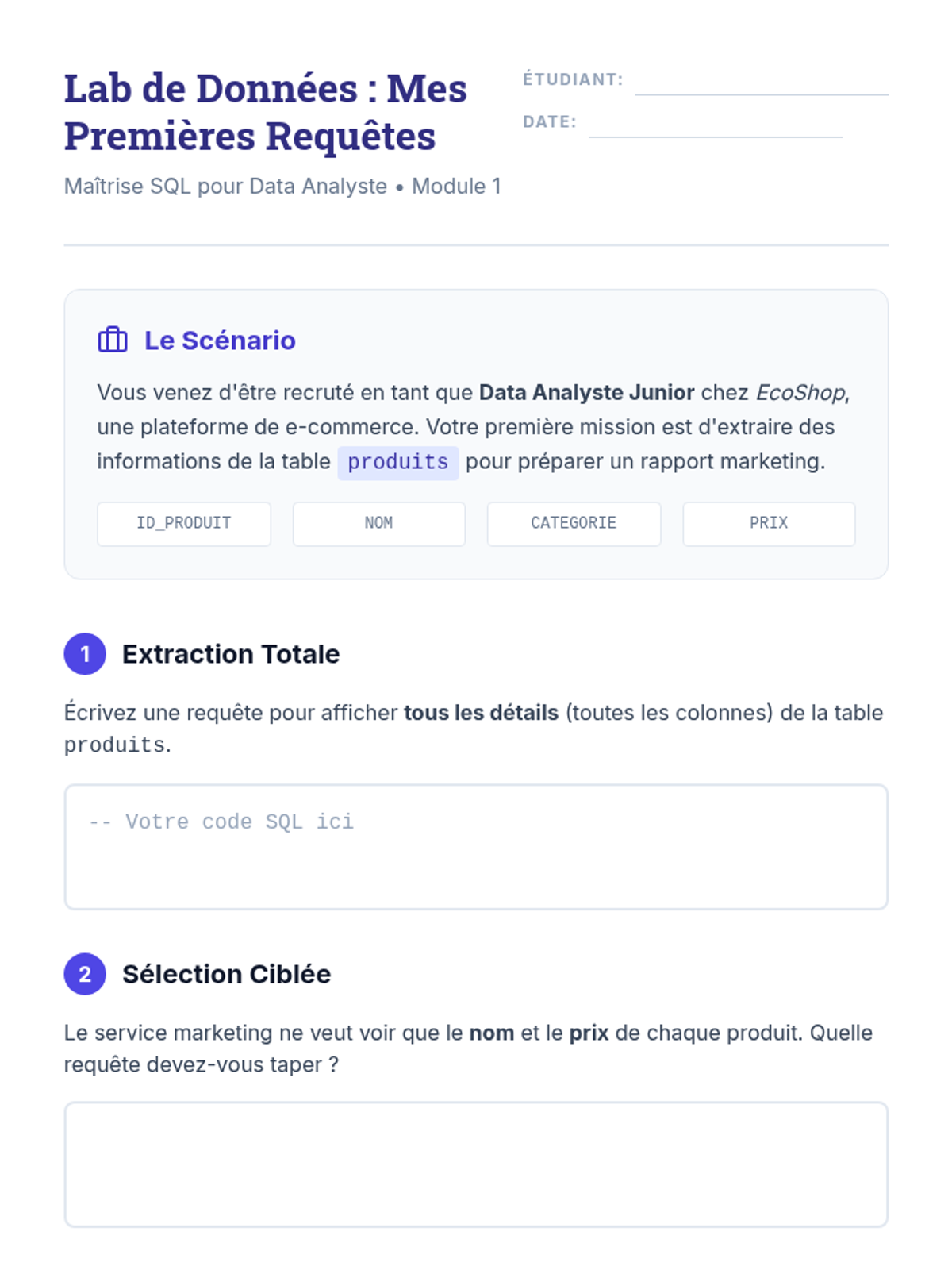
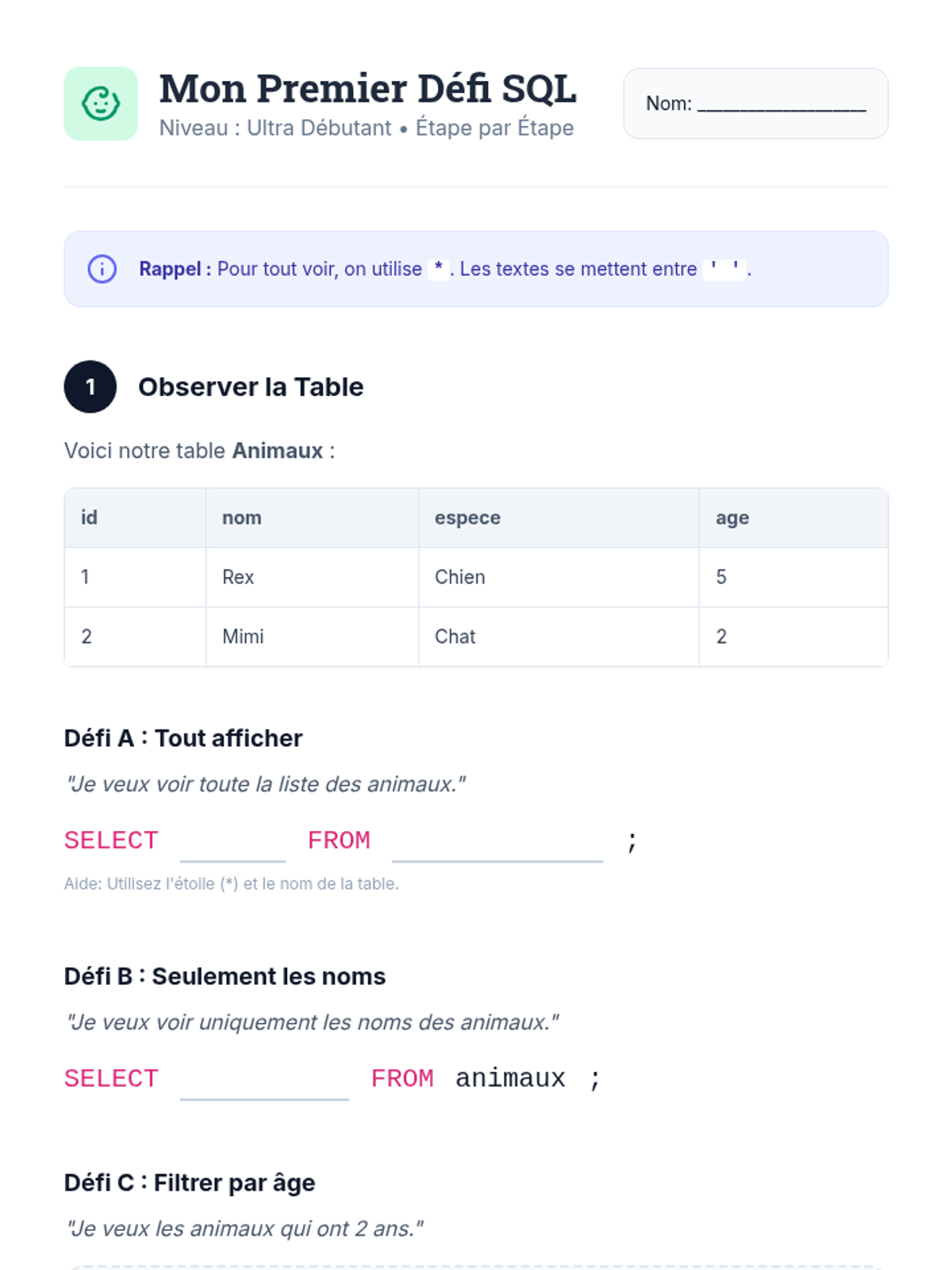
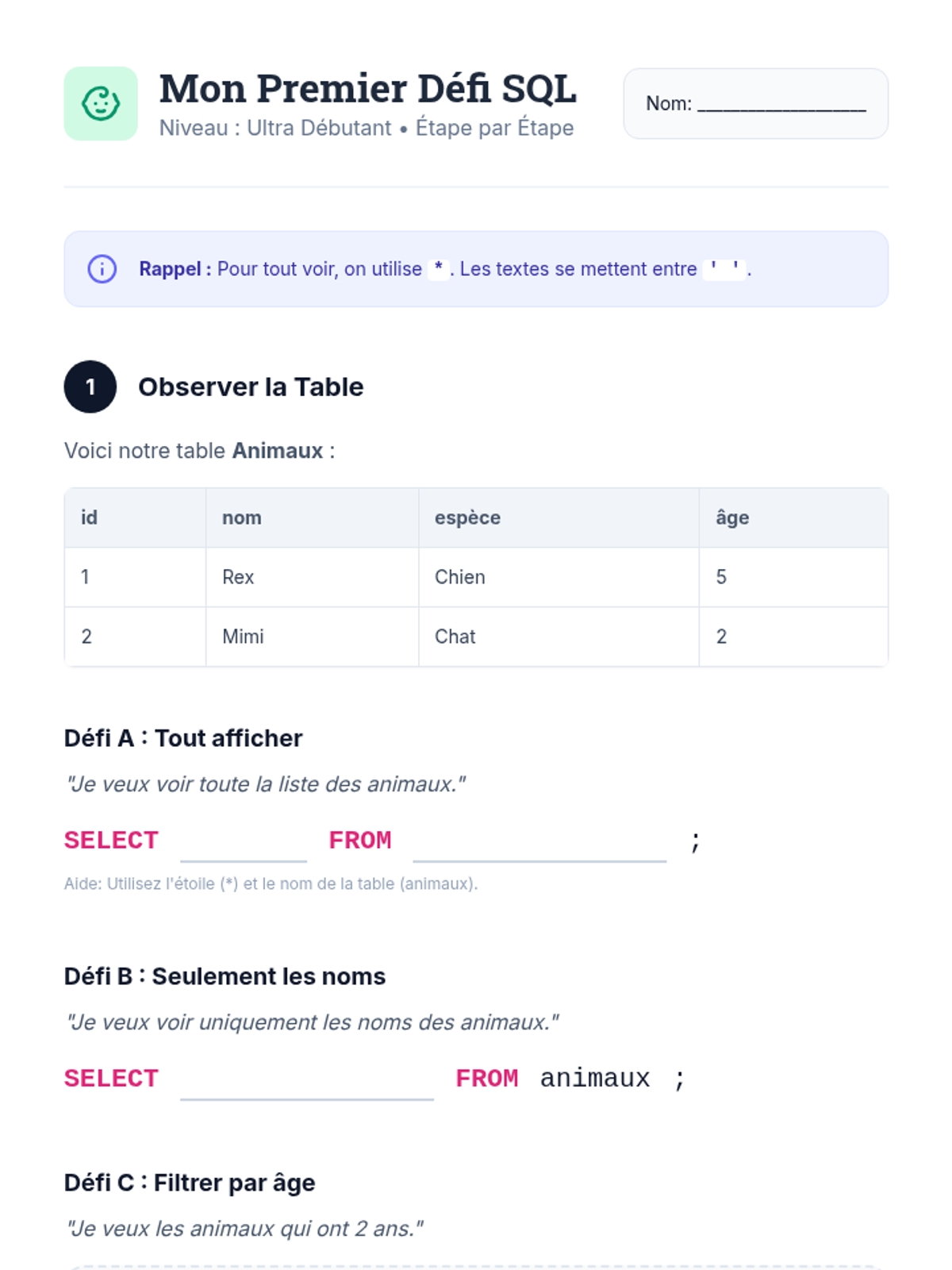
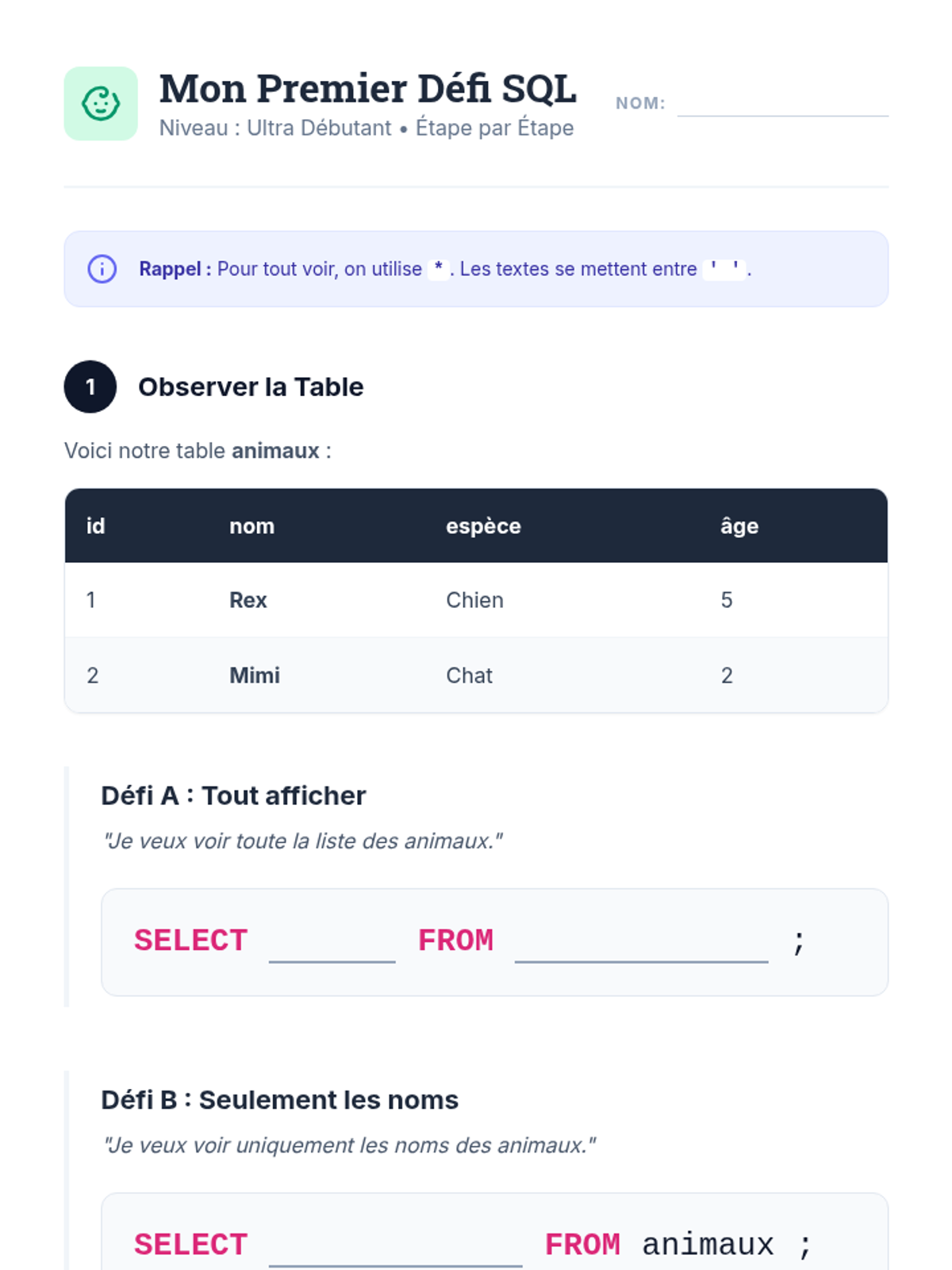
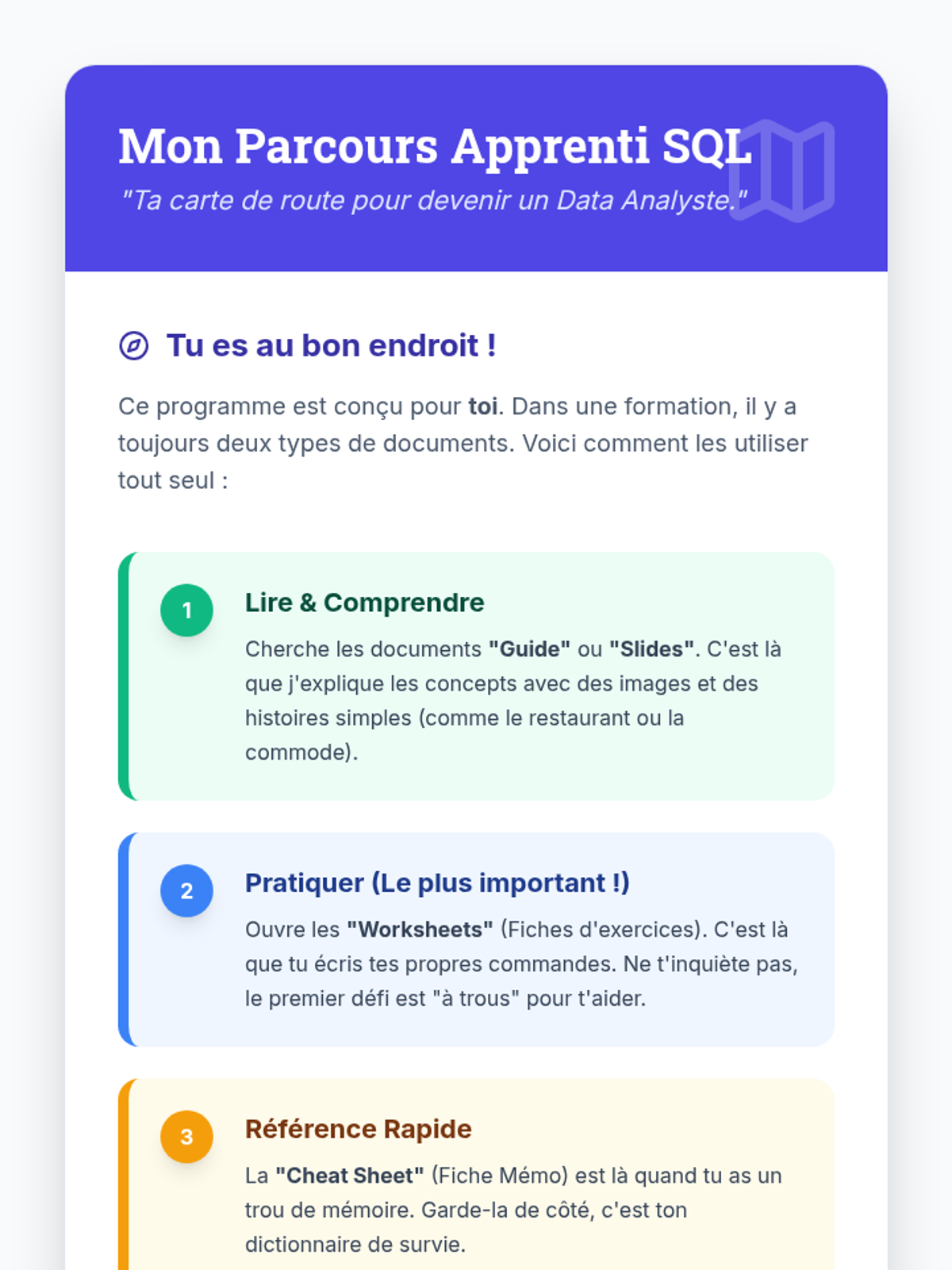
