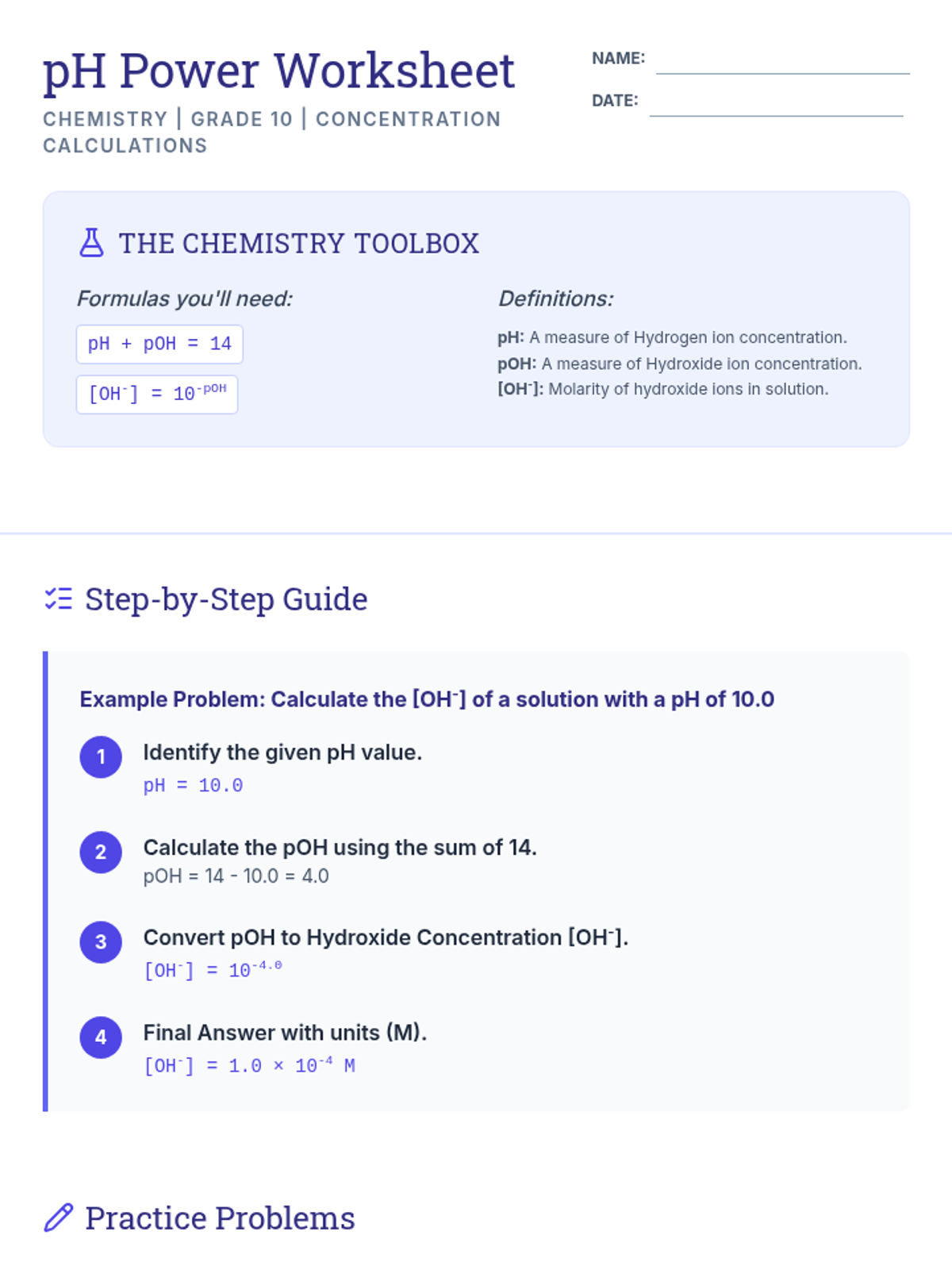
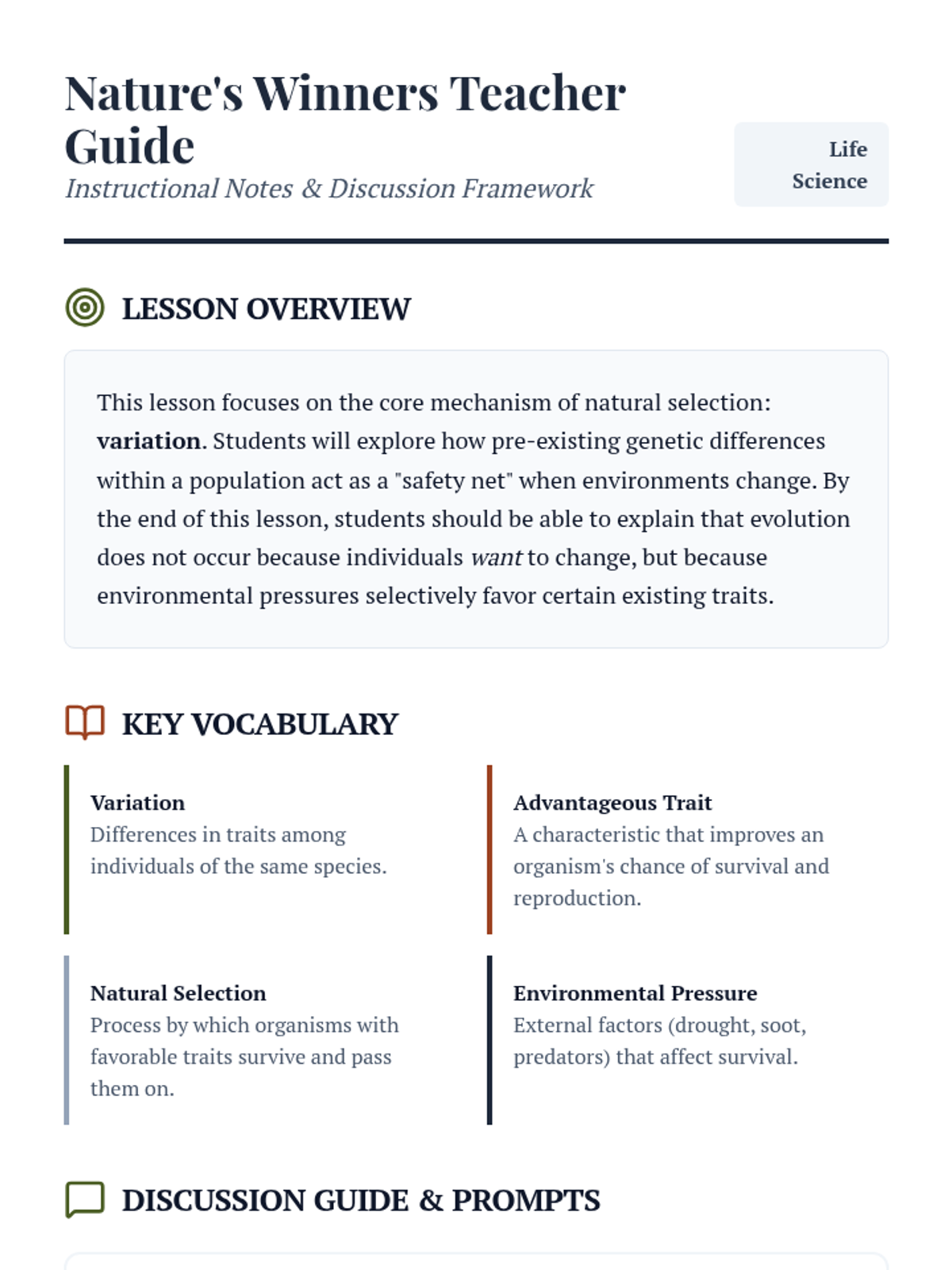
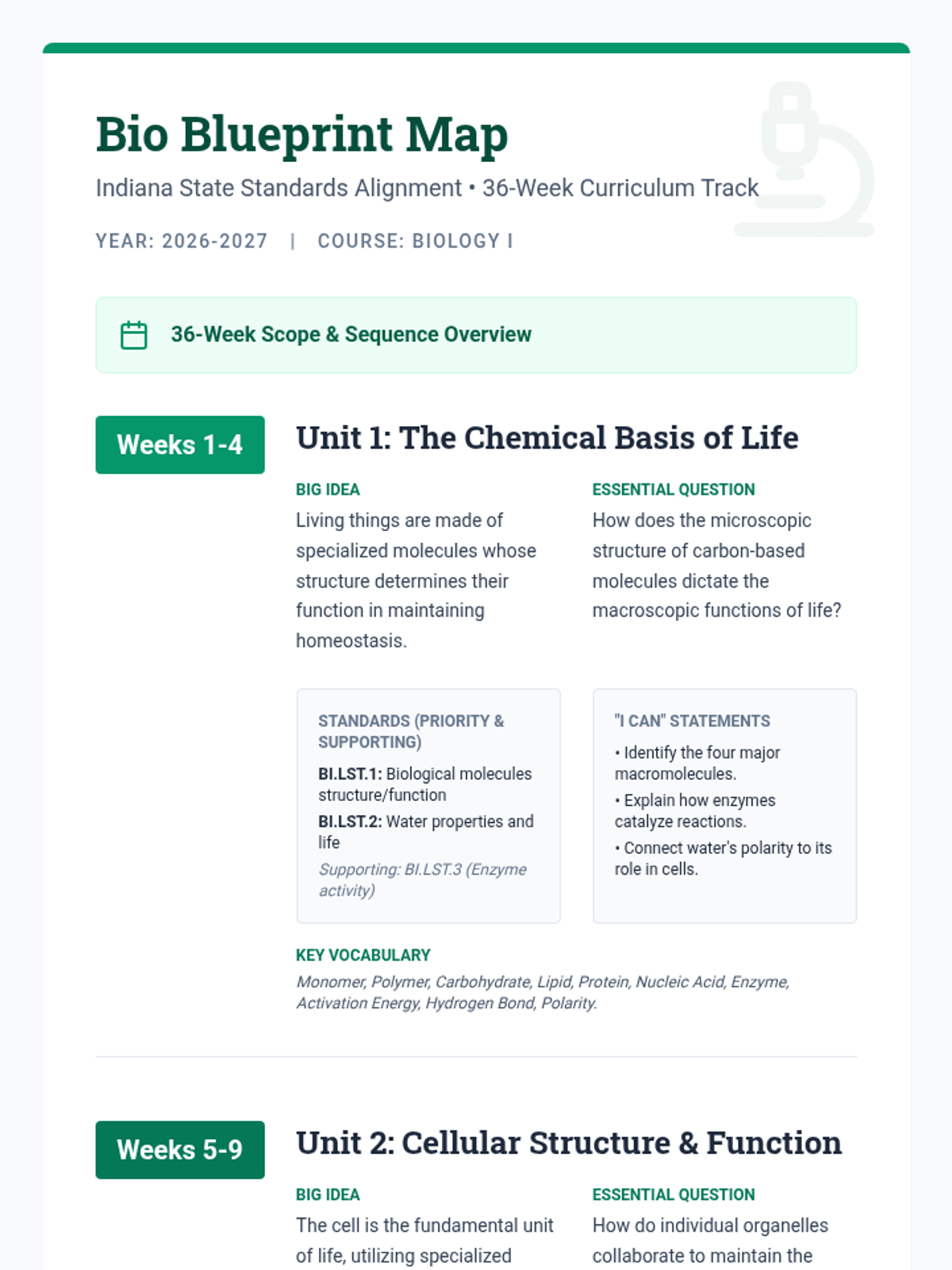
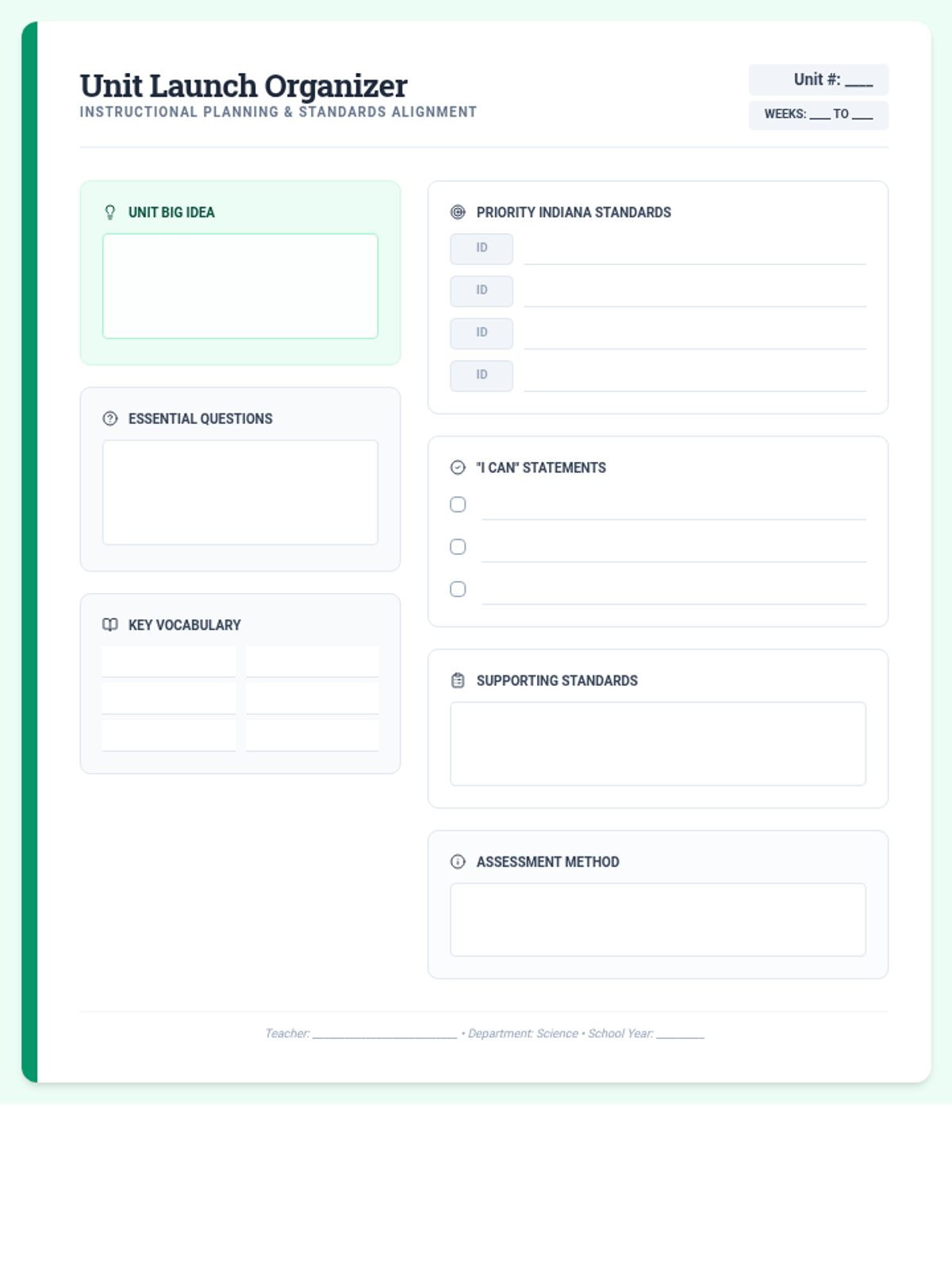
