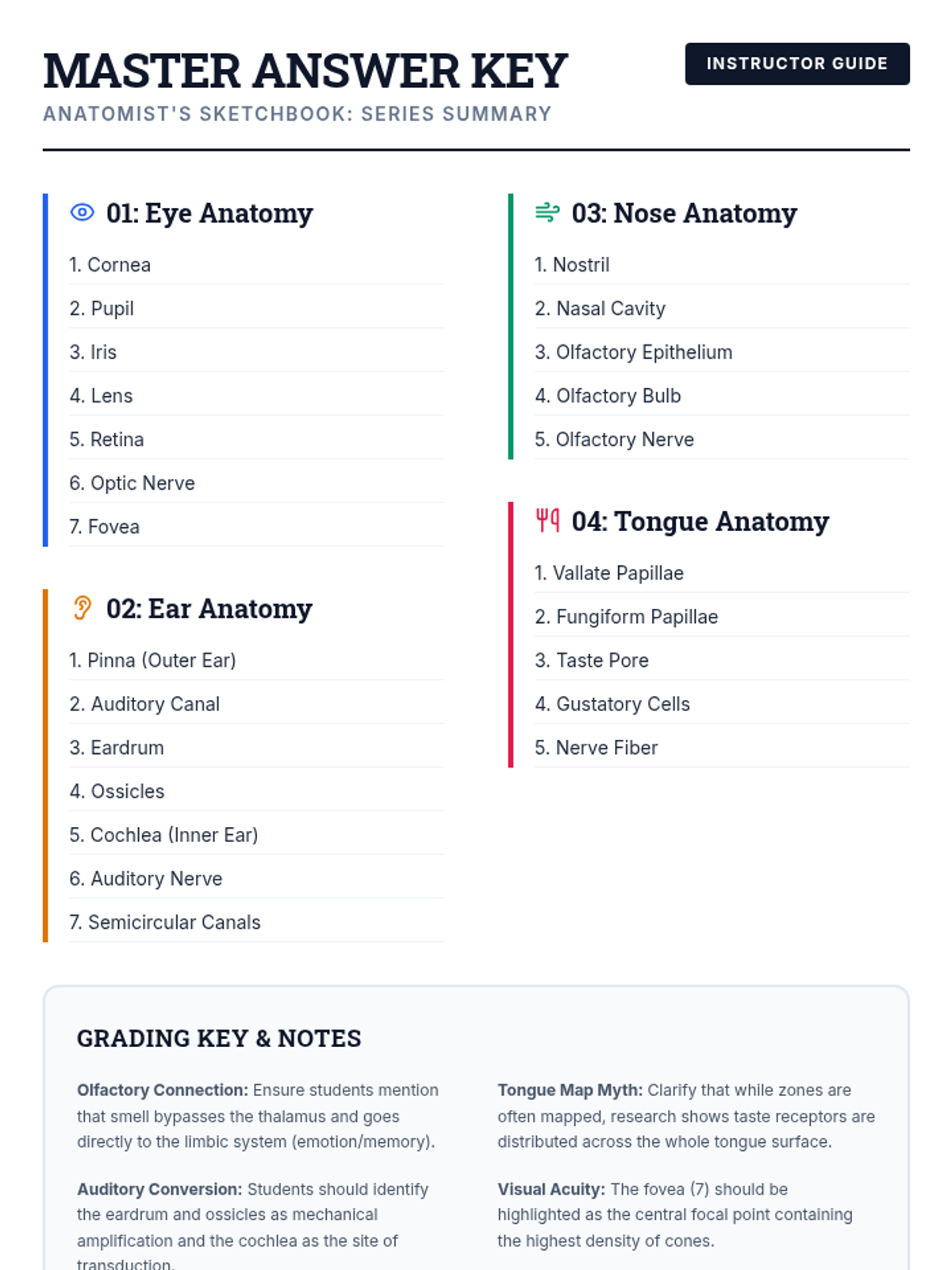
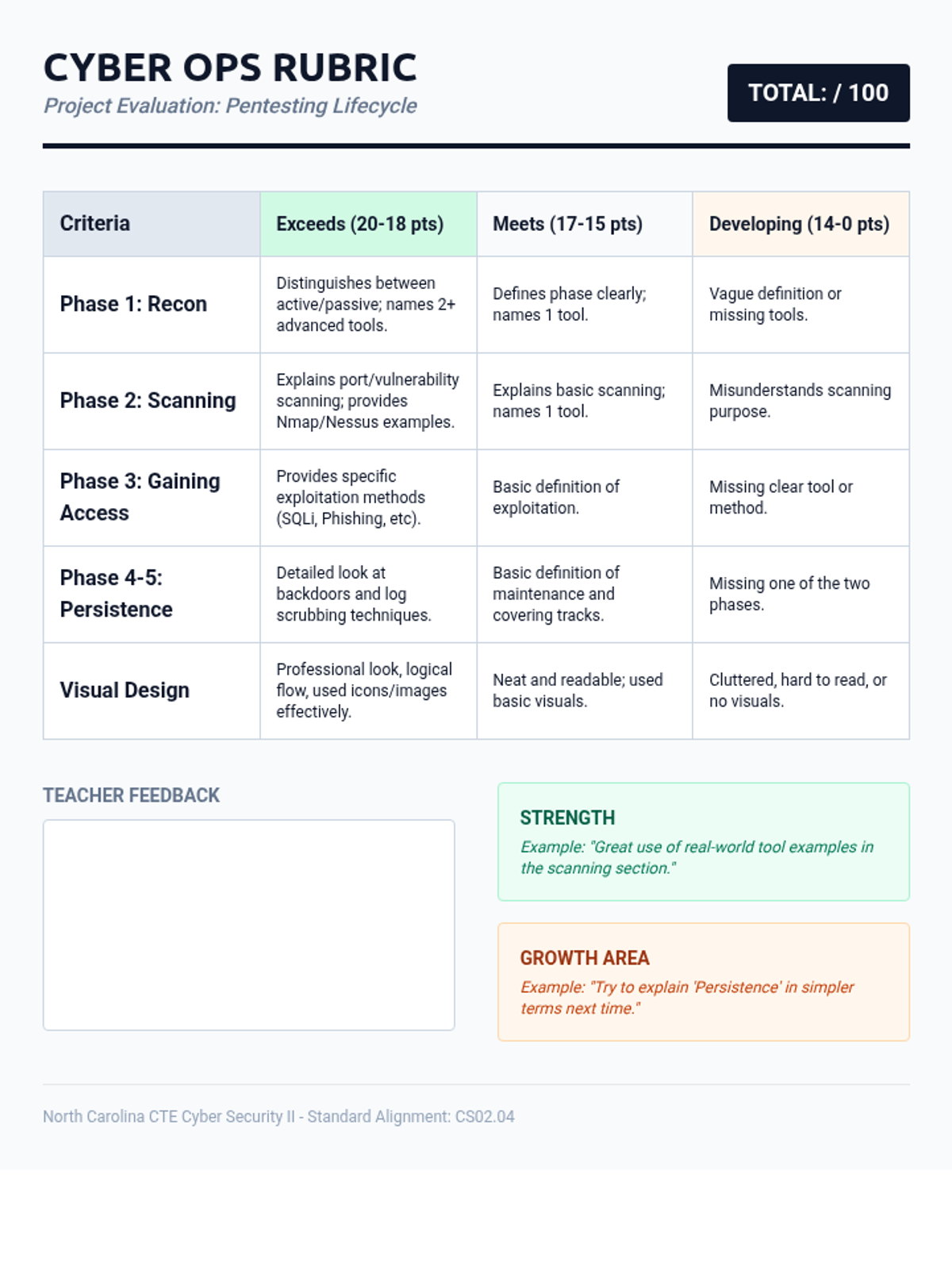
This 11th-grade chemistry sequence explores the microscopic origins of enthalpy changes by focusing on the energy required to break chemical bonds and the energy released when new bonds form. Students will transition from conceptual physical models to quantitative calculations of reaction enthalpies, eventually analyzing real-world fuels and the limitations of average bond energy data.