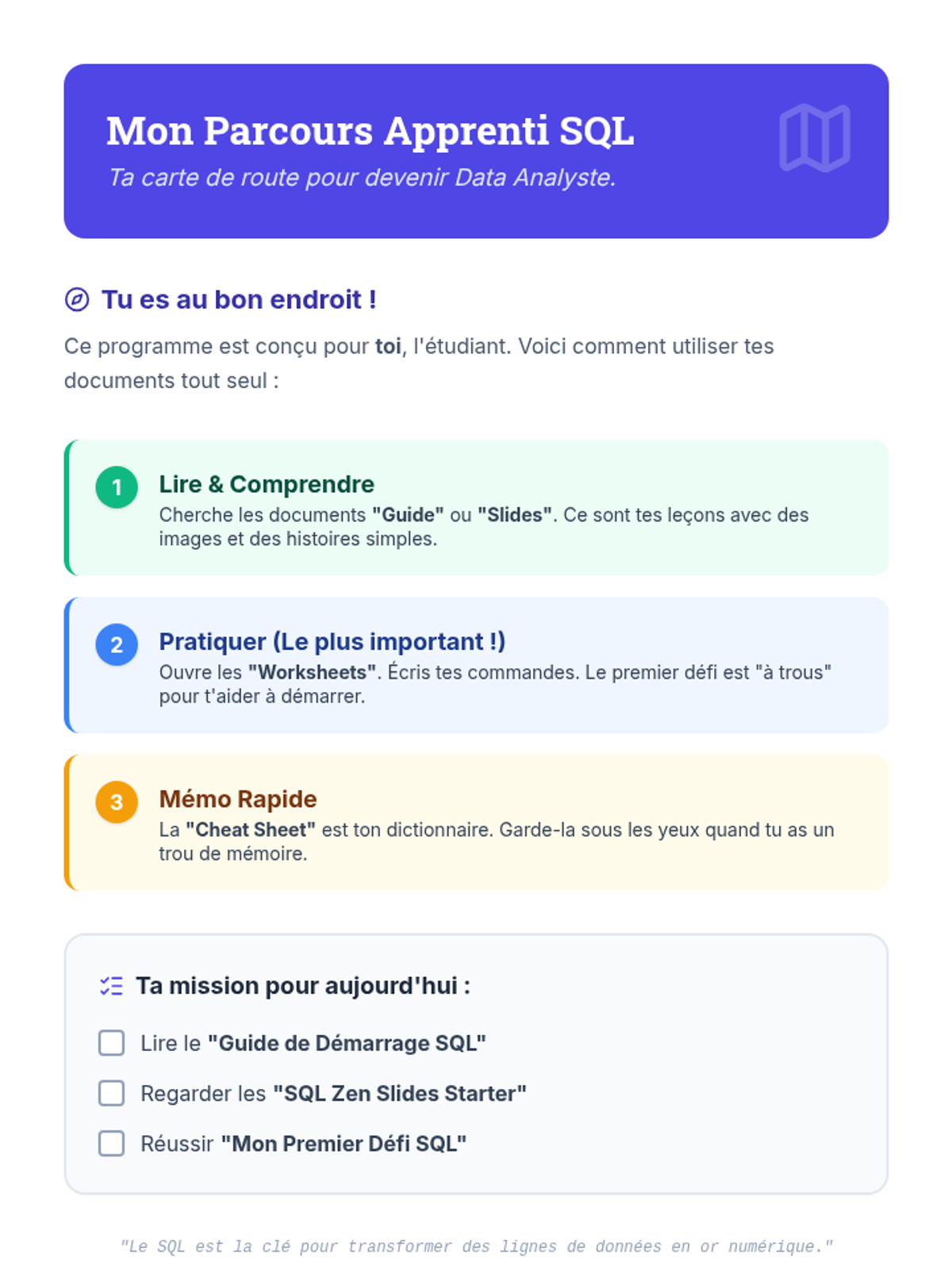
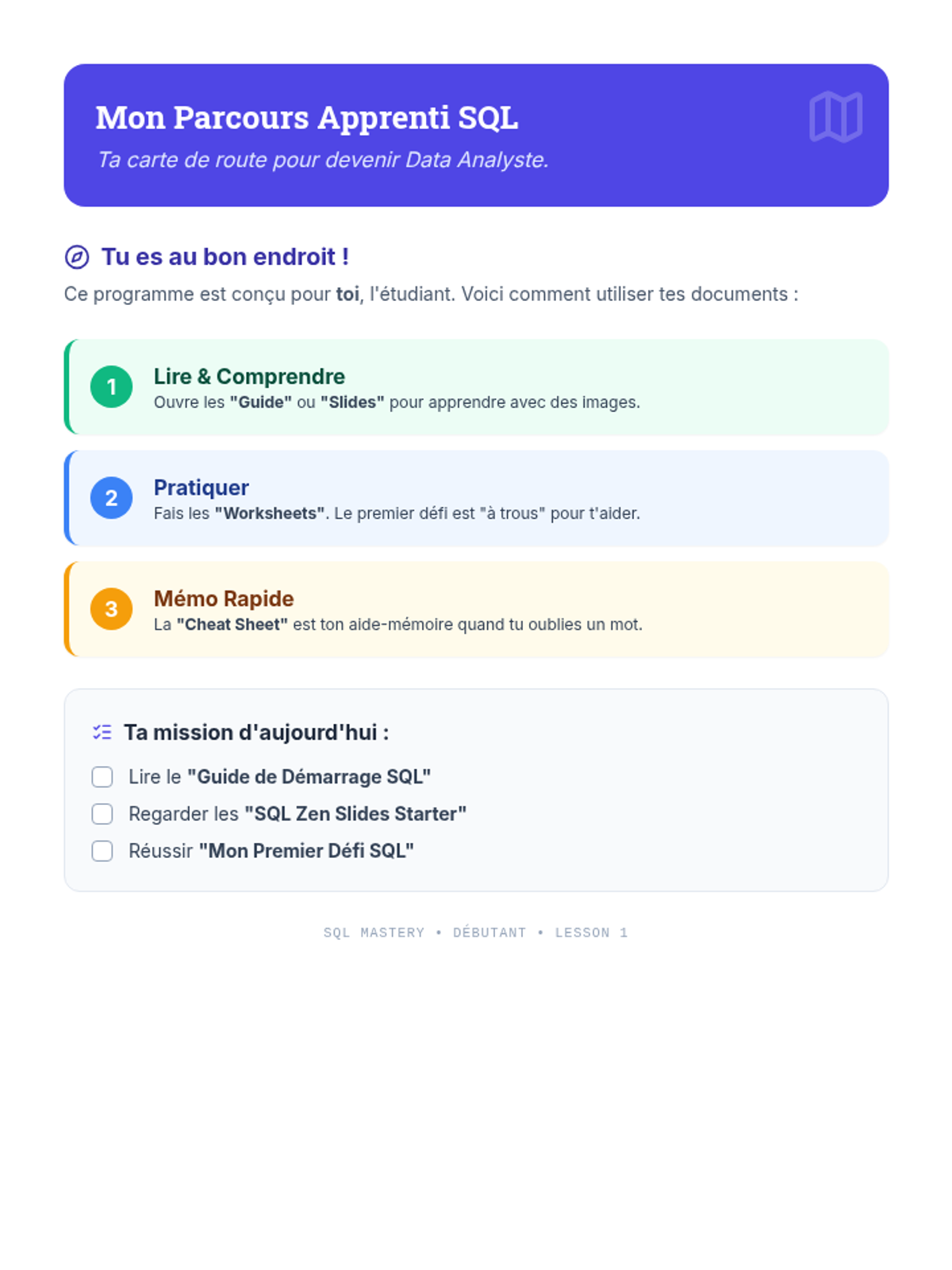
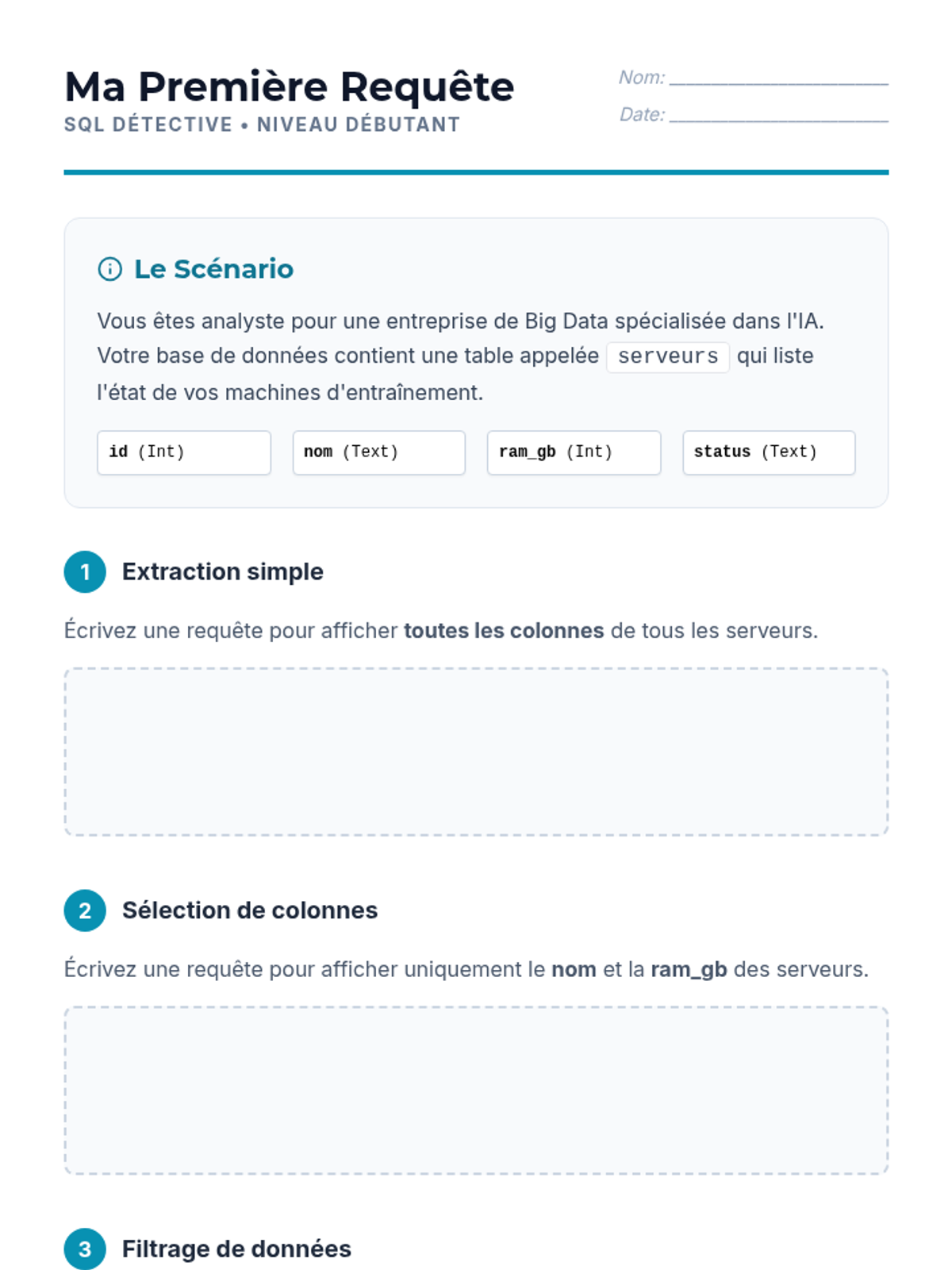
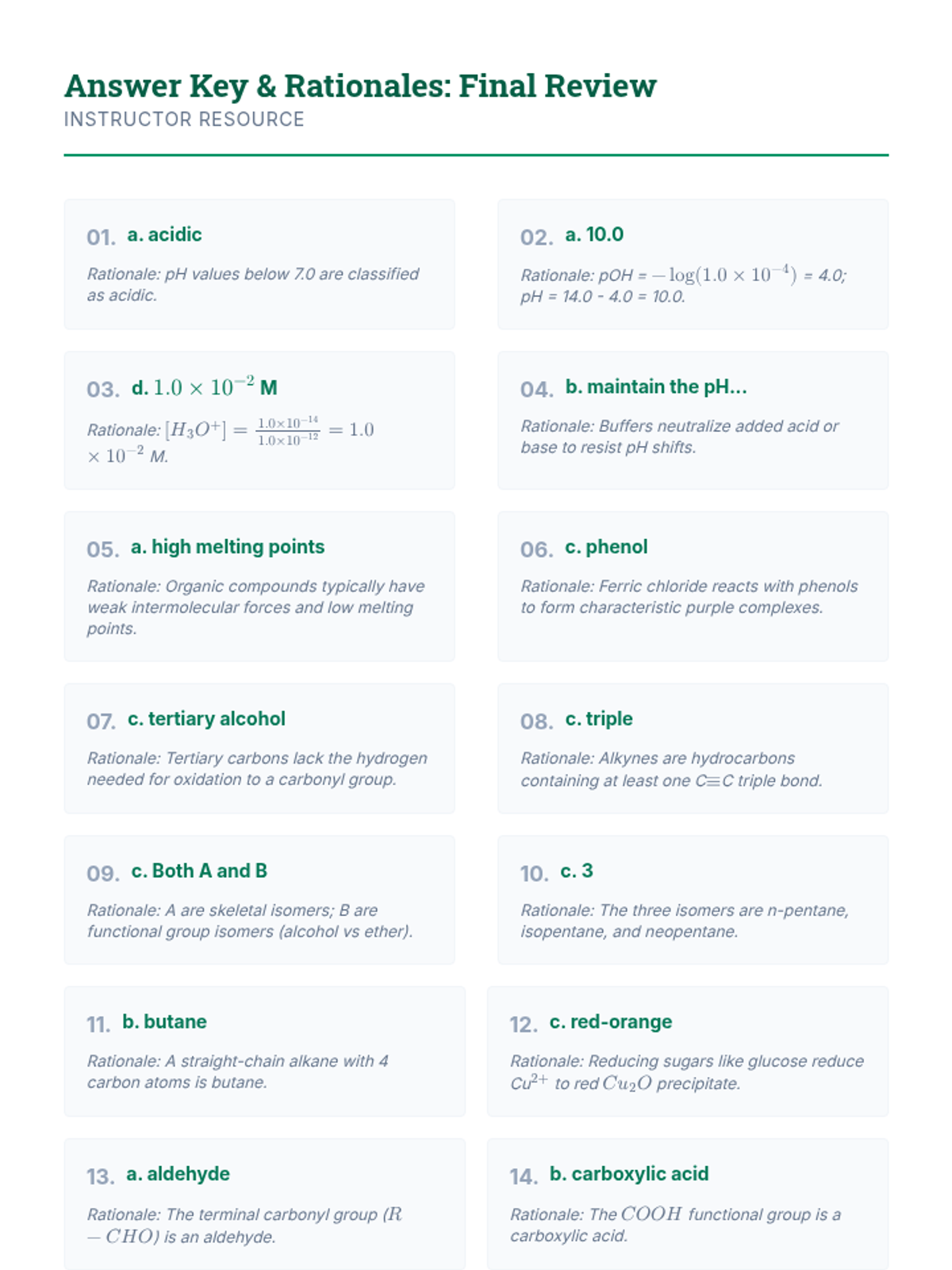