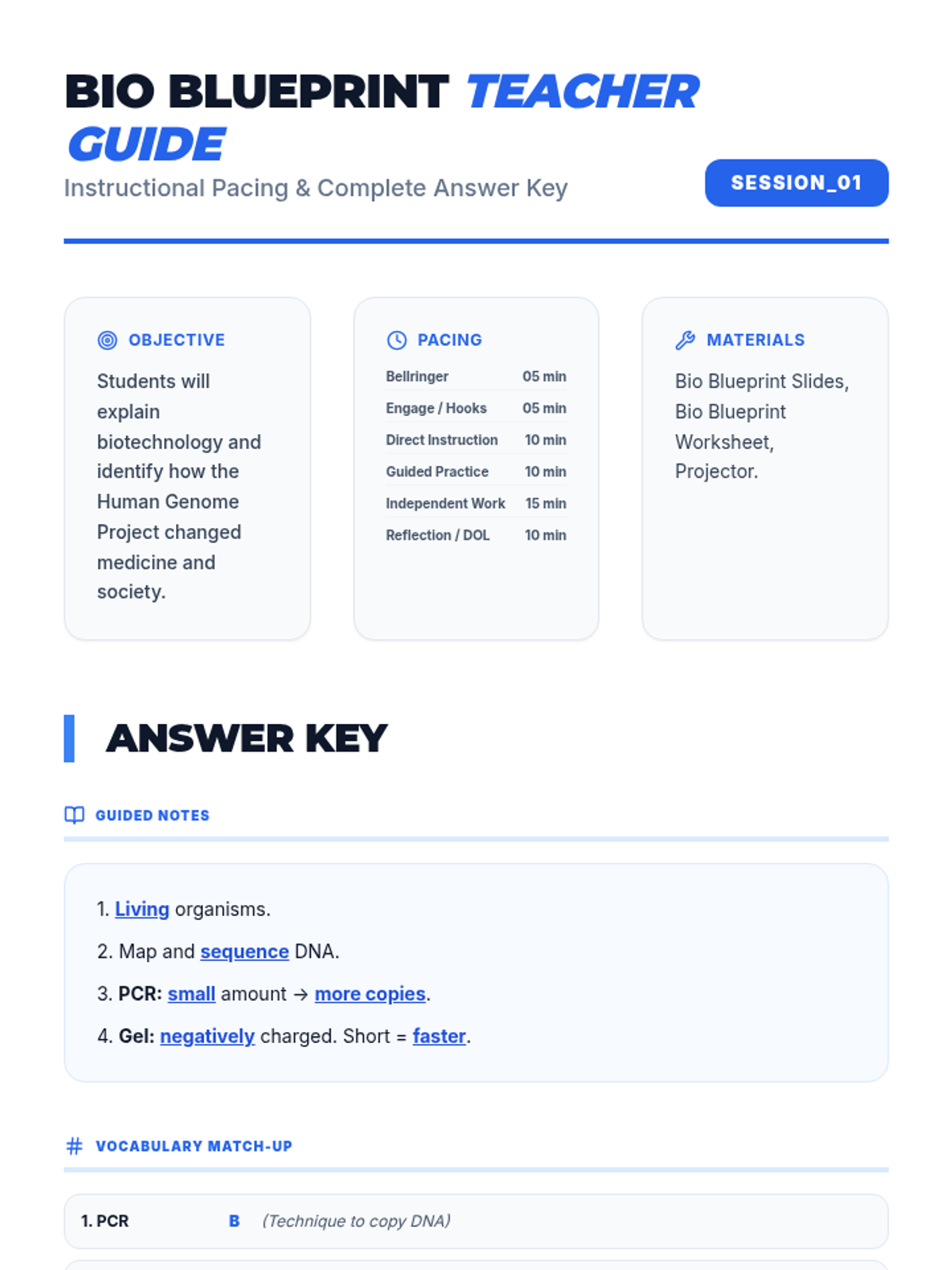
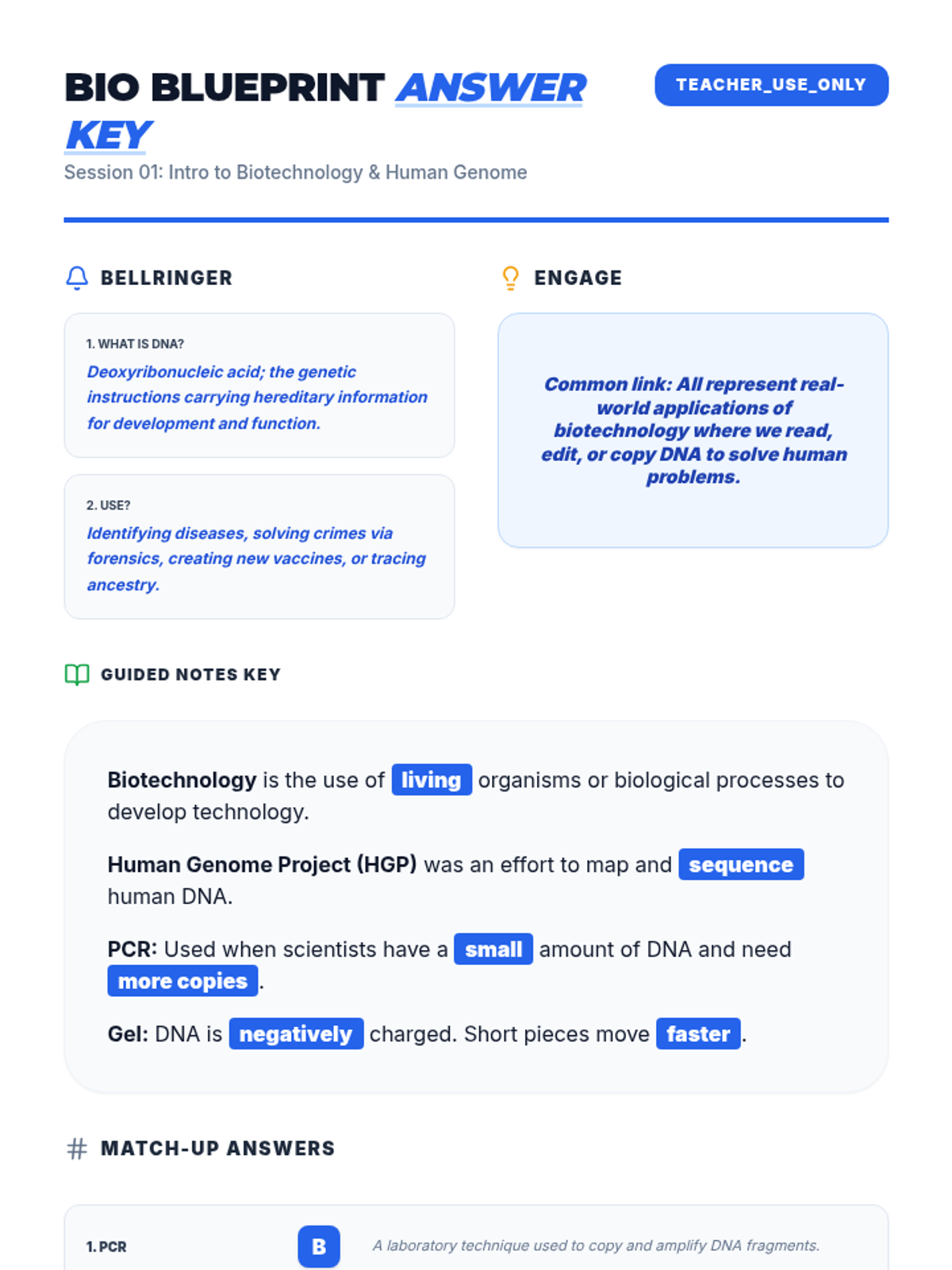
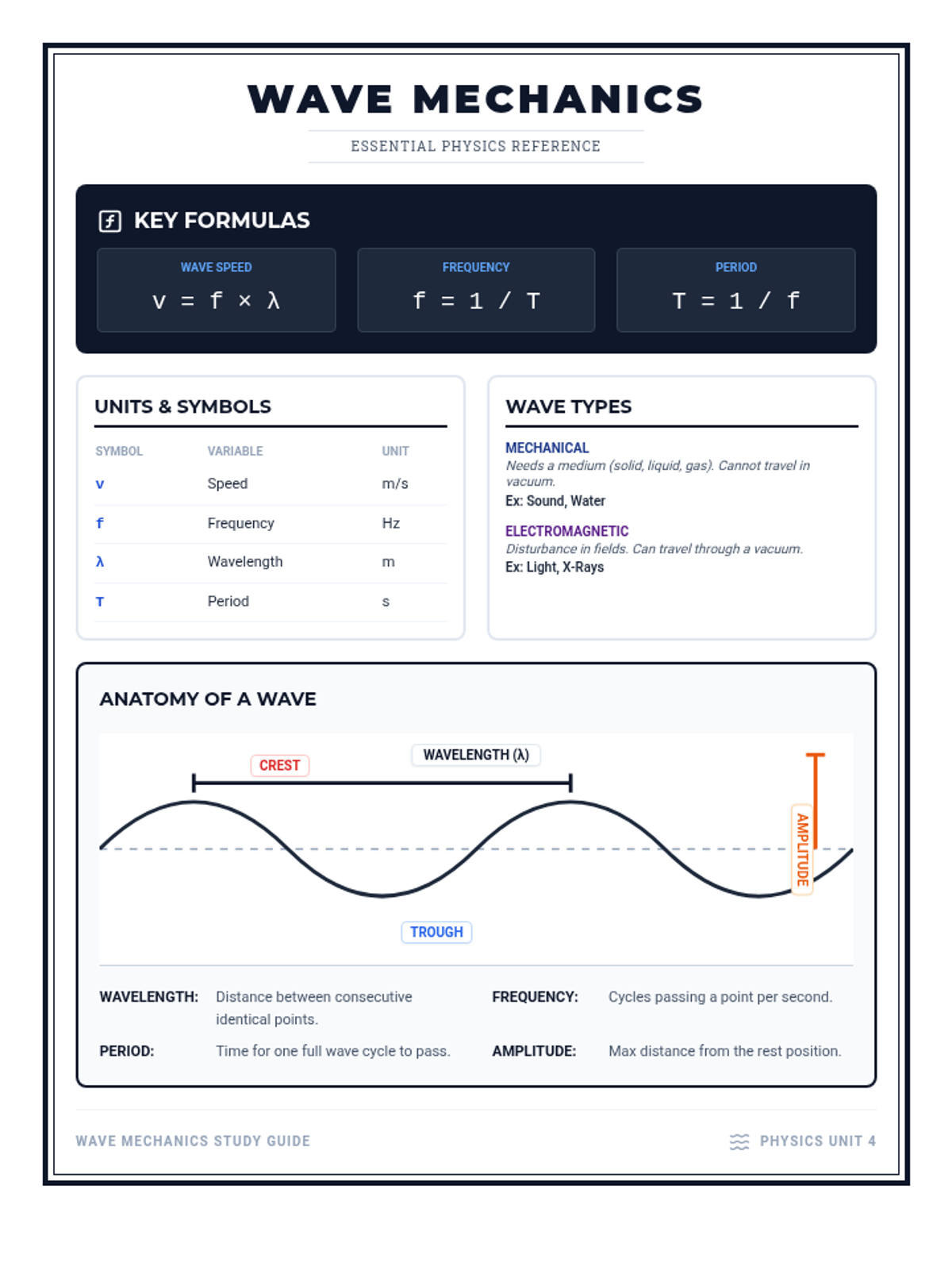
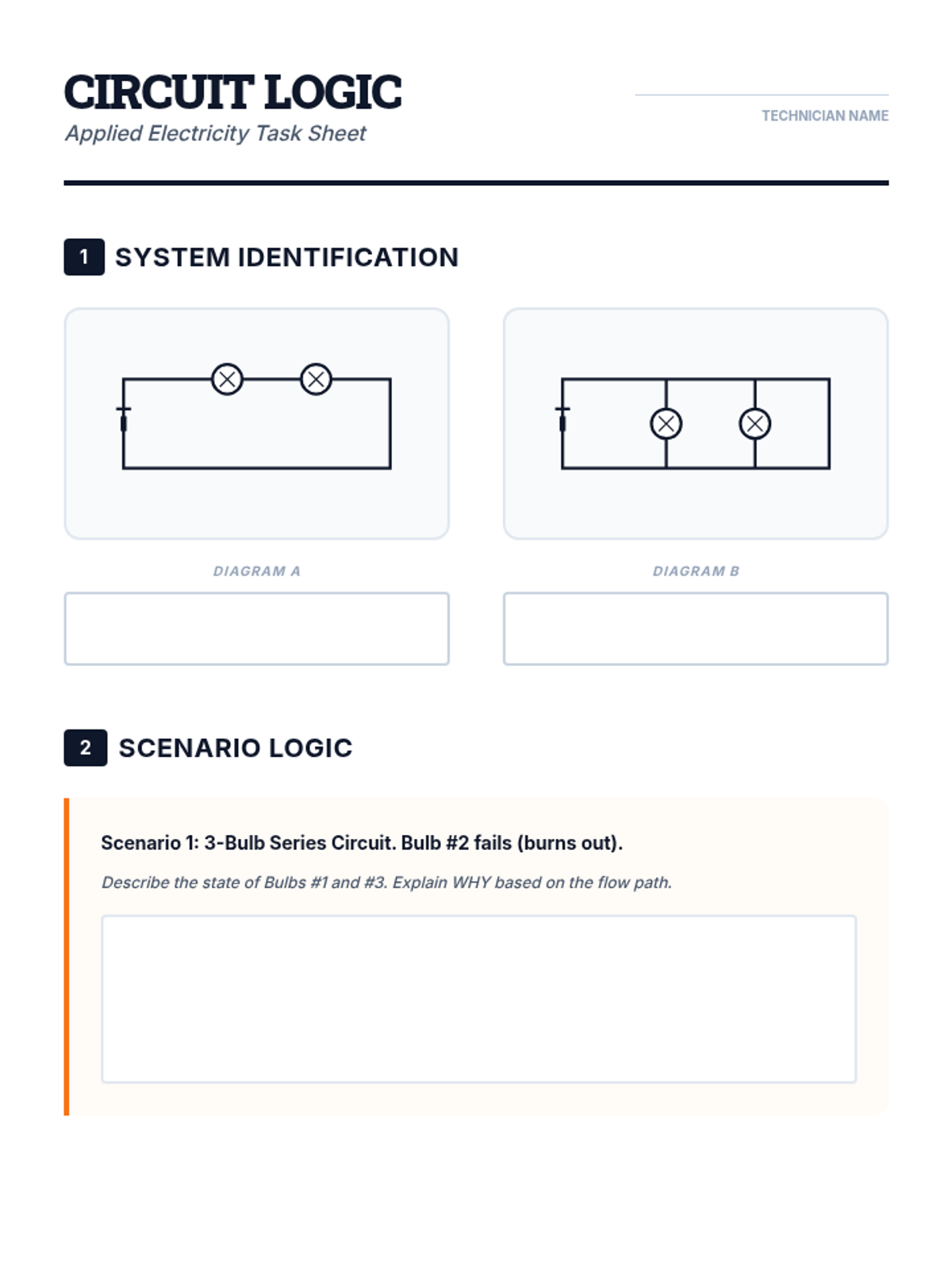