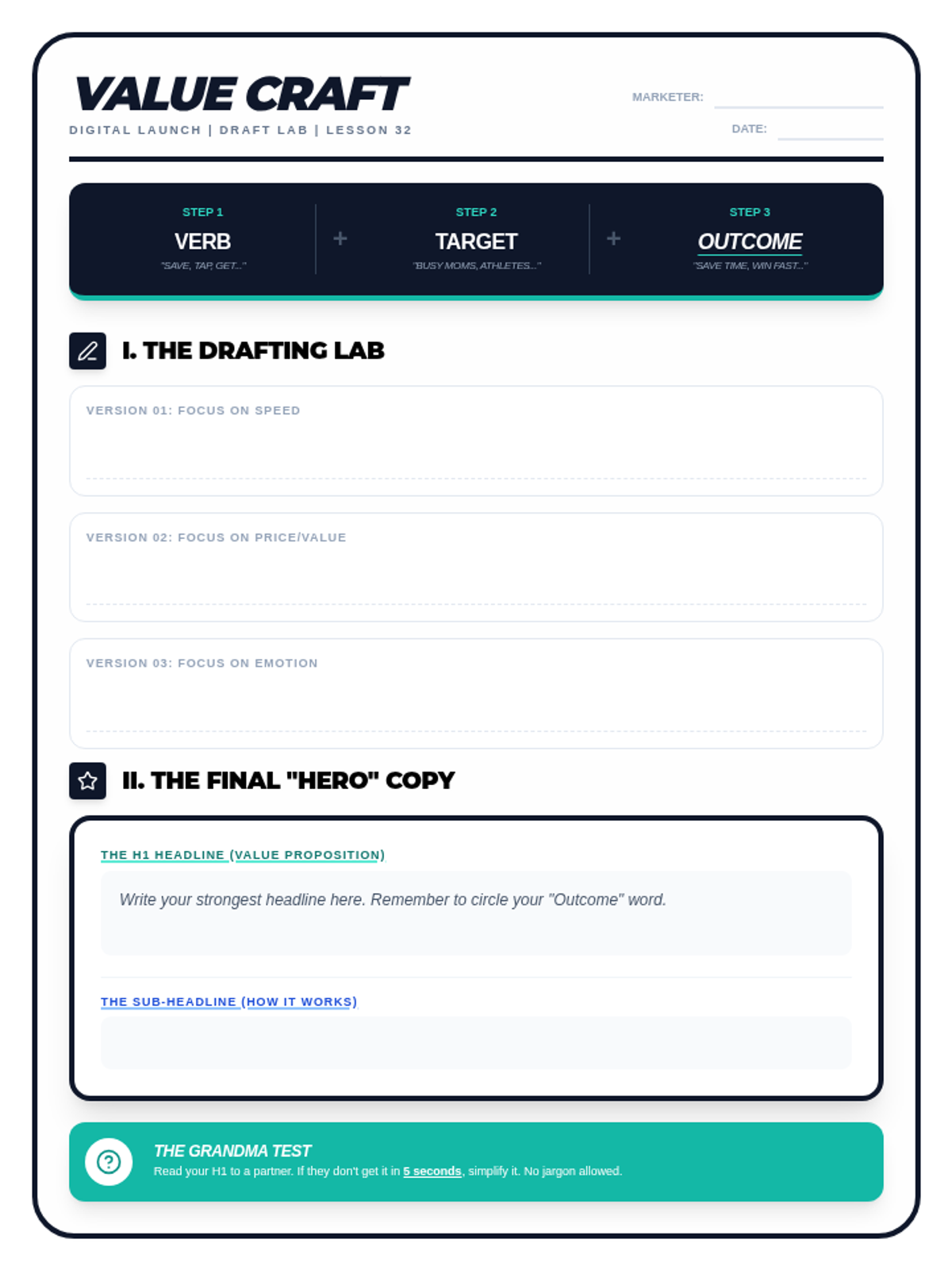
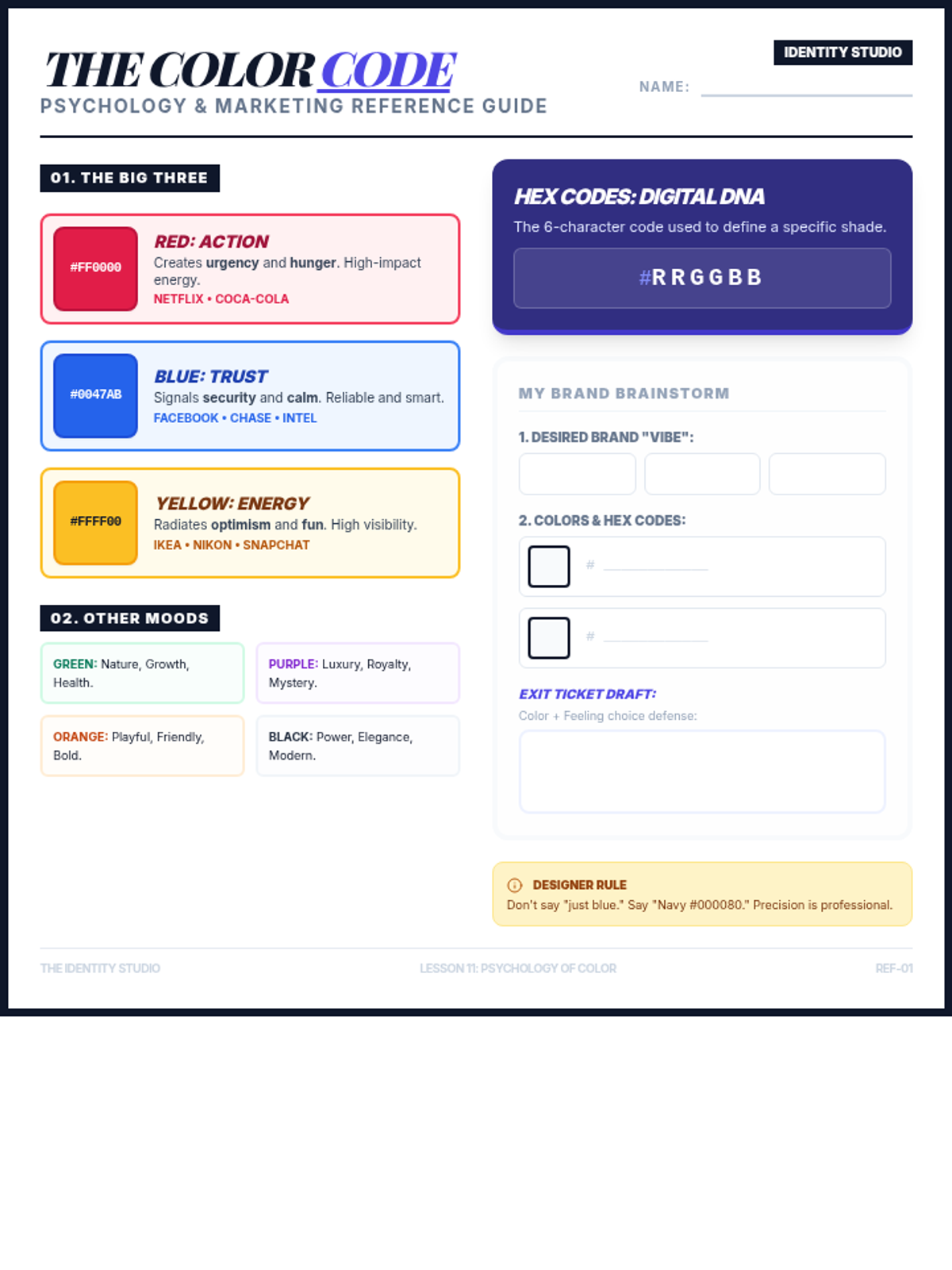
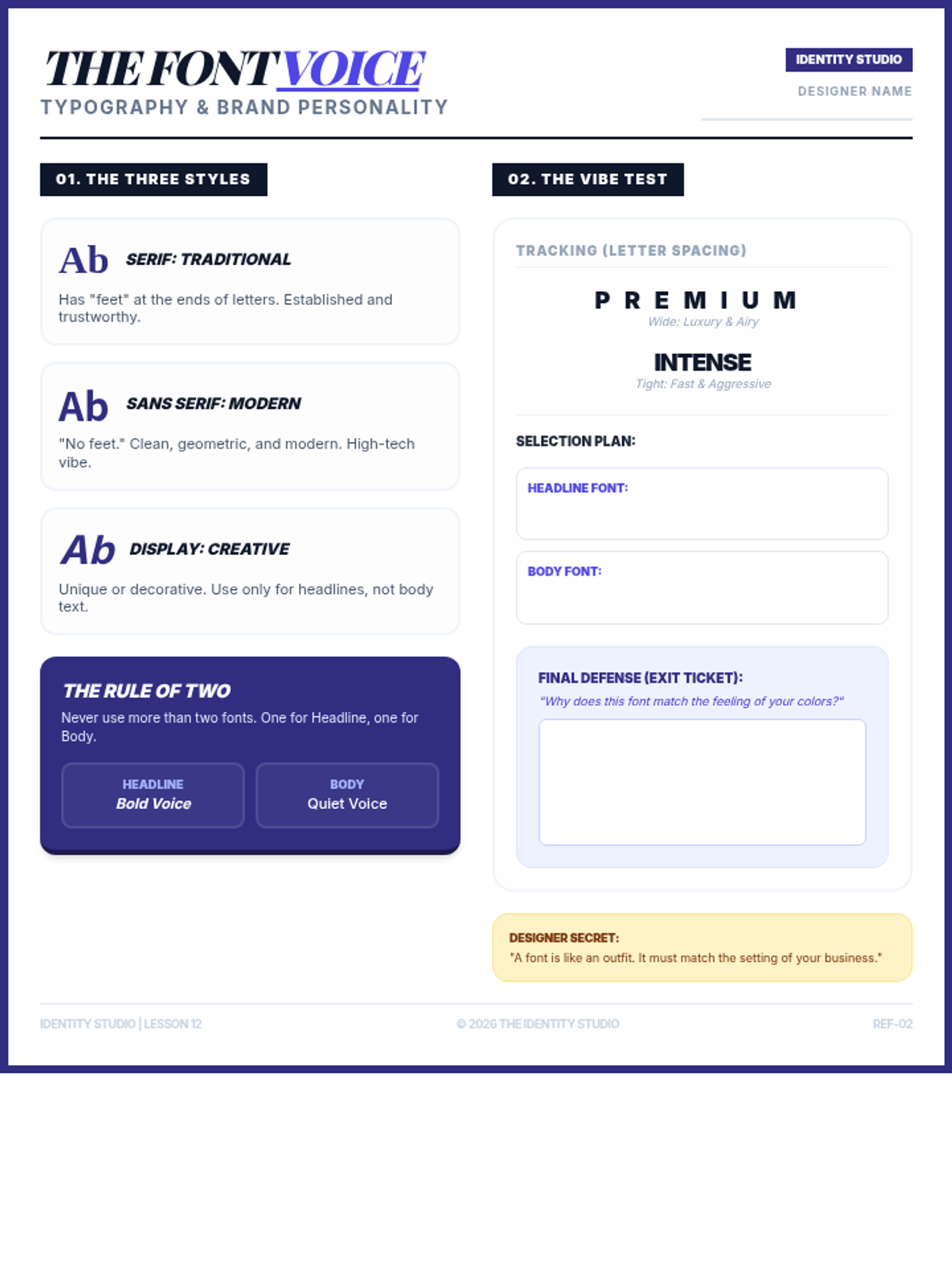
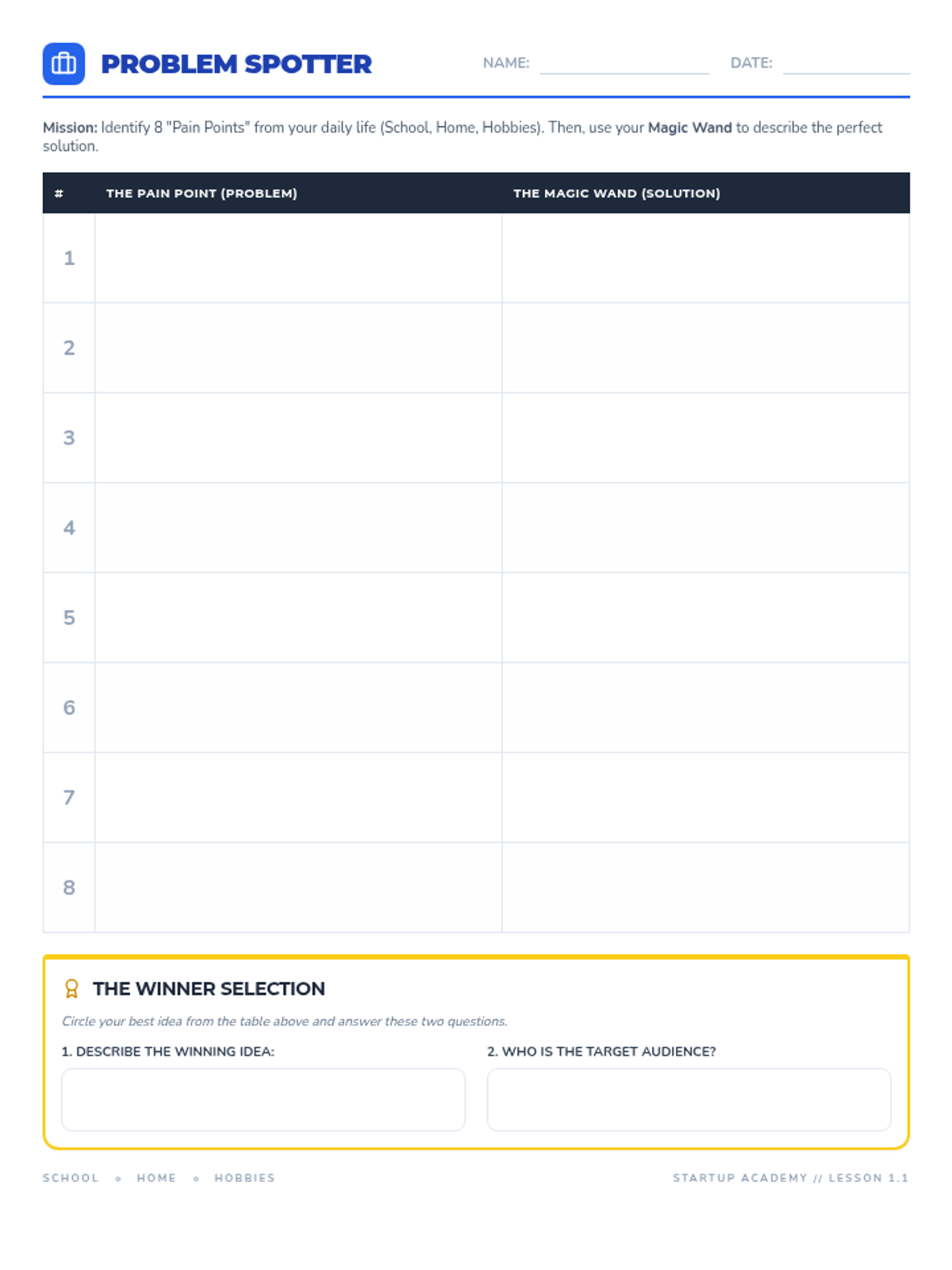
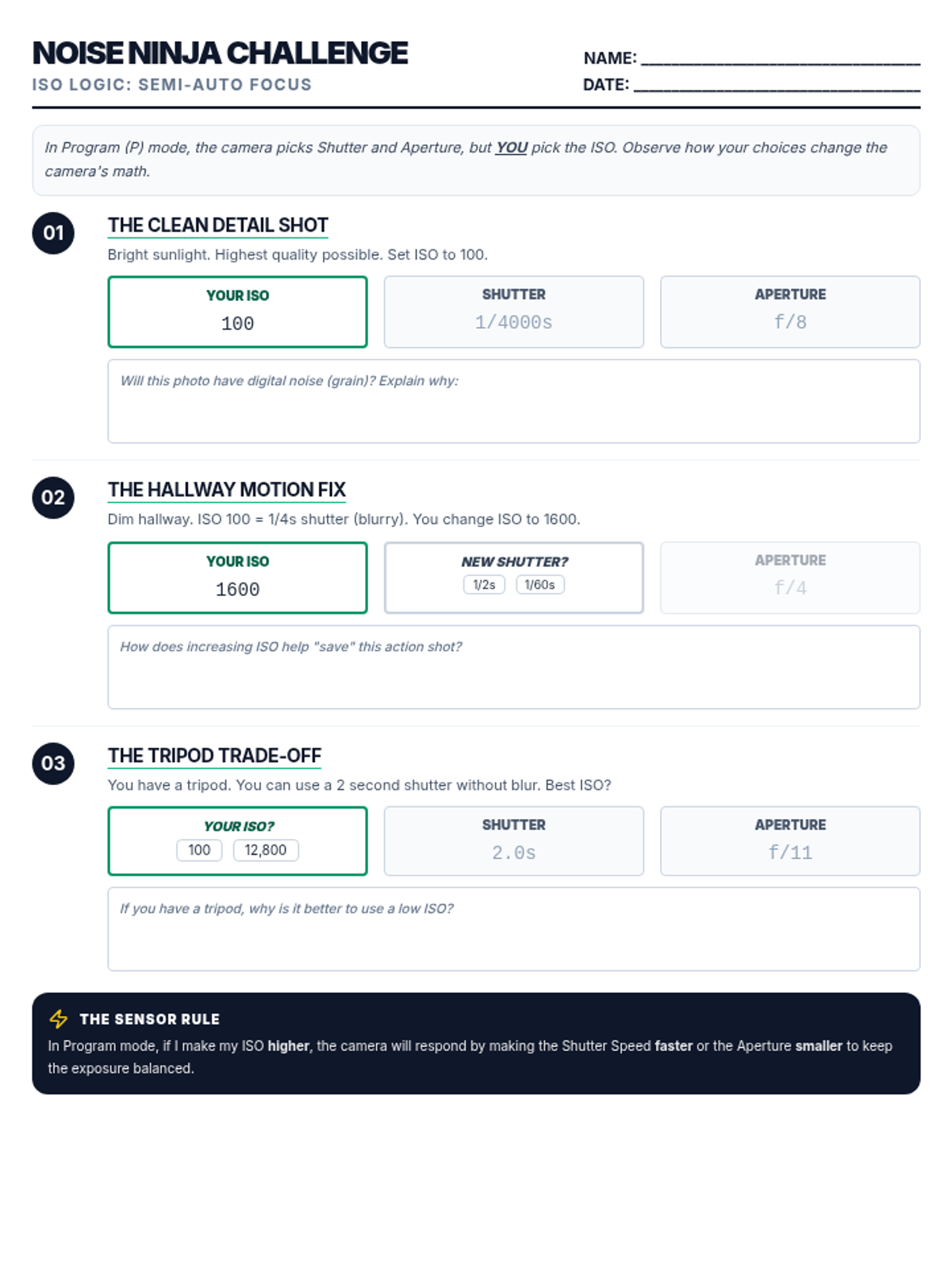
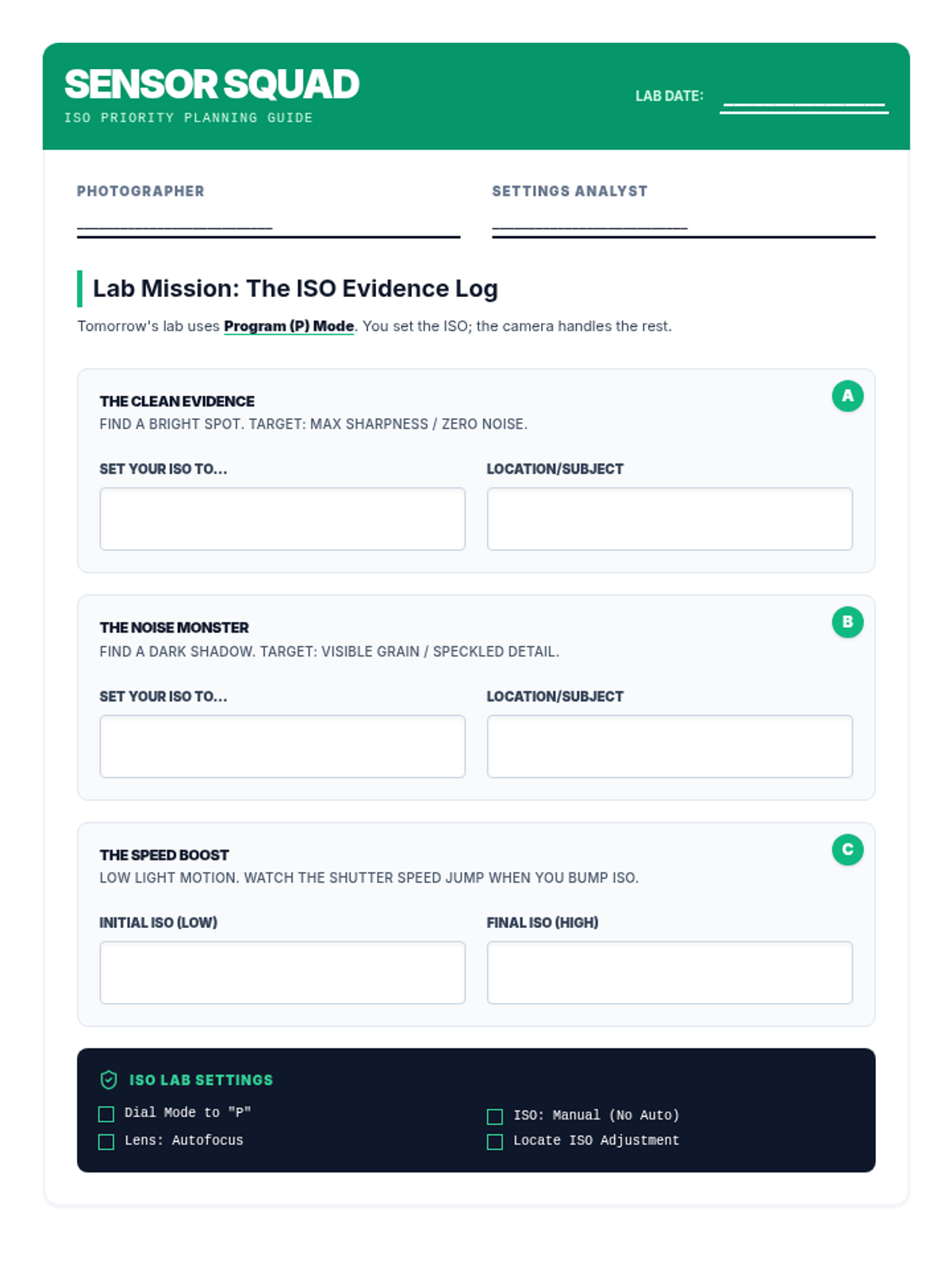
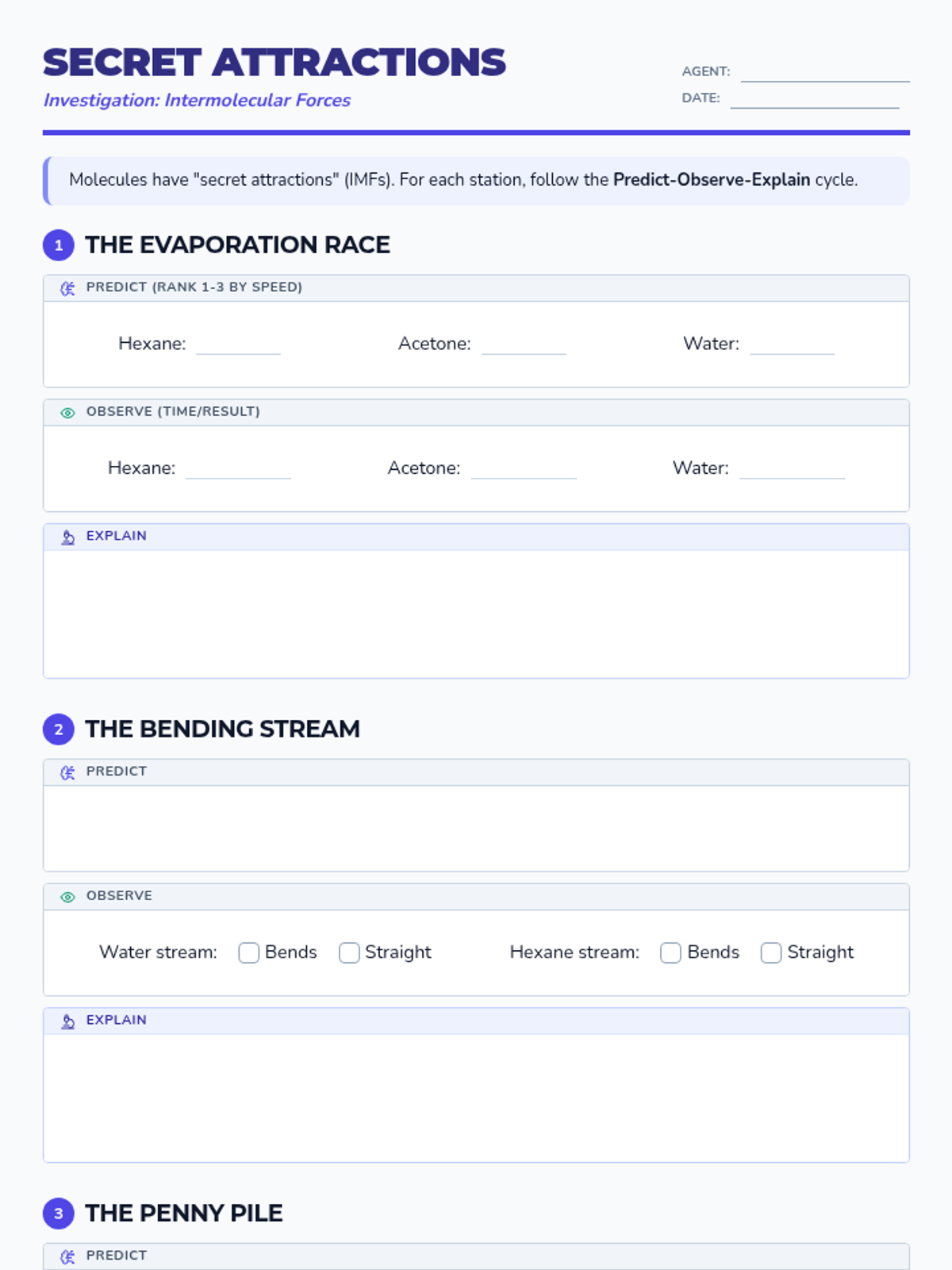
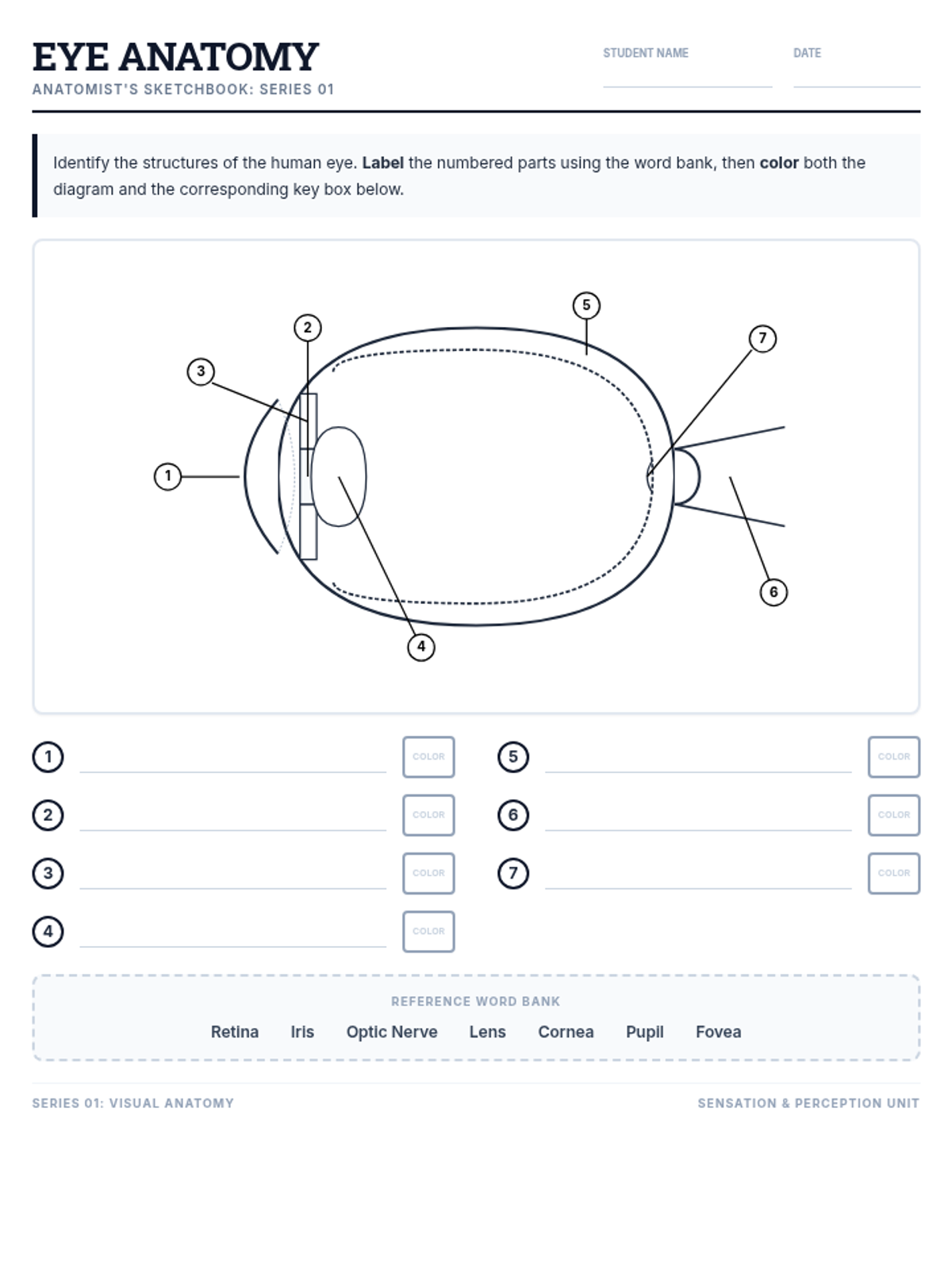
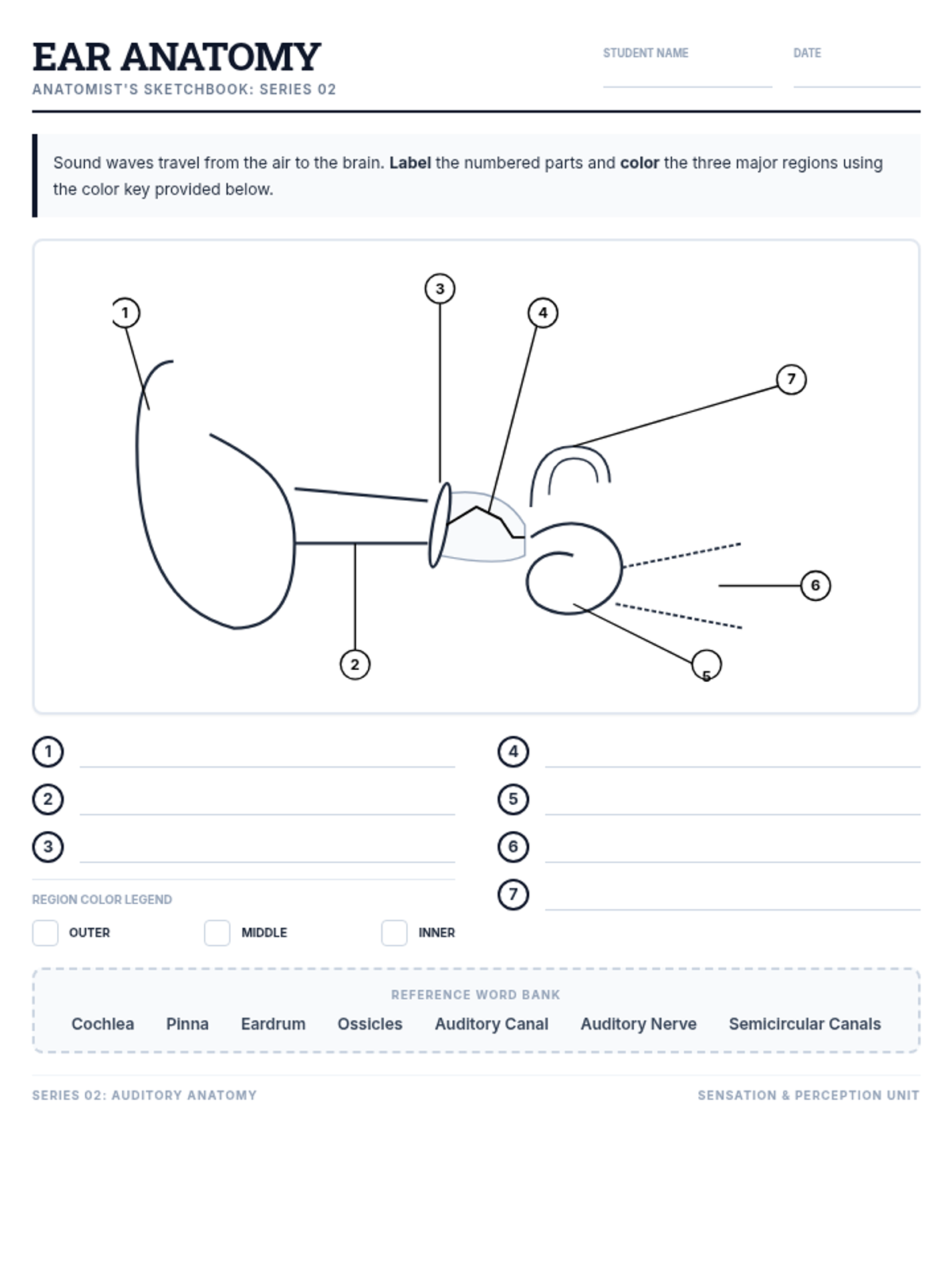