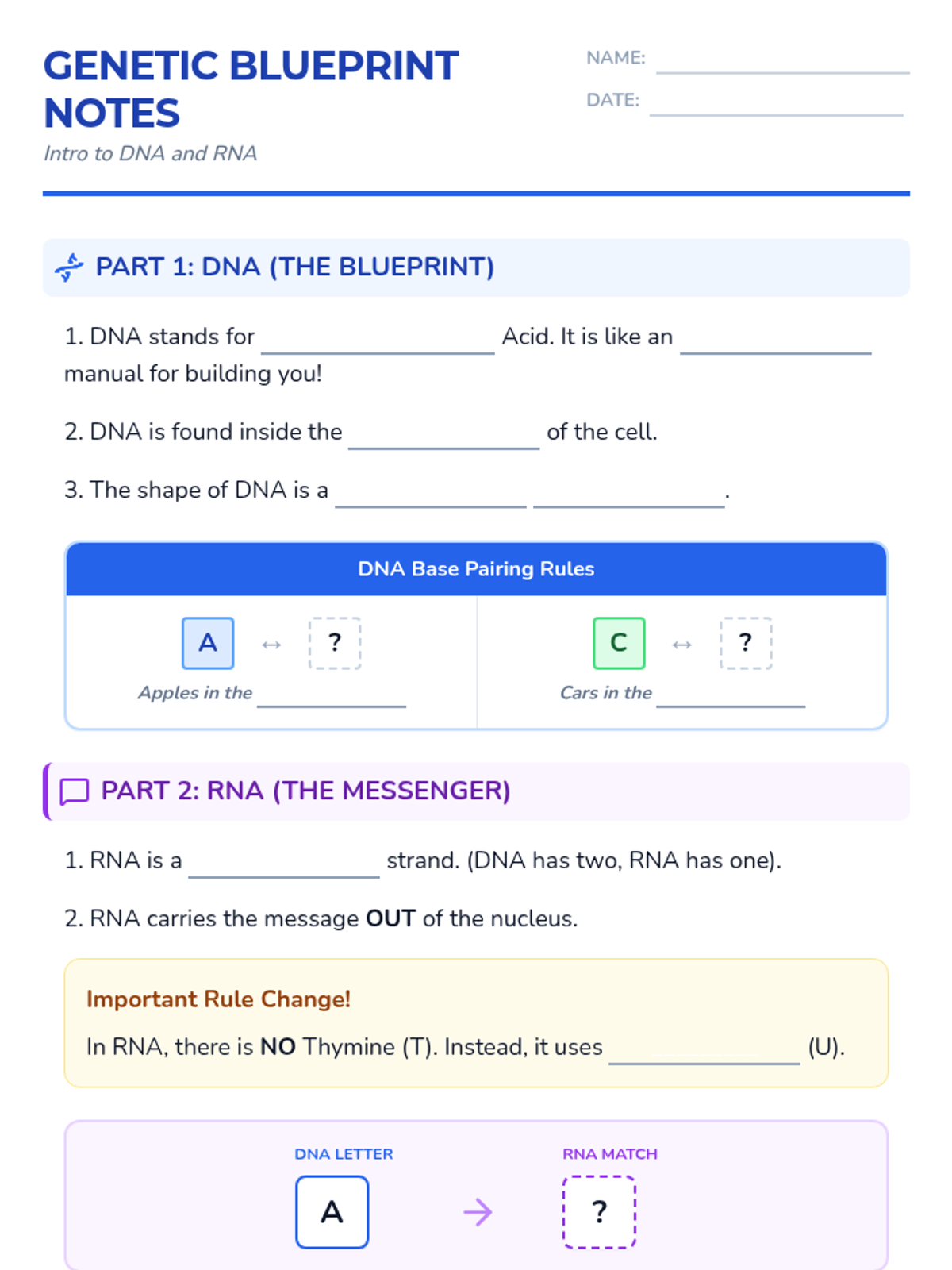
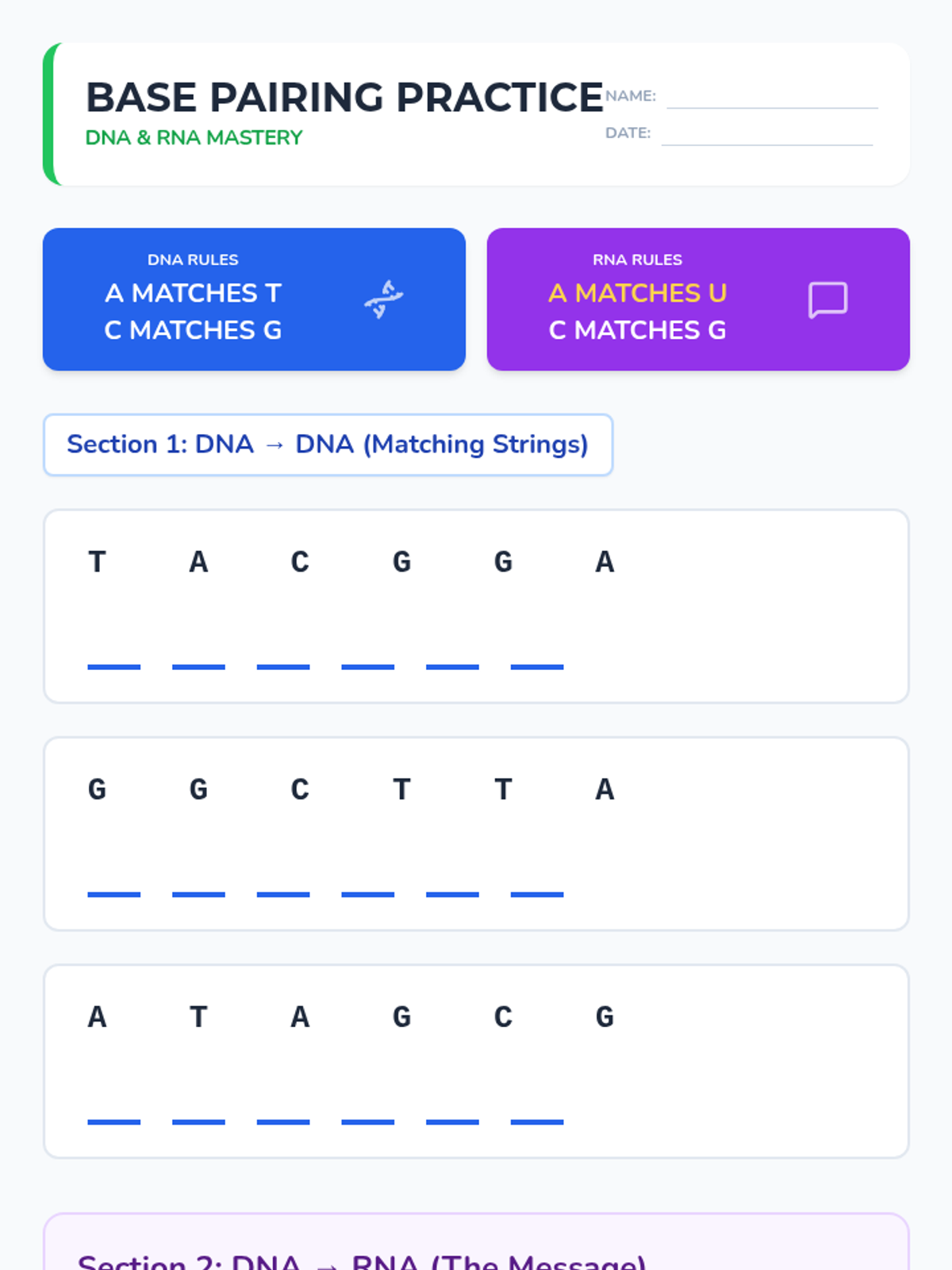
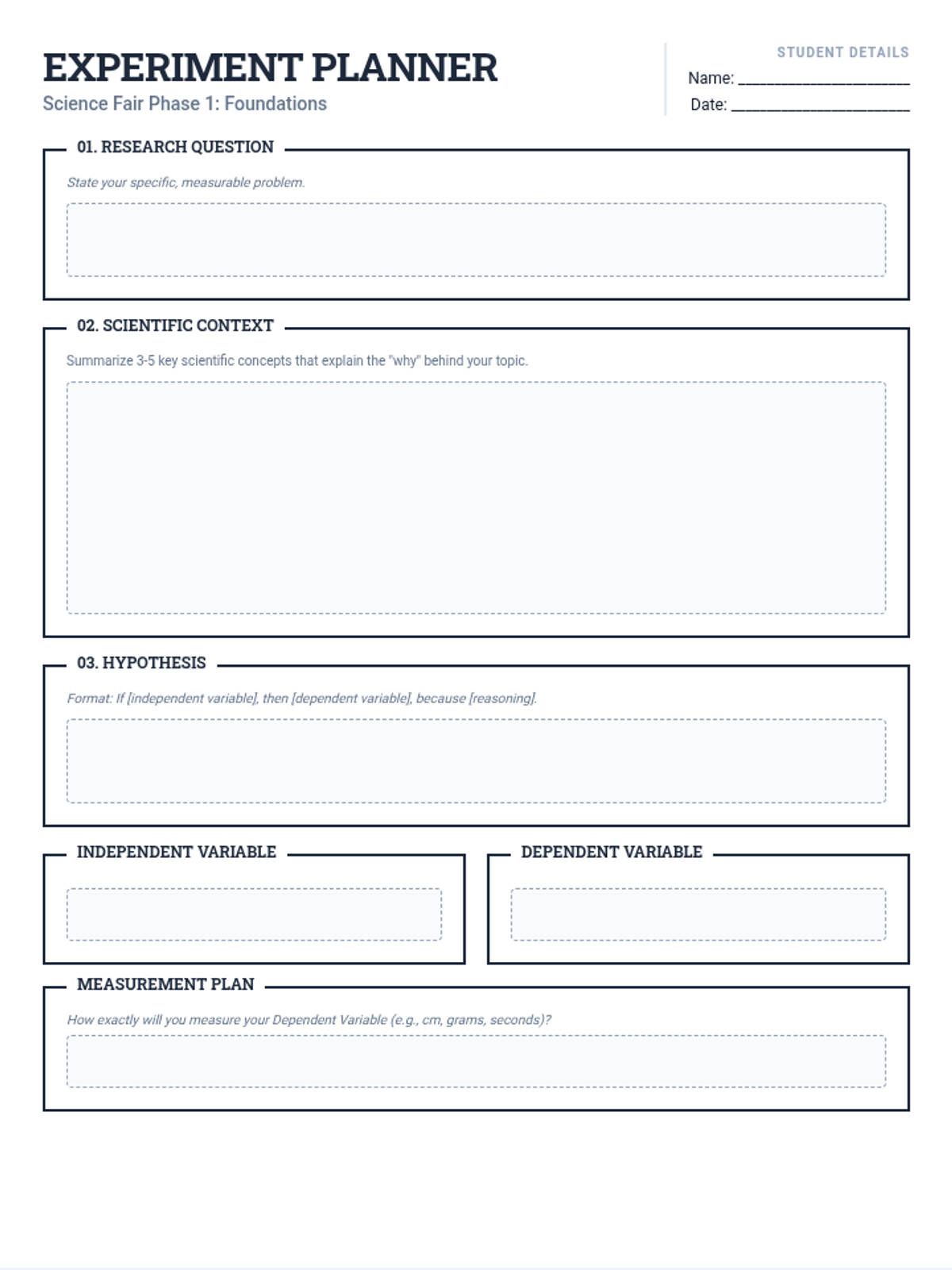
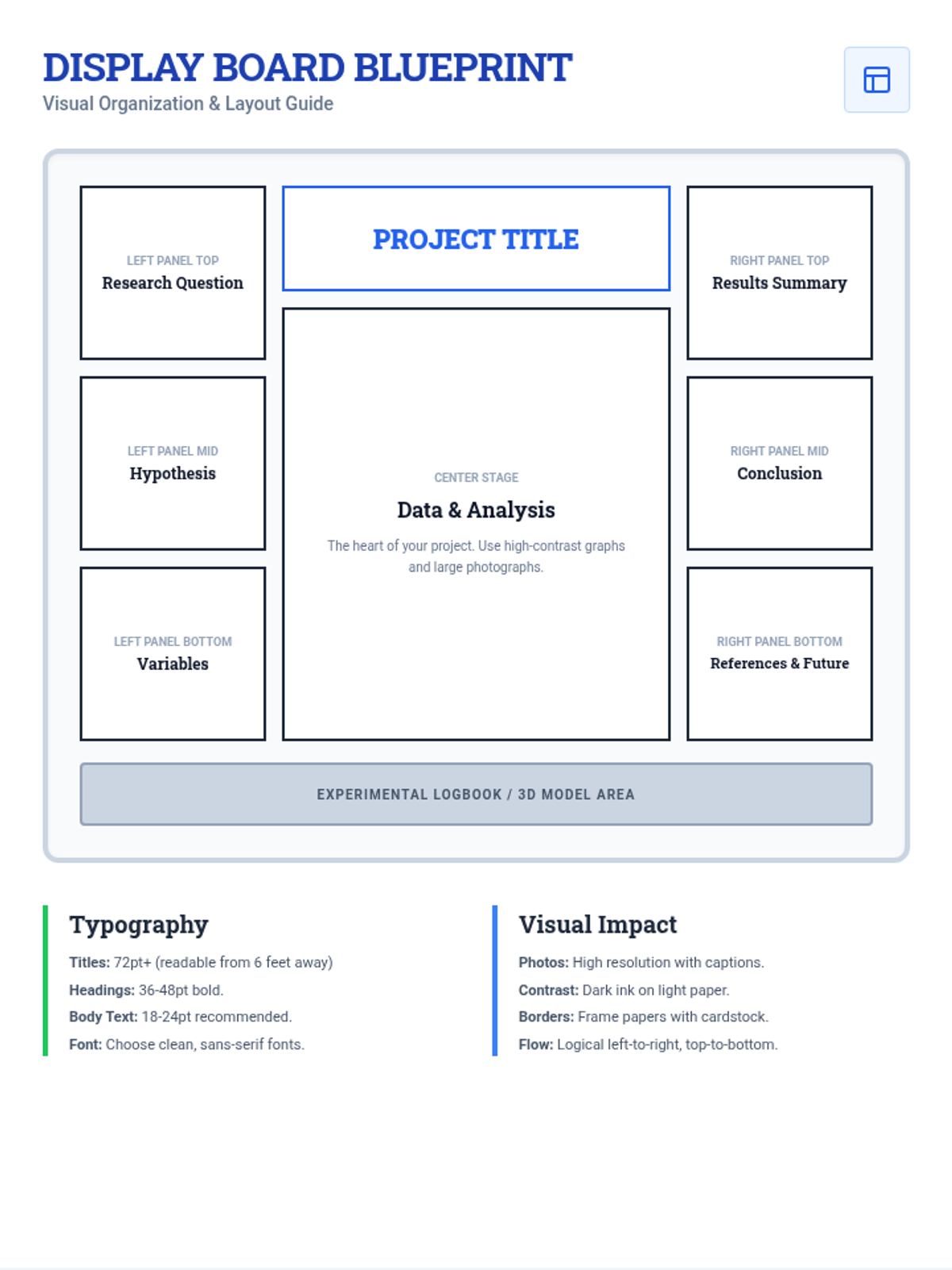