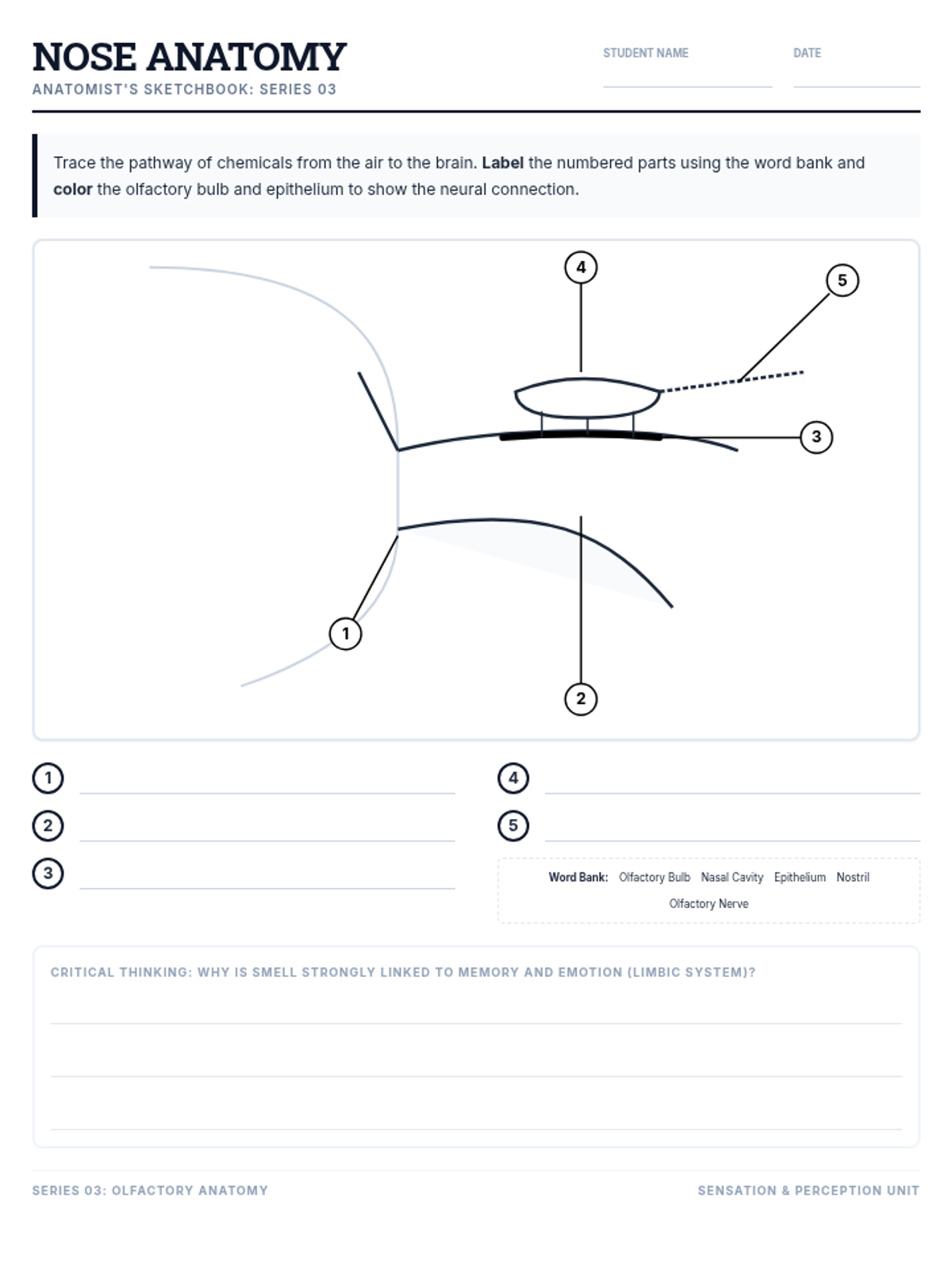
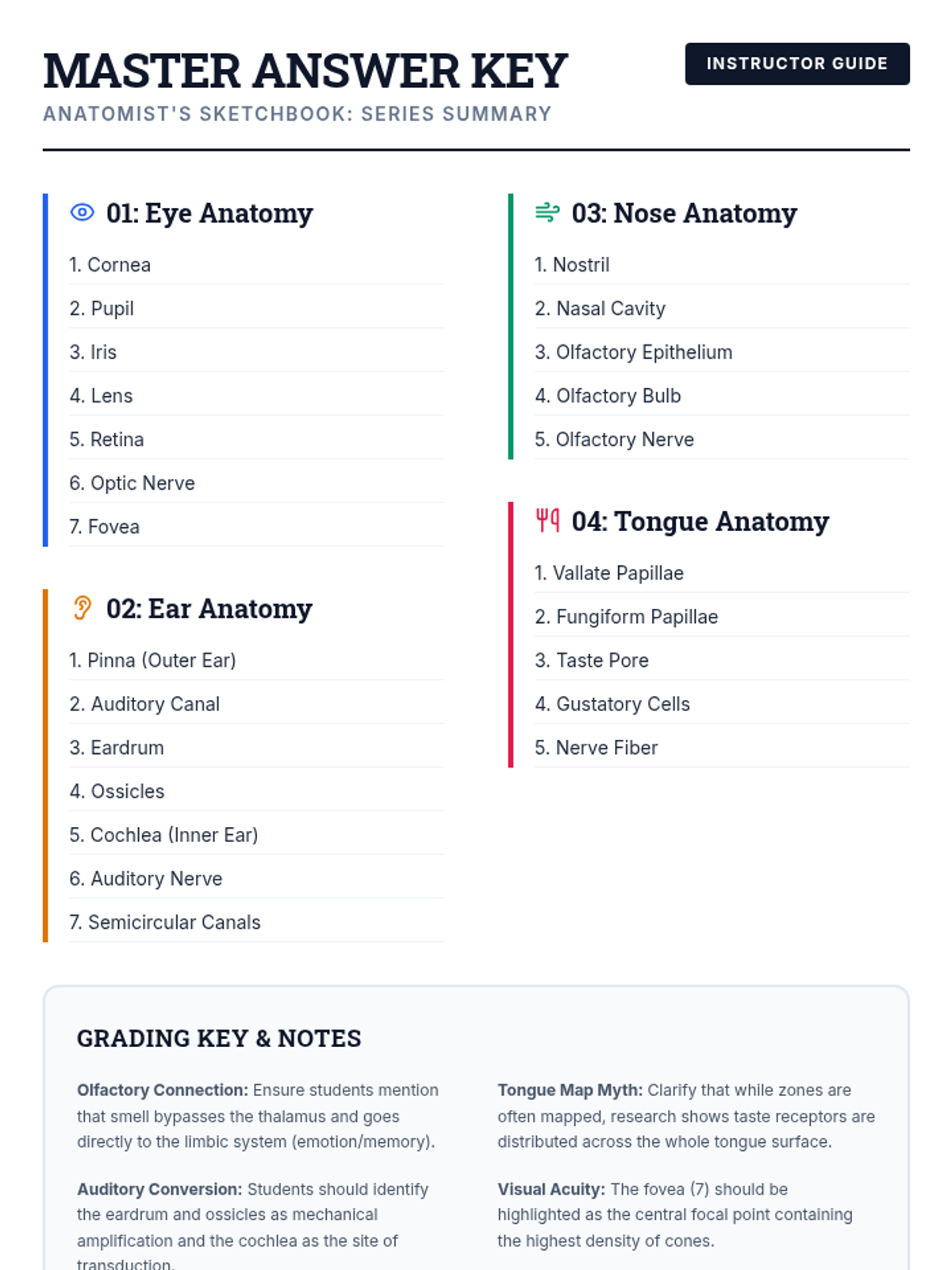
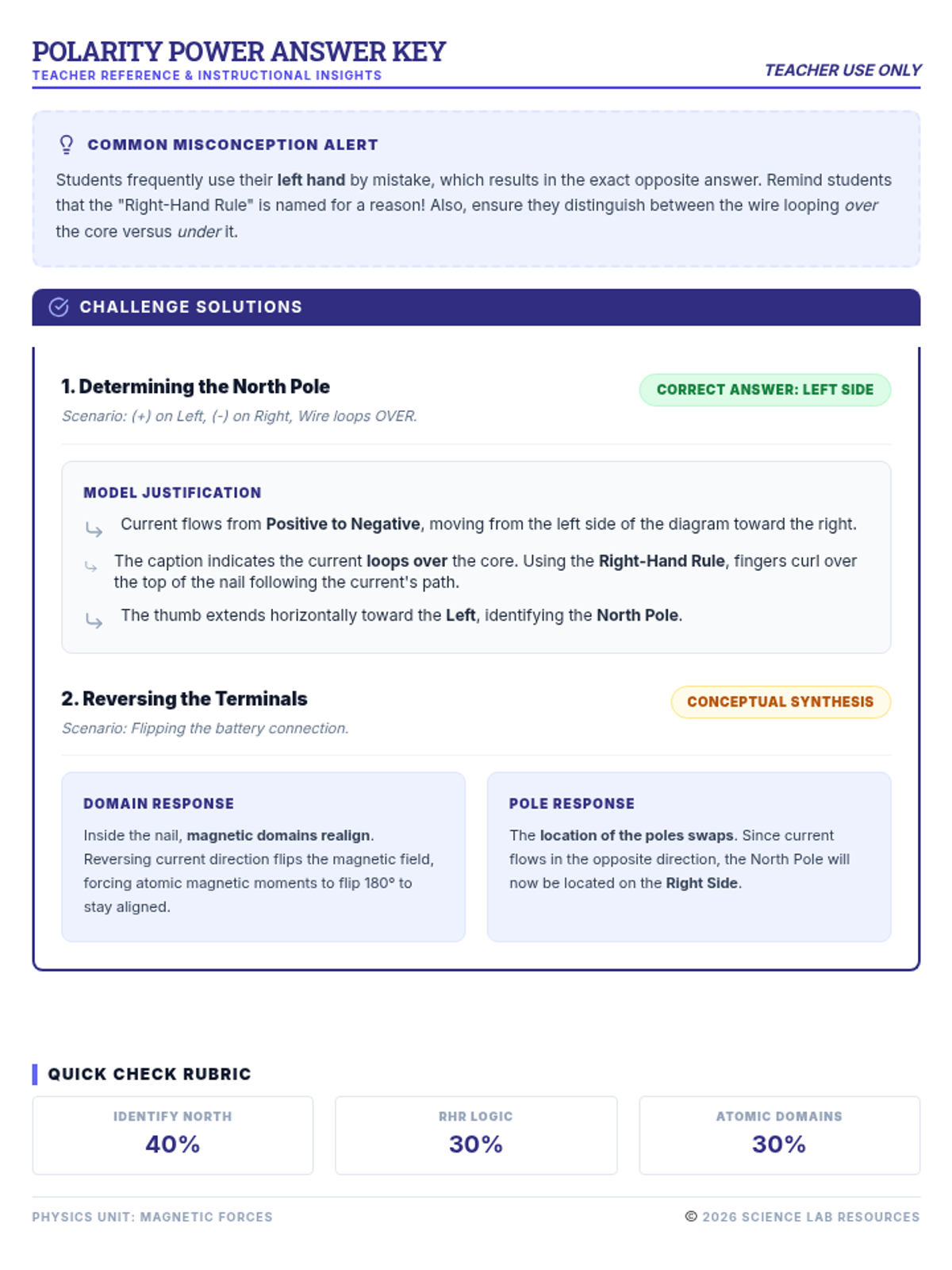
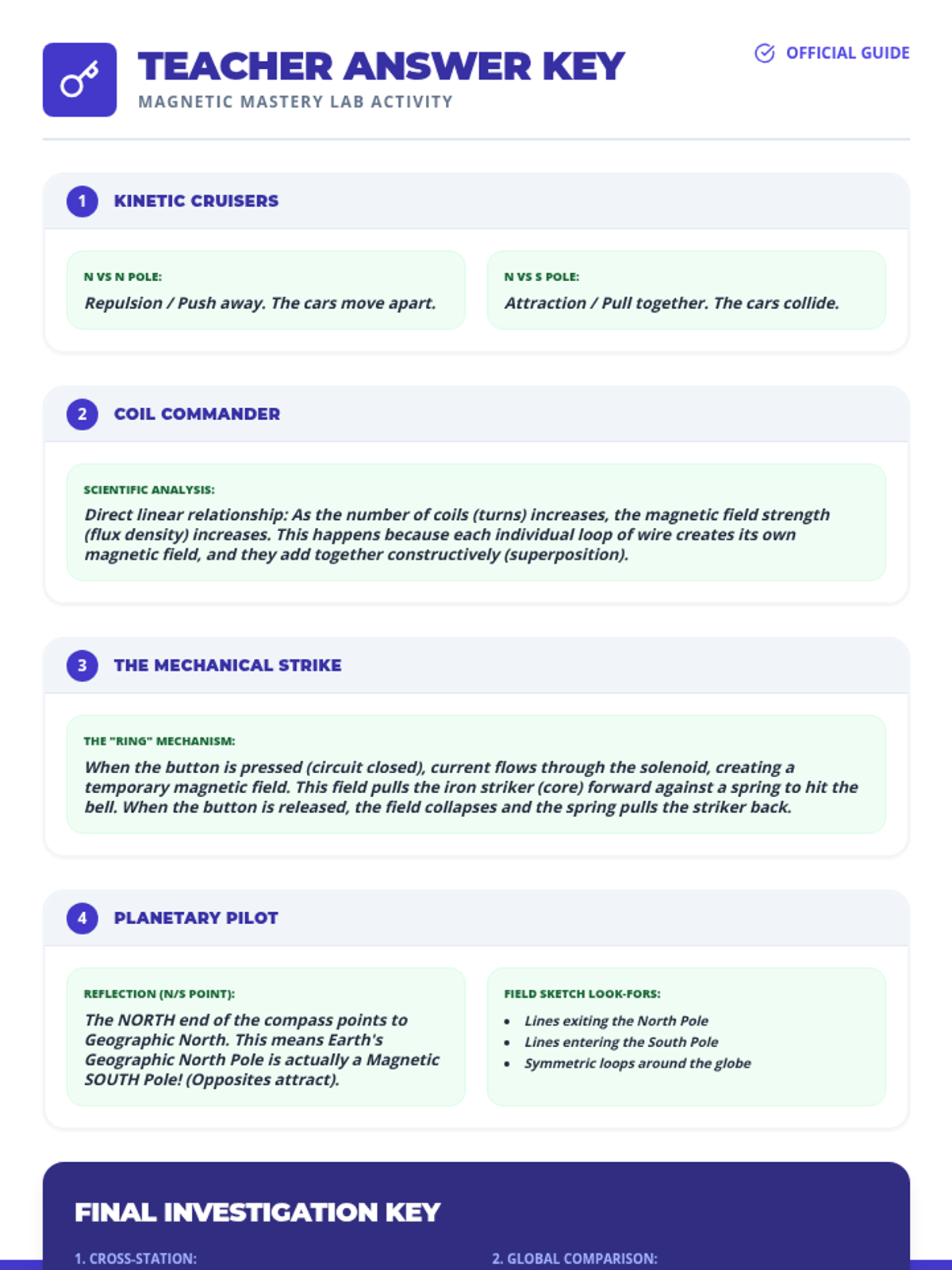
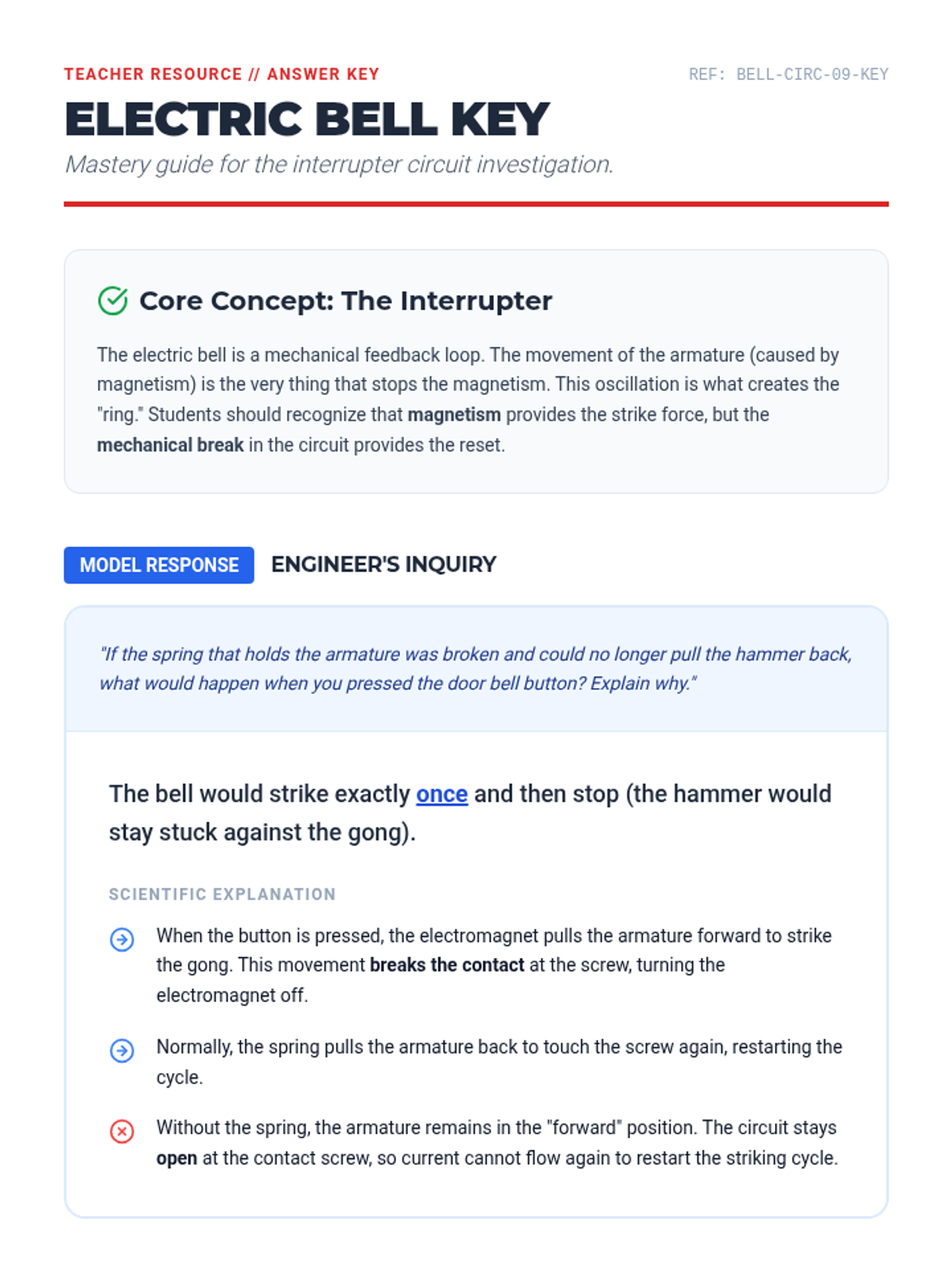