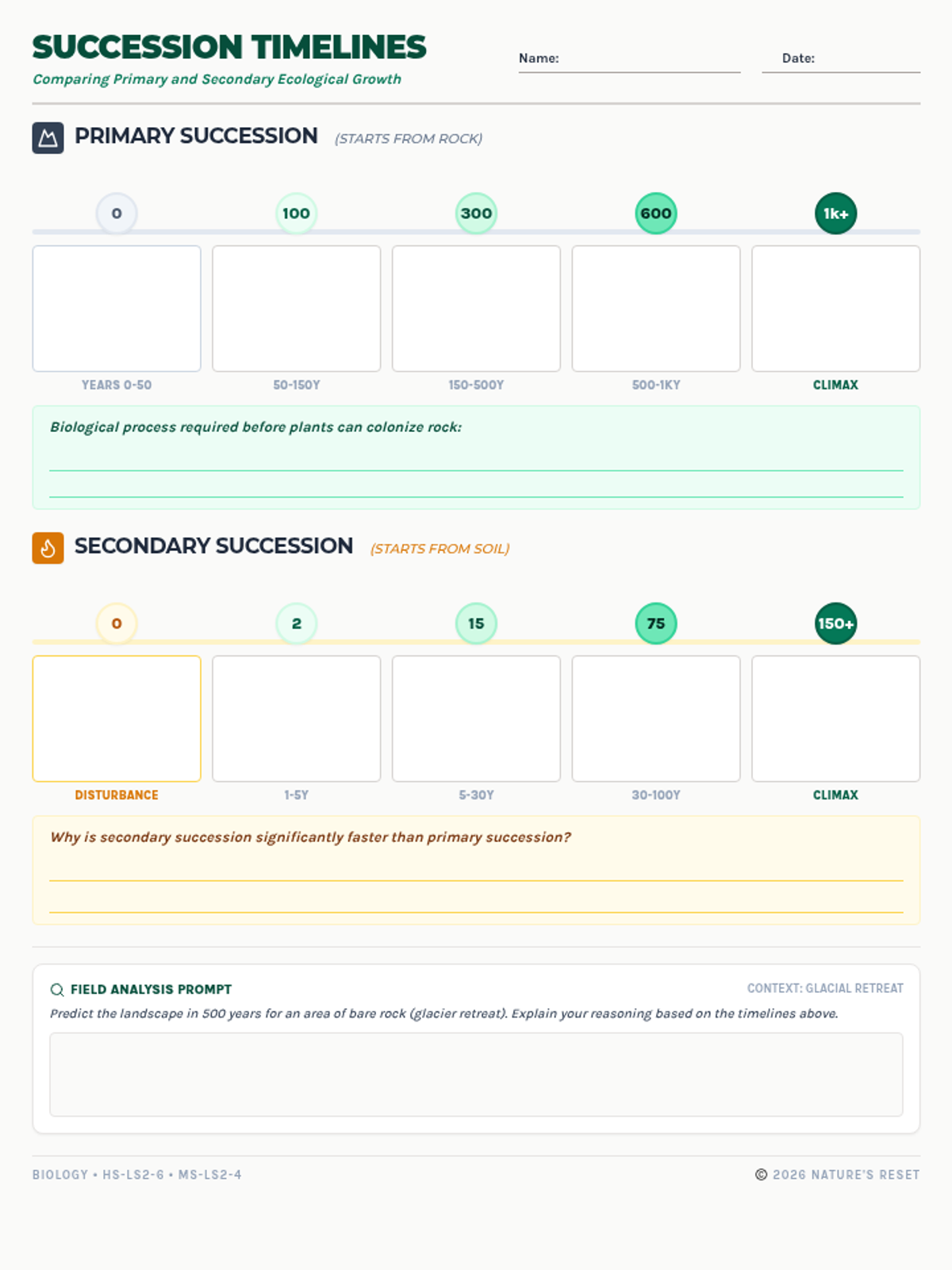
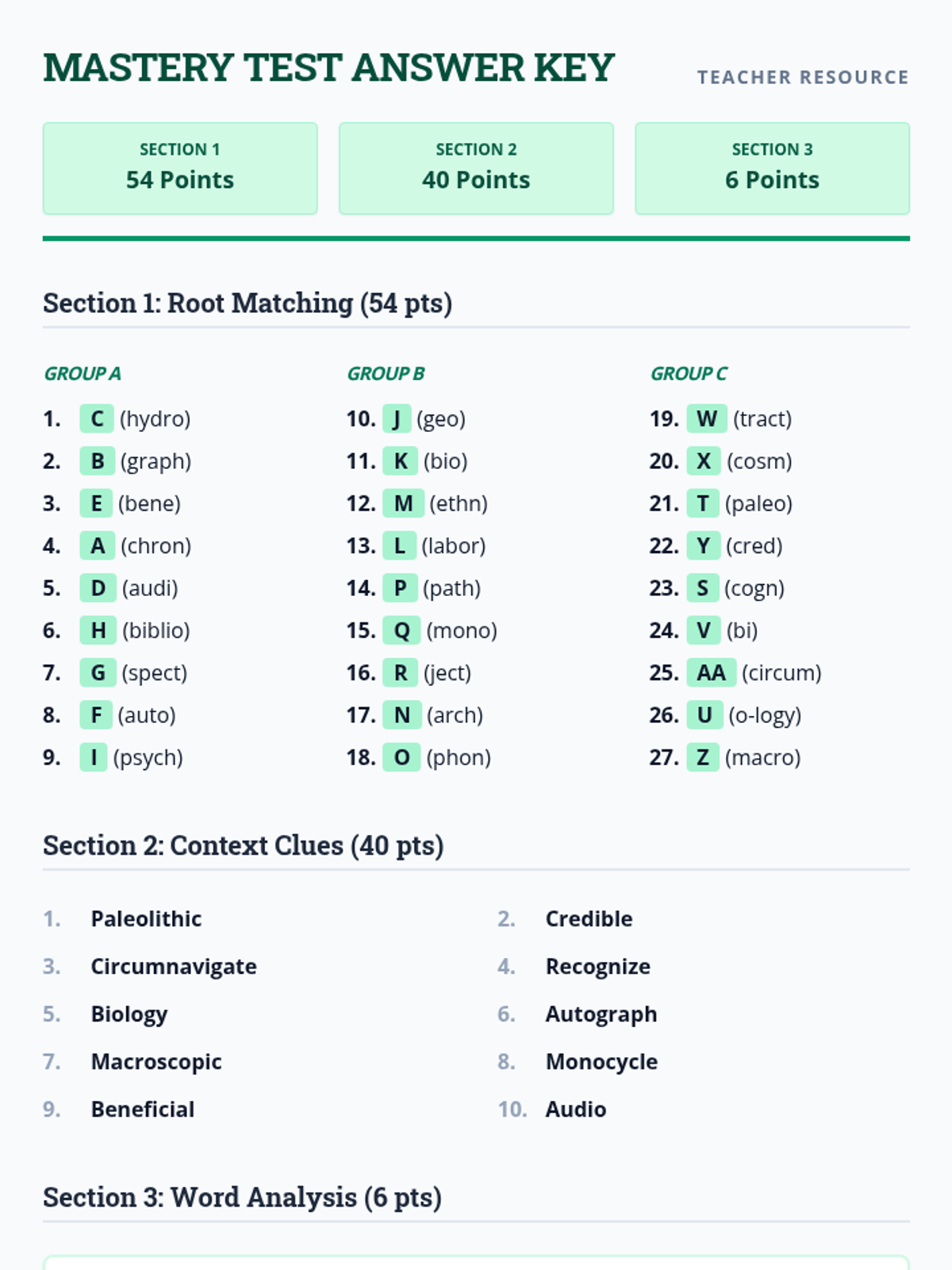
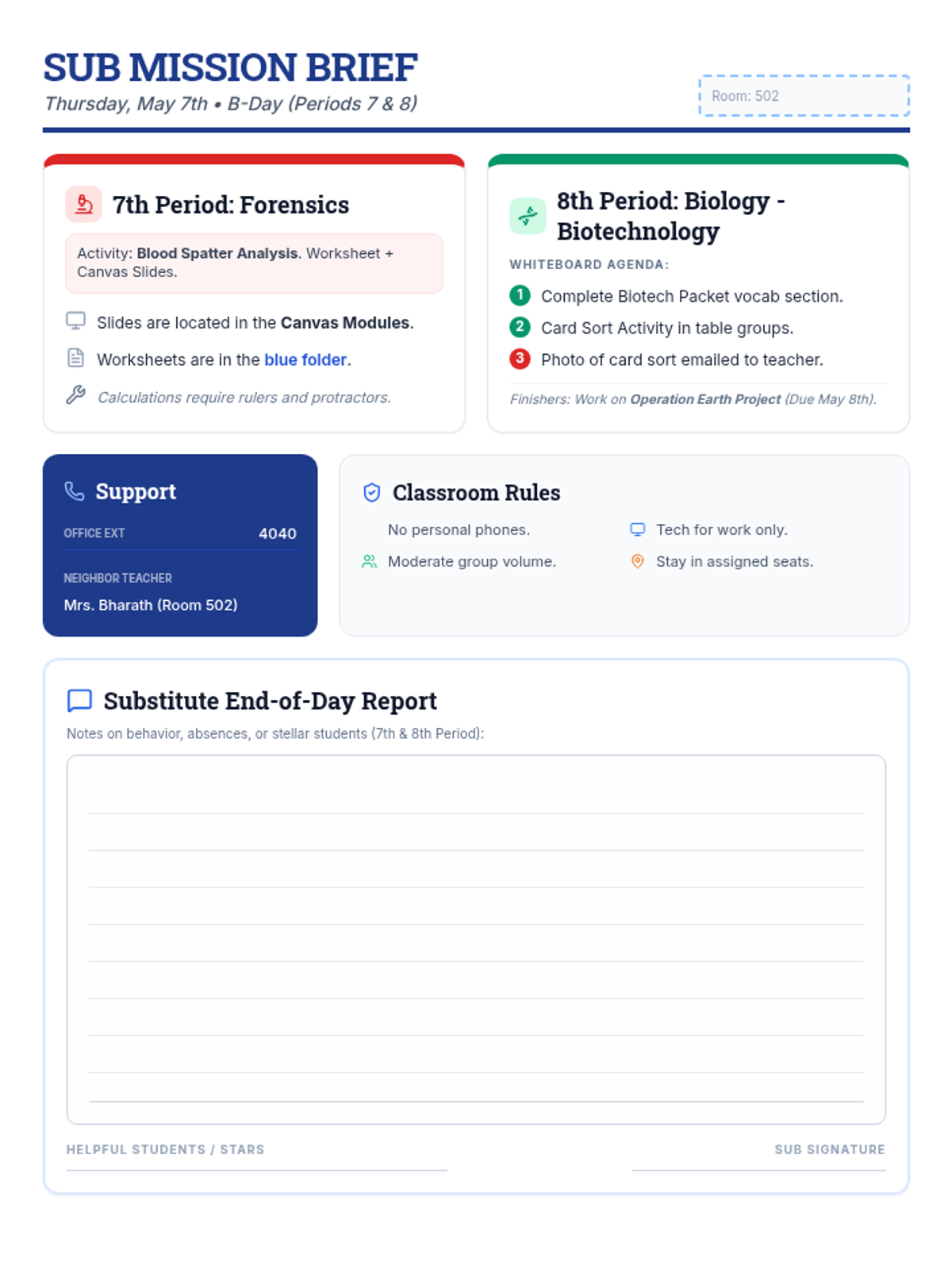
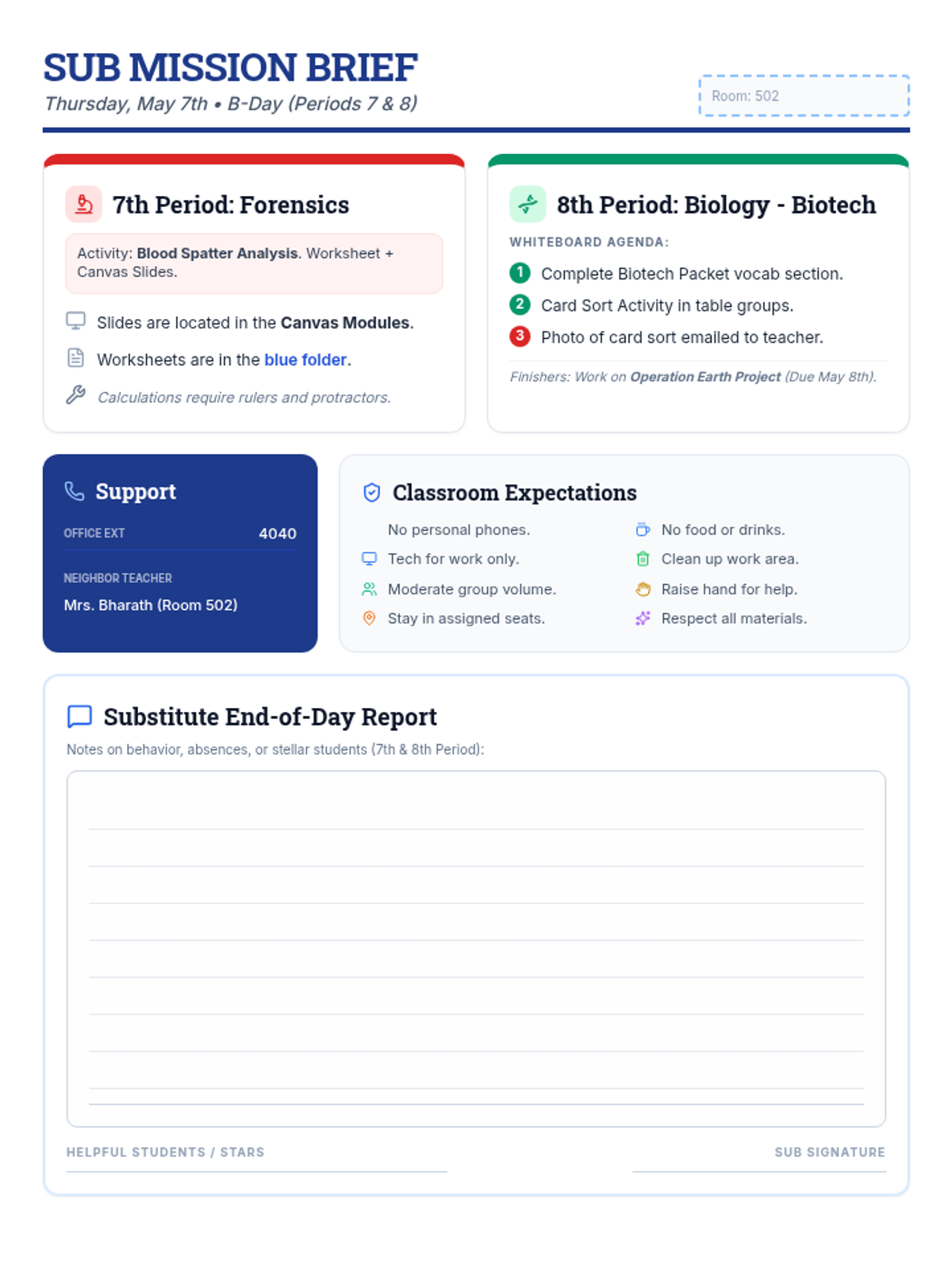
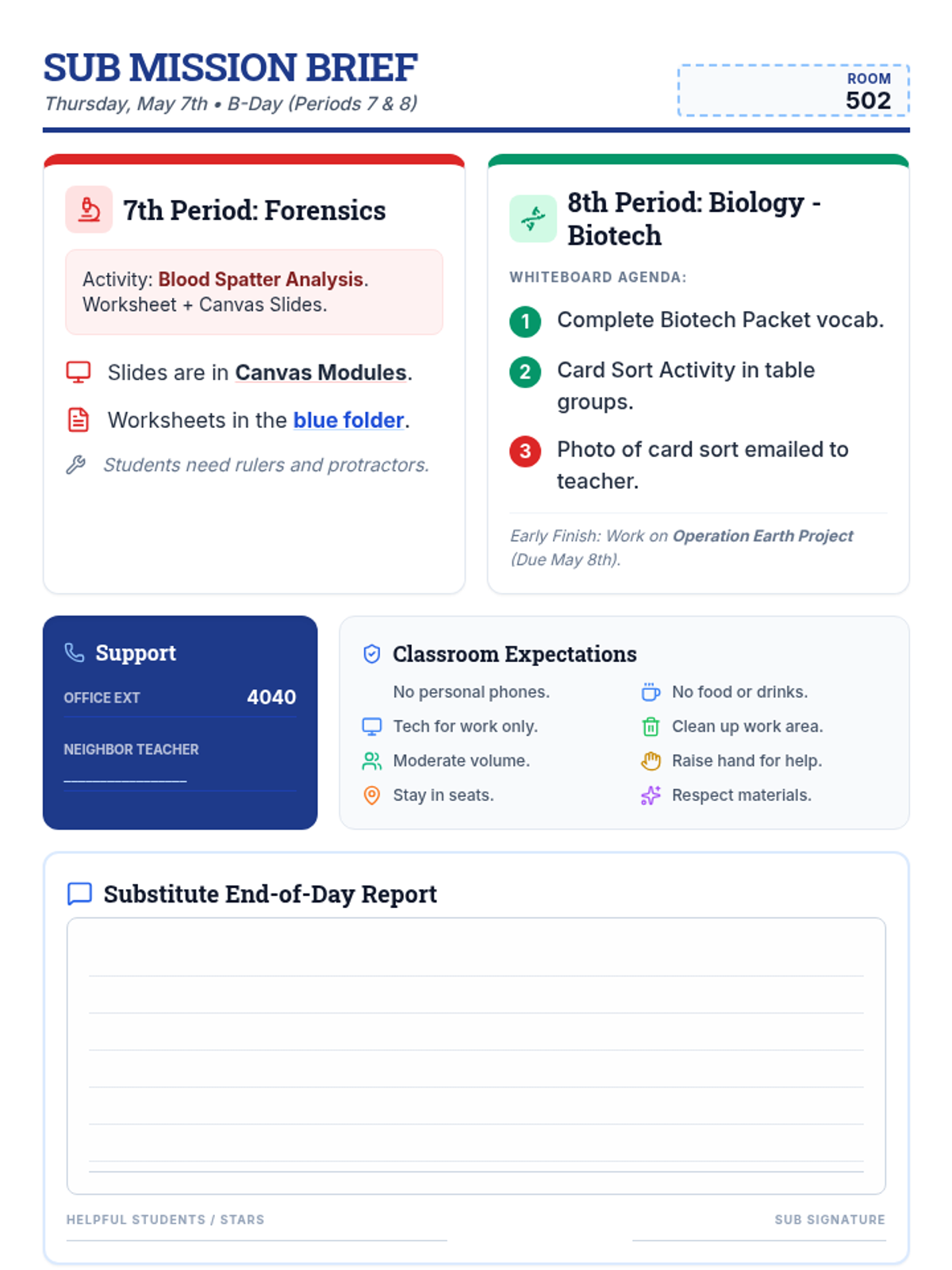
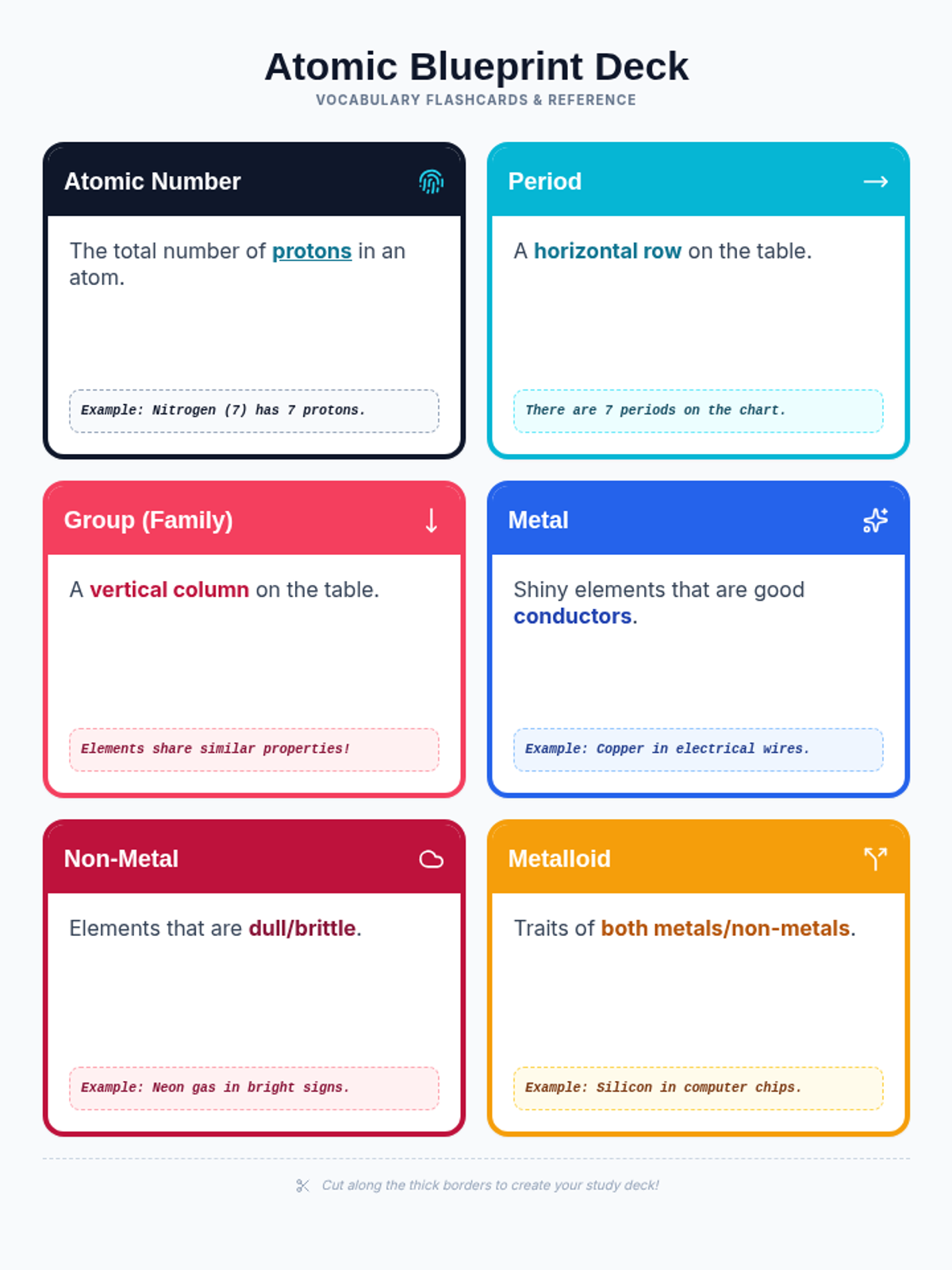
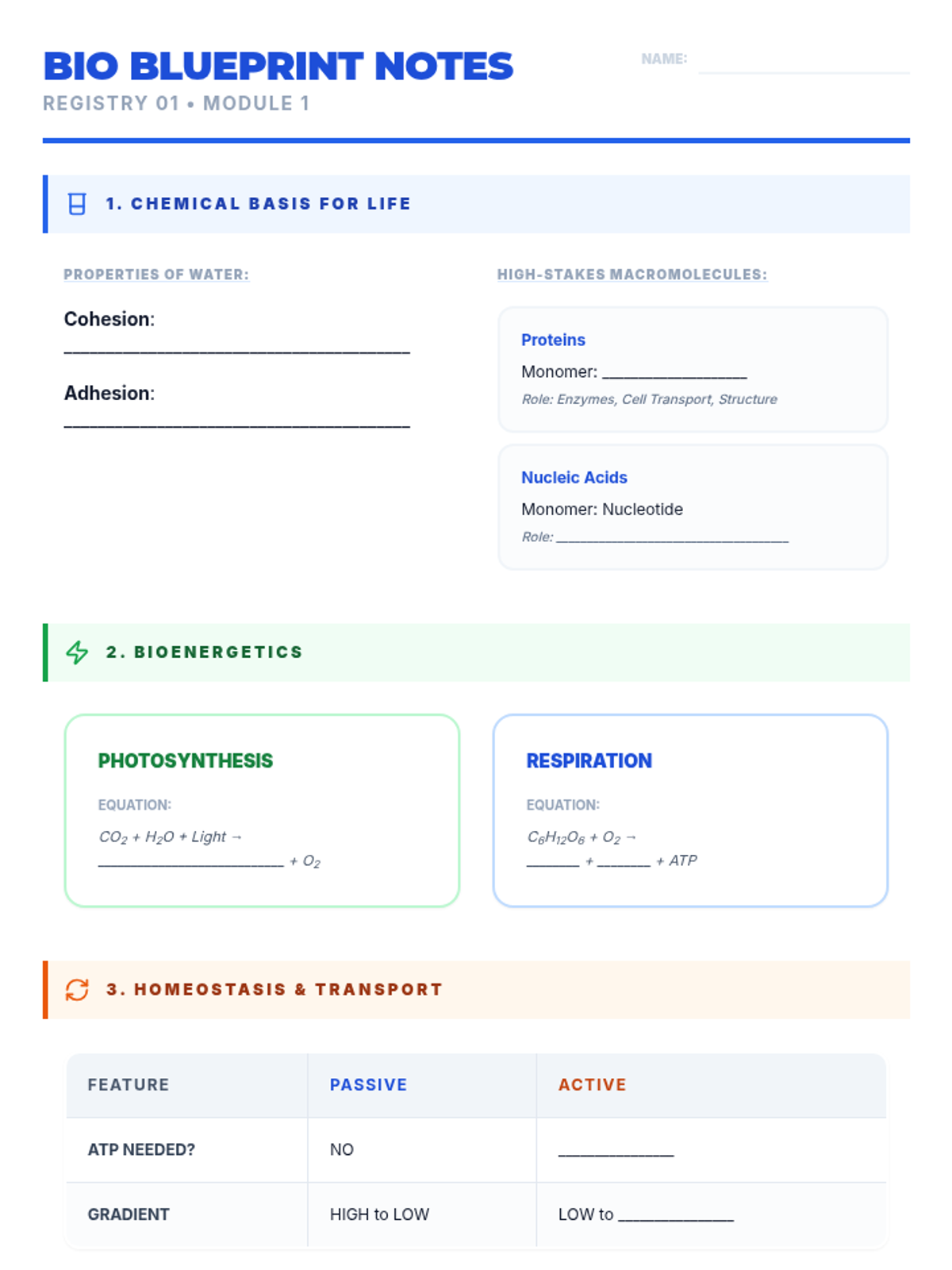
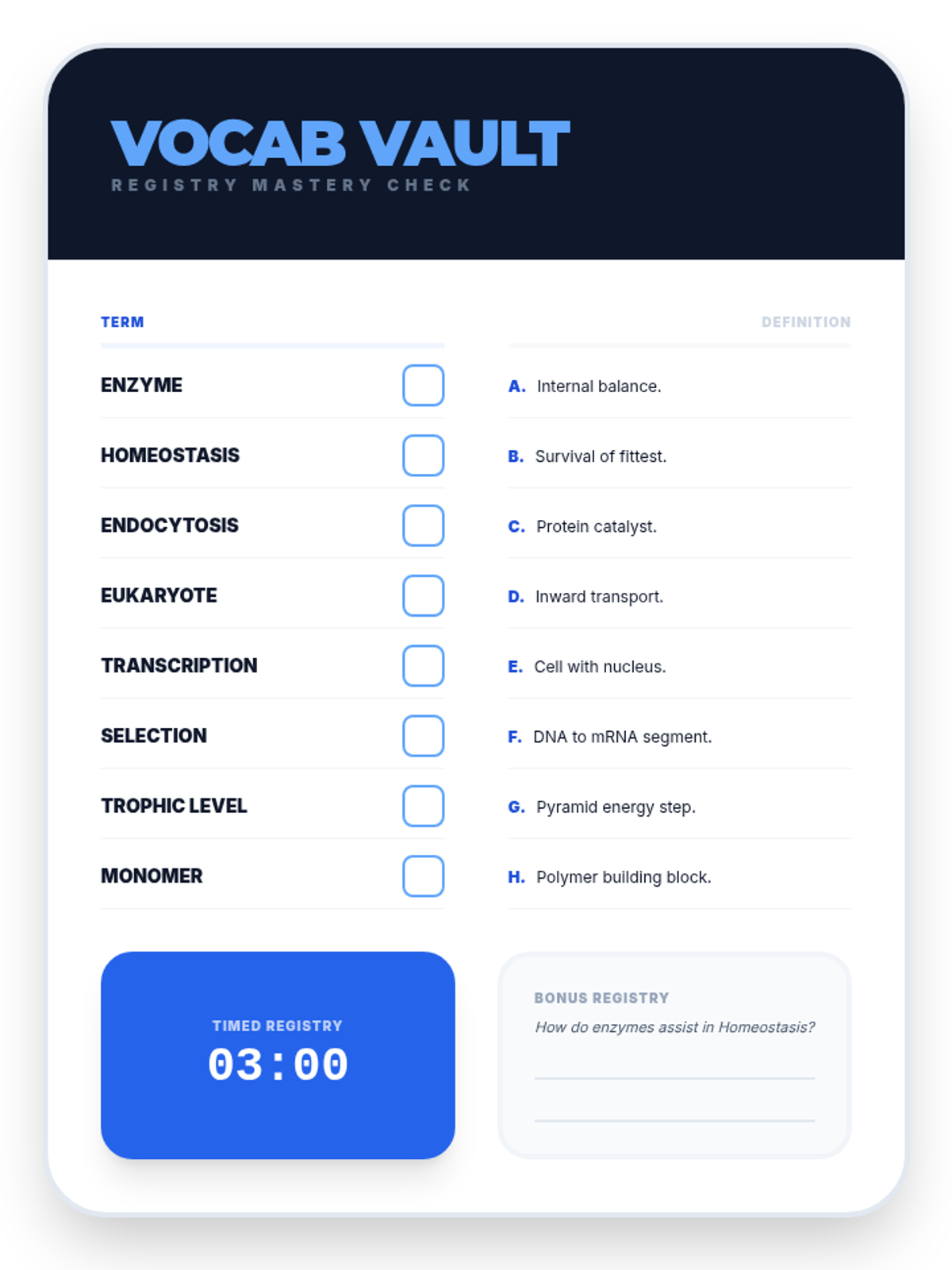