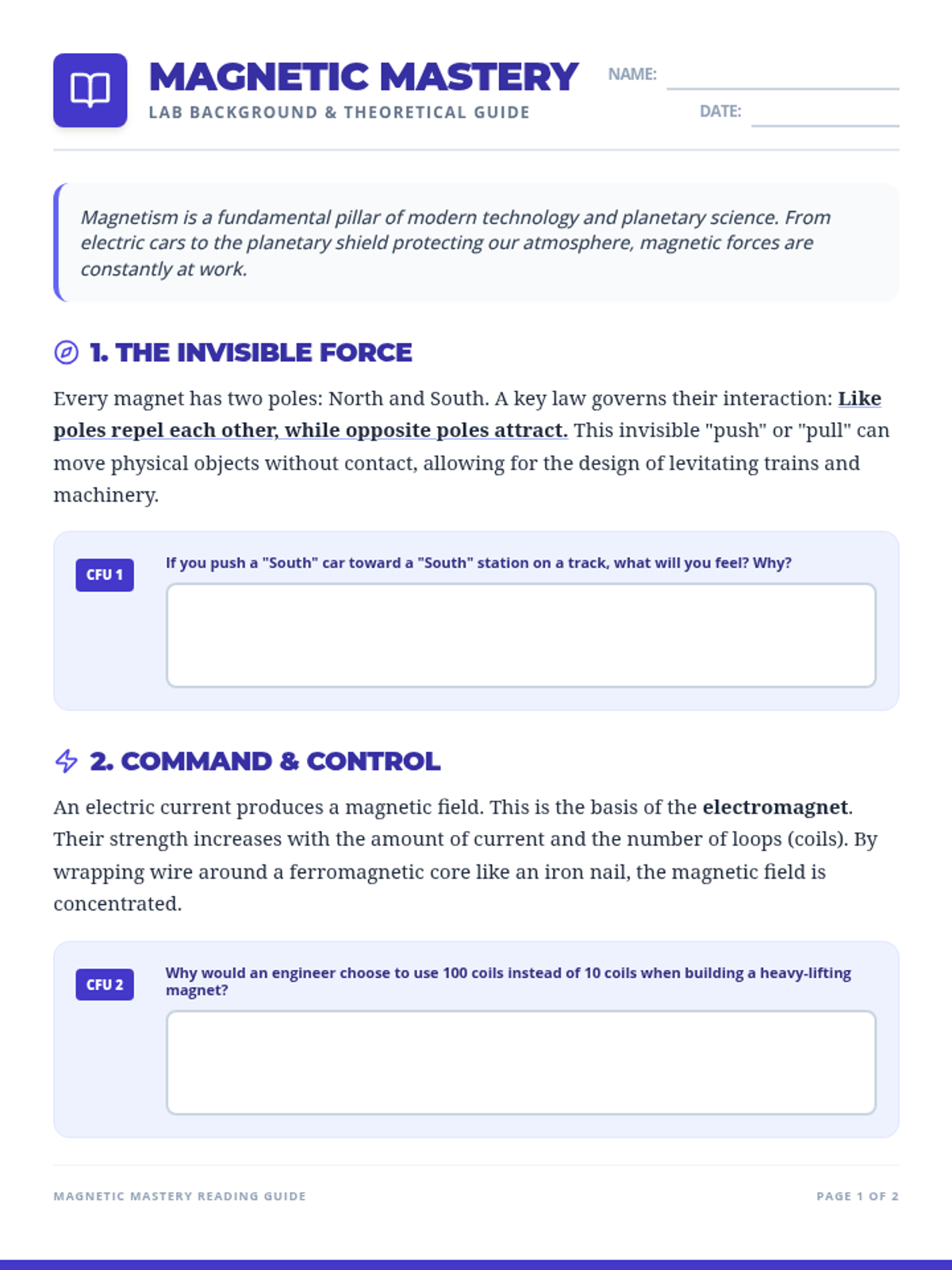
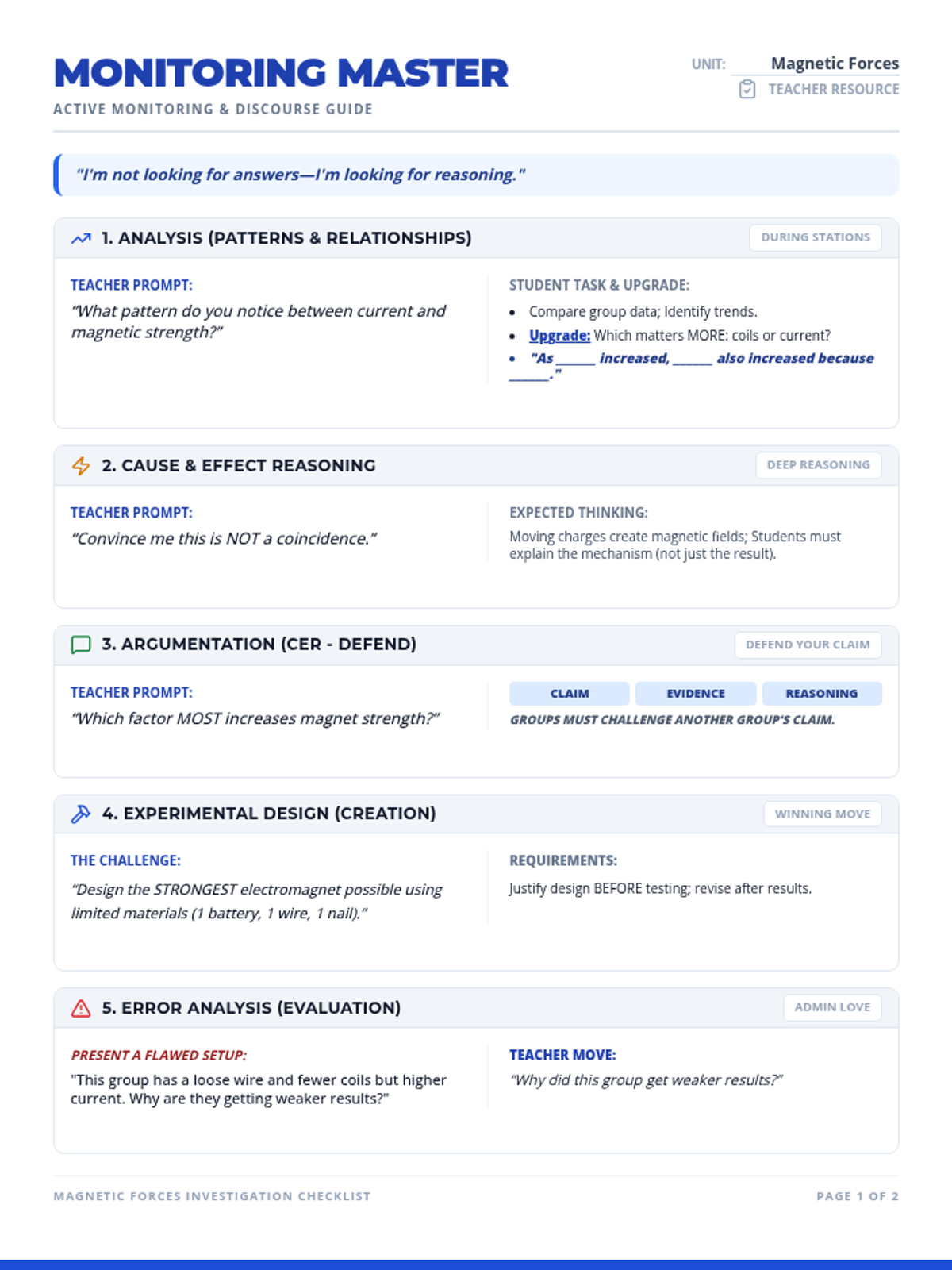
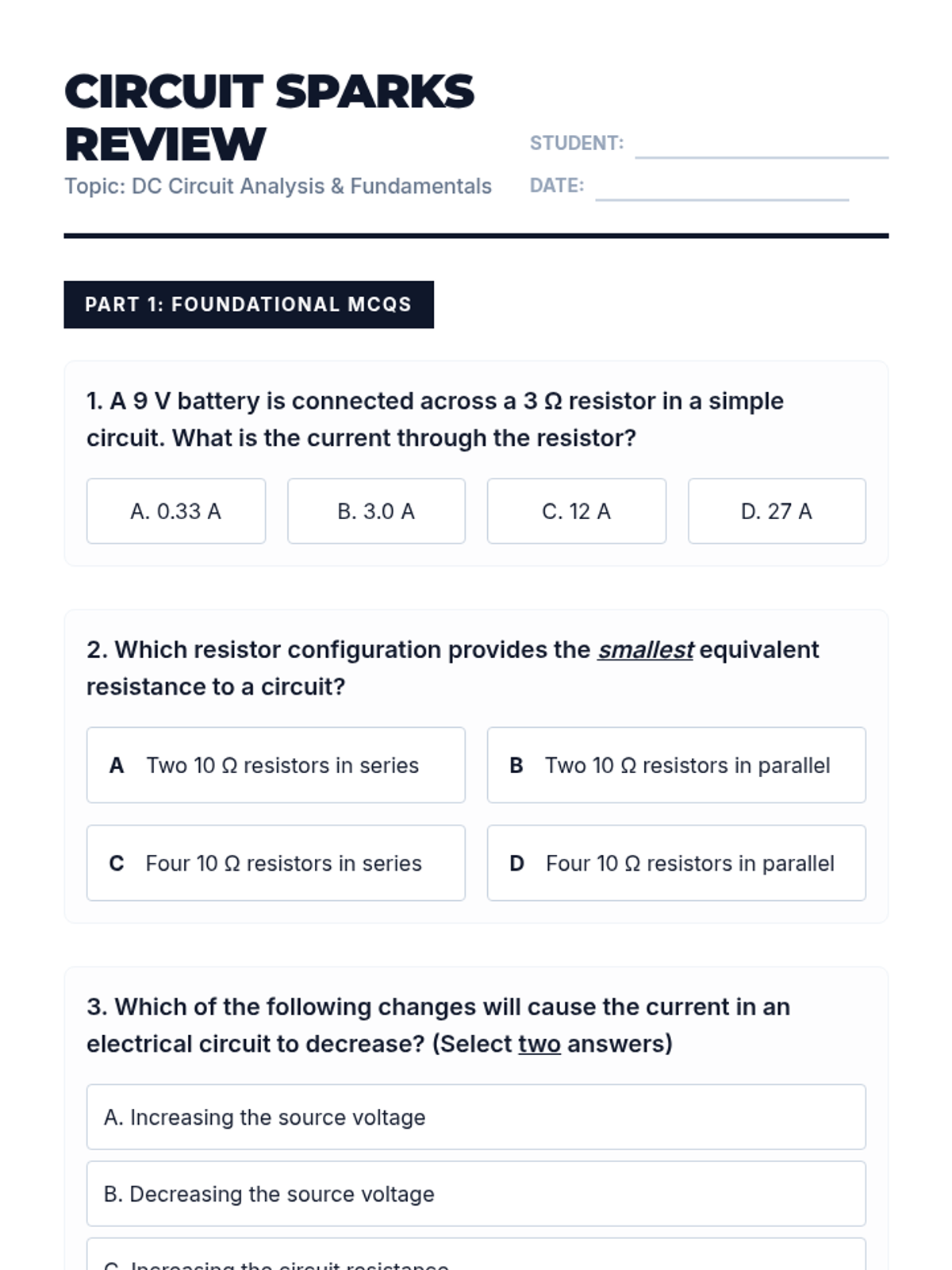
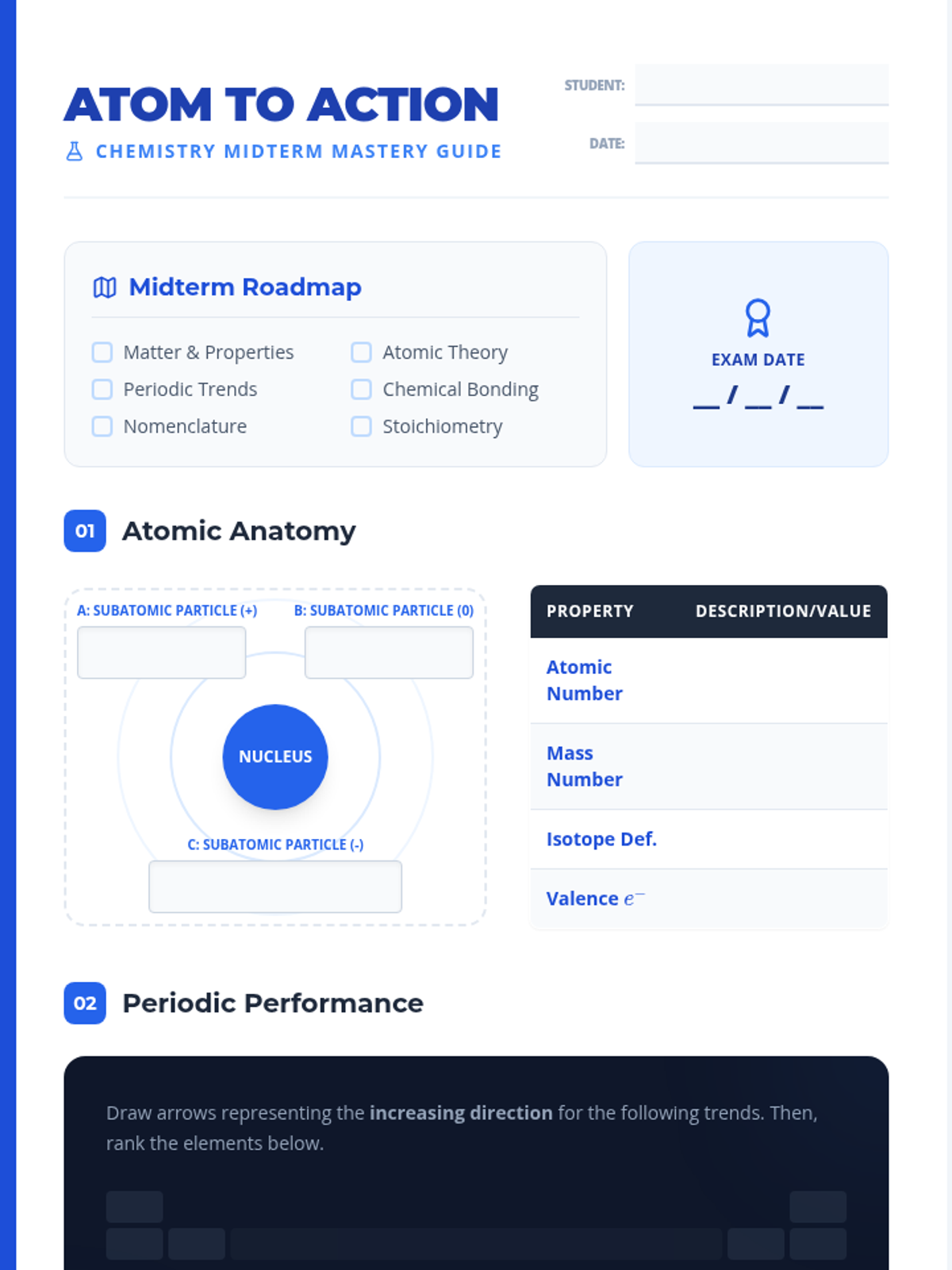