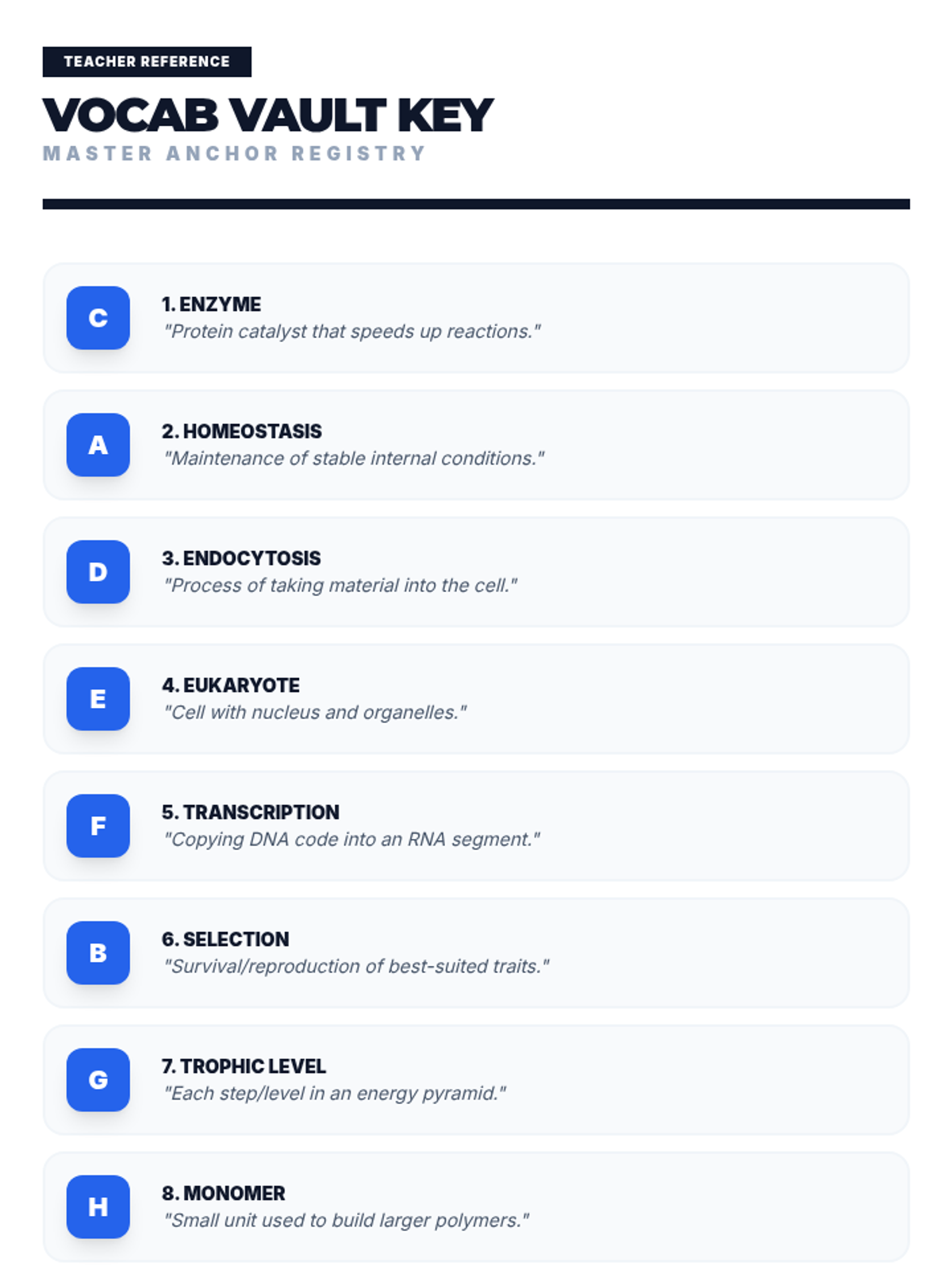
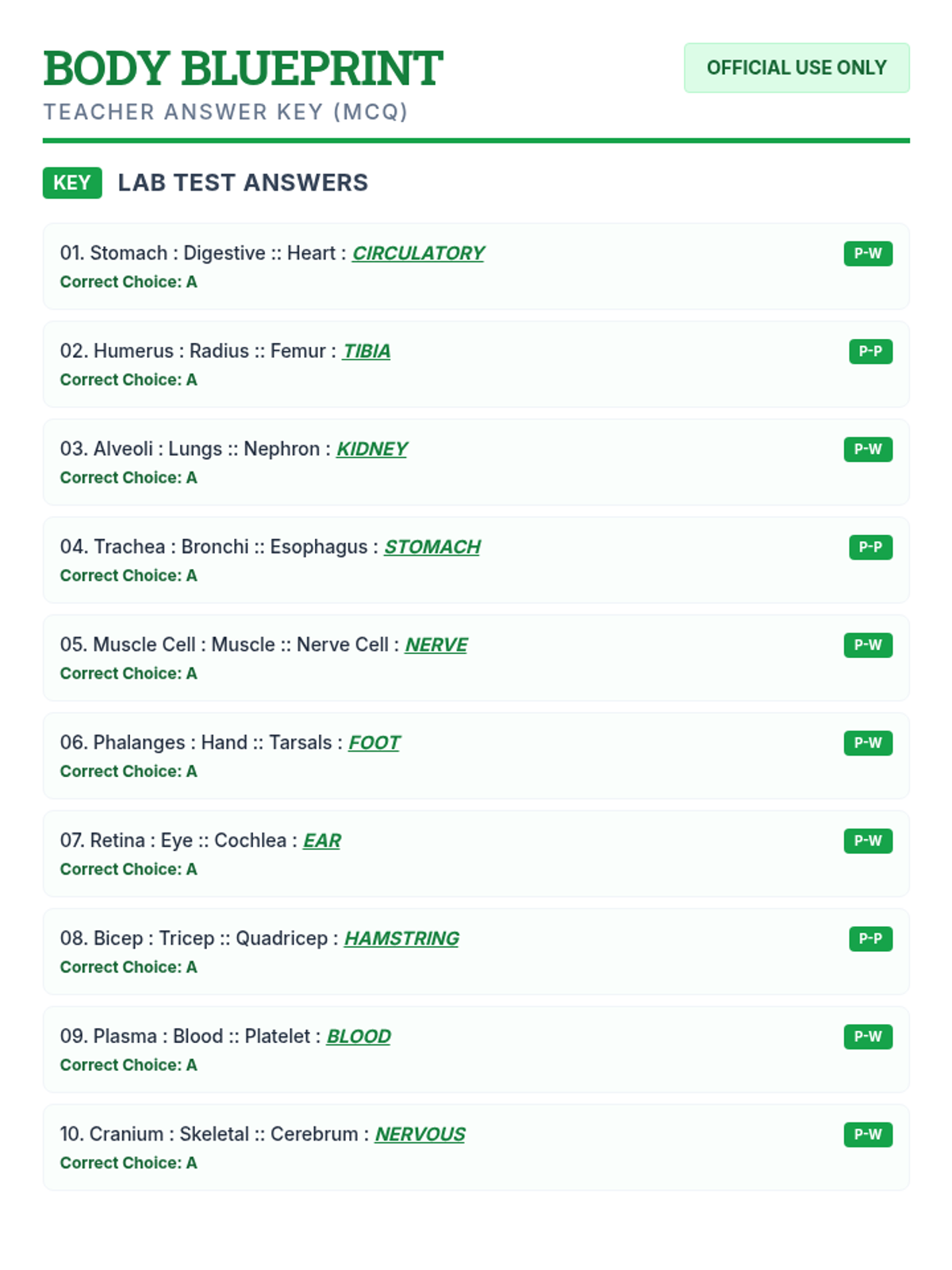
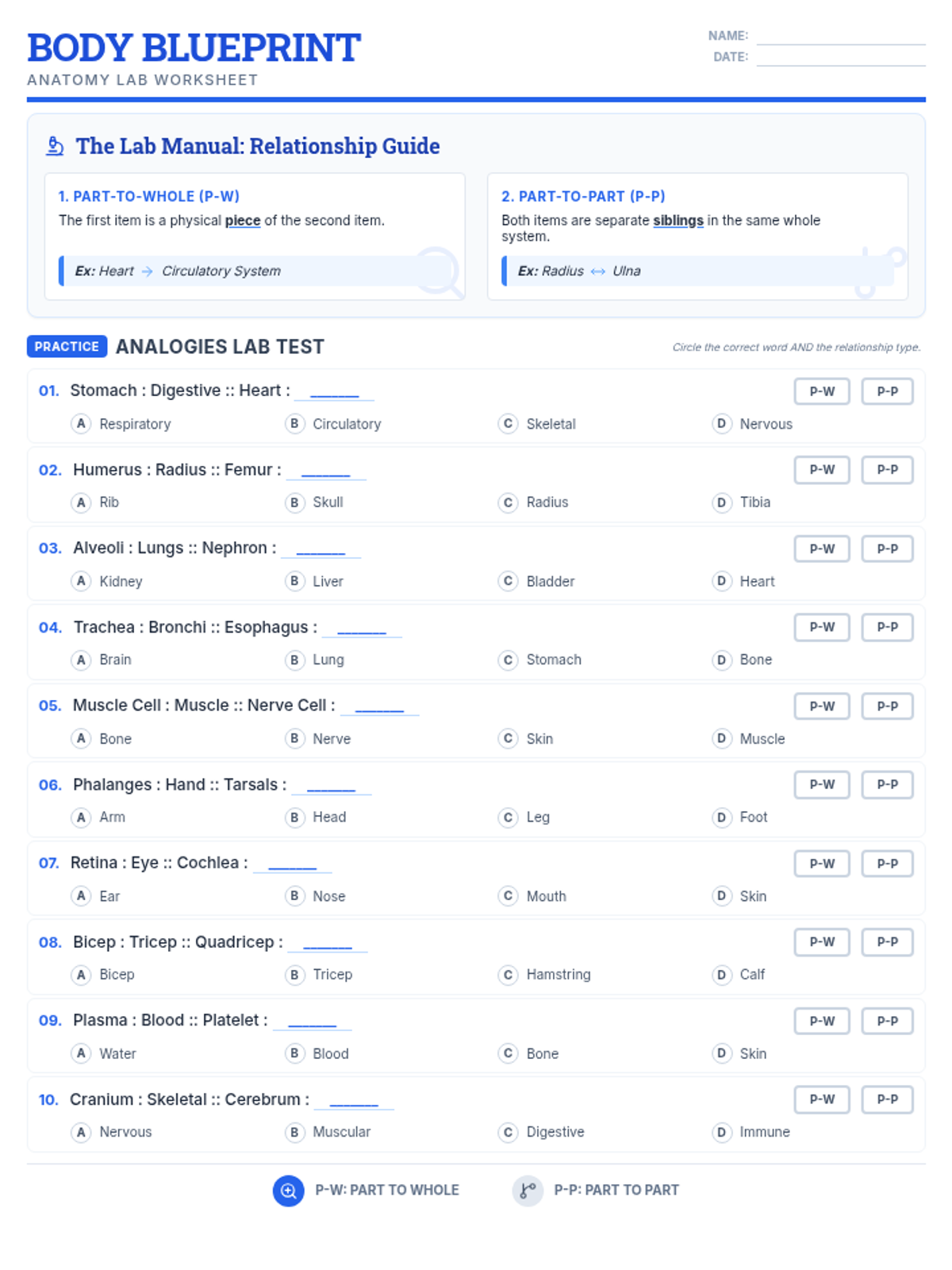
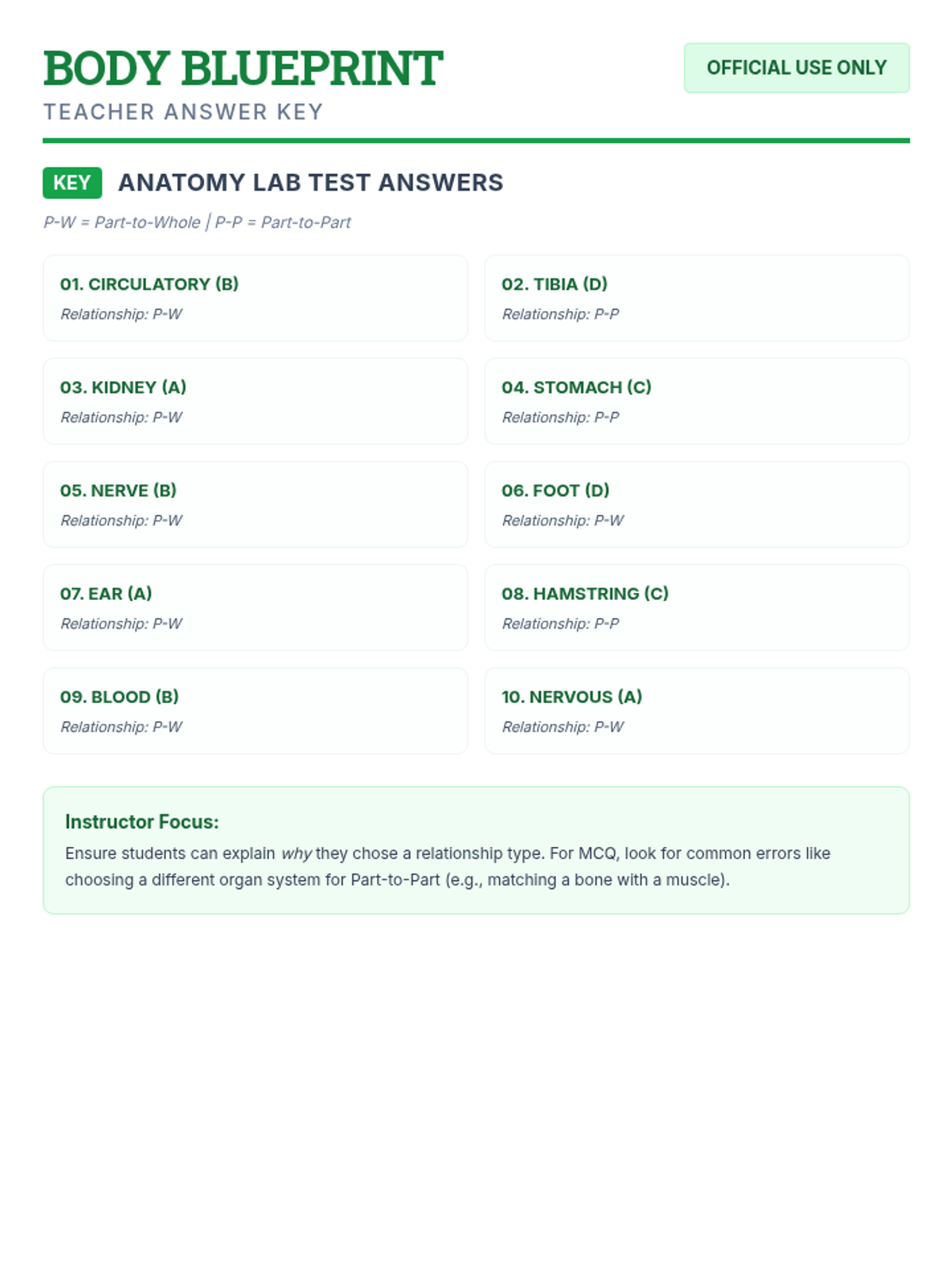
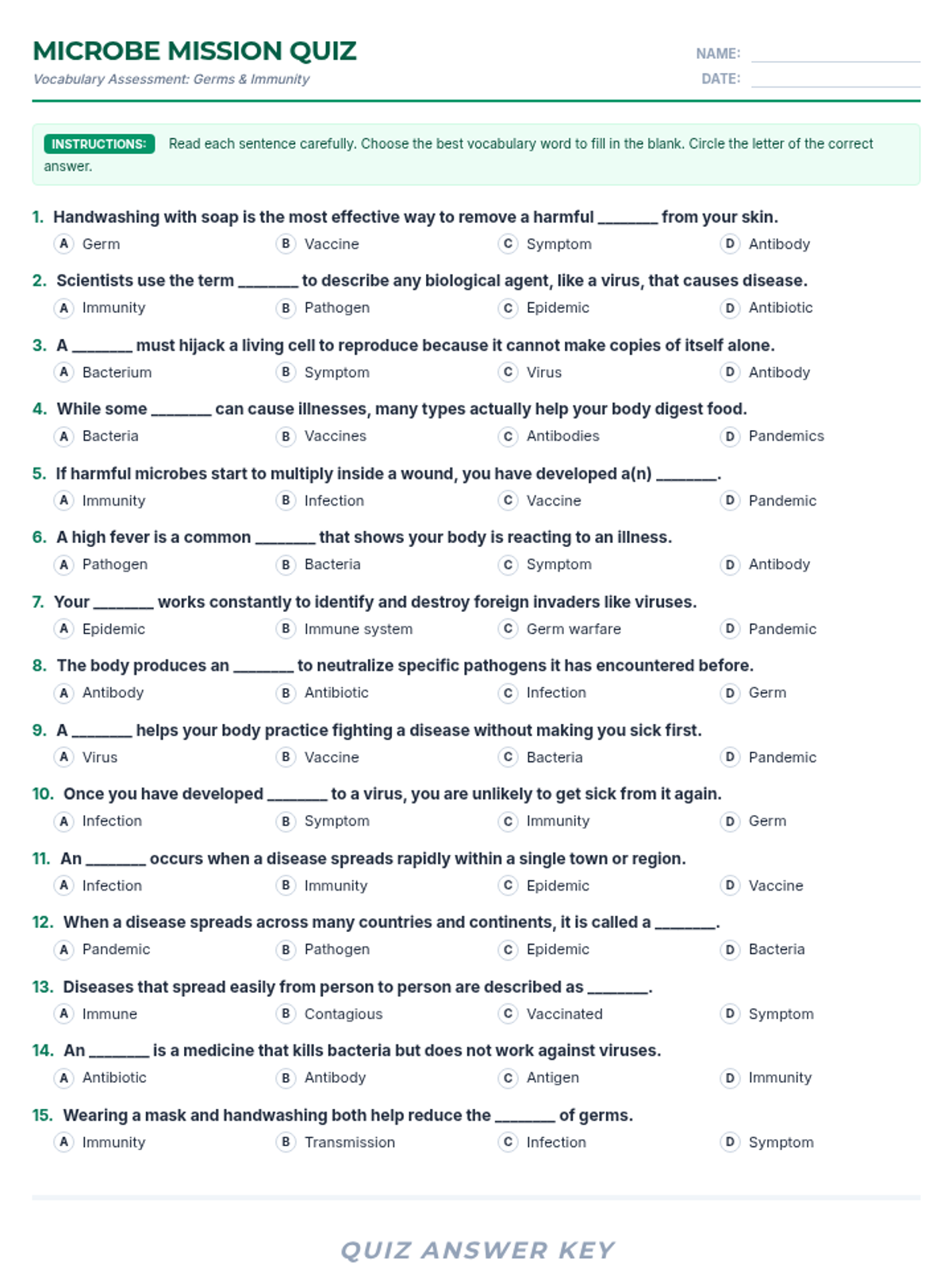
Students bridge the gap between Lewis structures and Valence Bond Theory by exploring the concept of orbital overlap. They visualize how s and p orbitals interact to form covalent bonds and discuss why Lewis theory is insufficient for explaining bond angles.