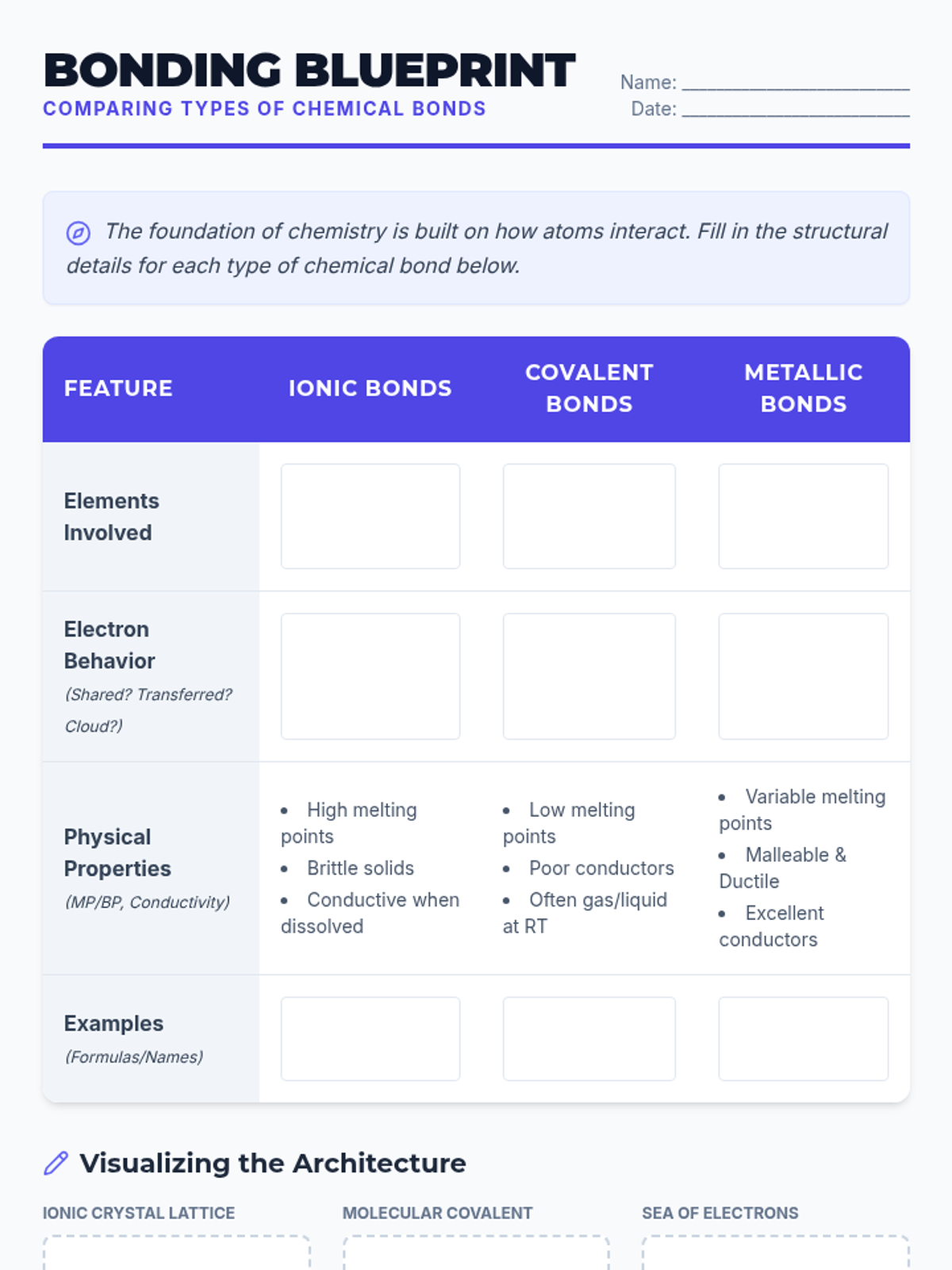
This undergraduate chemistry sequence covers the fundamental principles of chemical bonding, from electronegativity gradients and Lewis structure construction to formal charge analysis, resonance, and bond energetics. Students move from basic electron sharing to modeling complex hypervalent molecules and calculating reaction enthalpies.