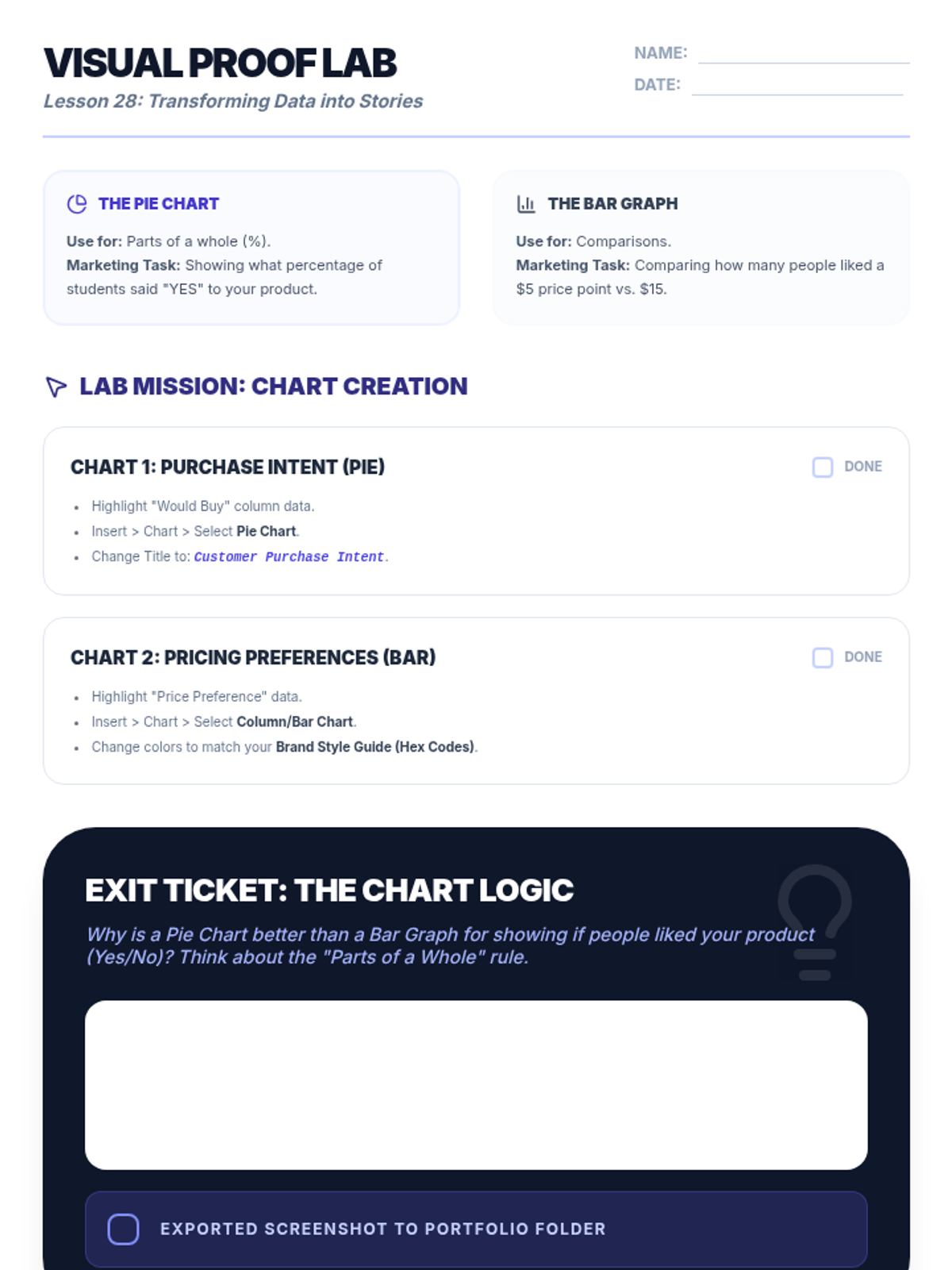
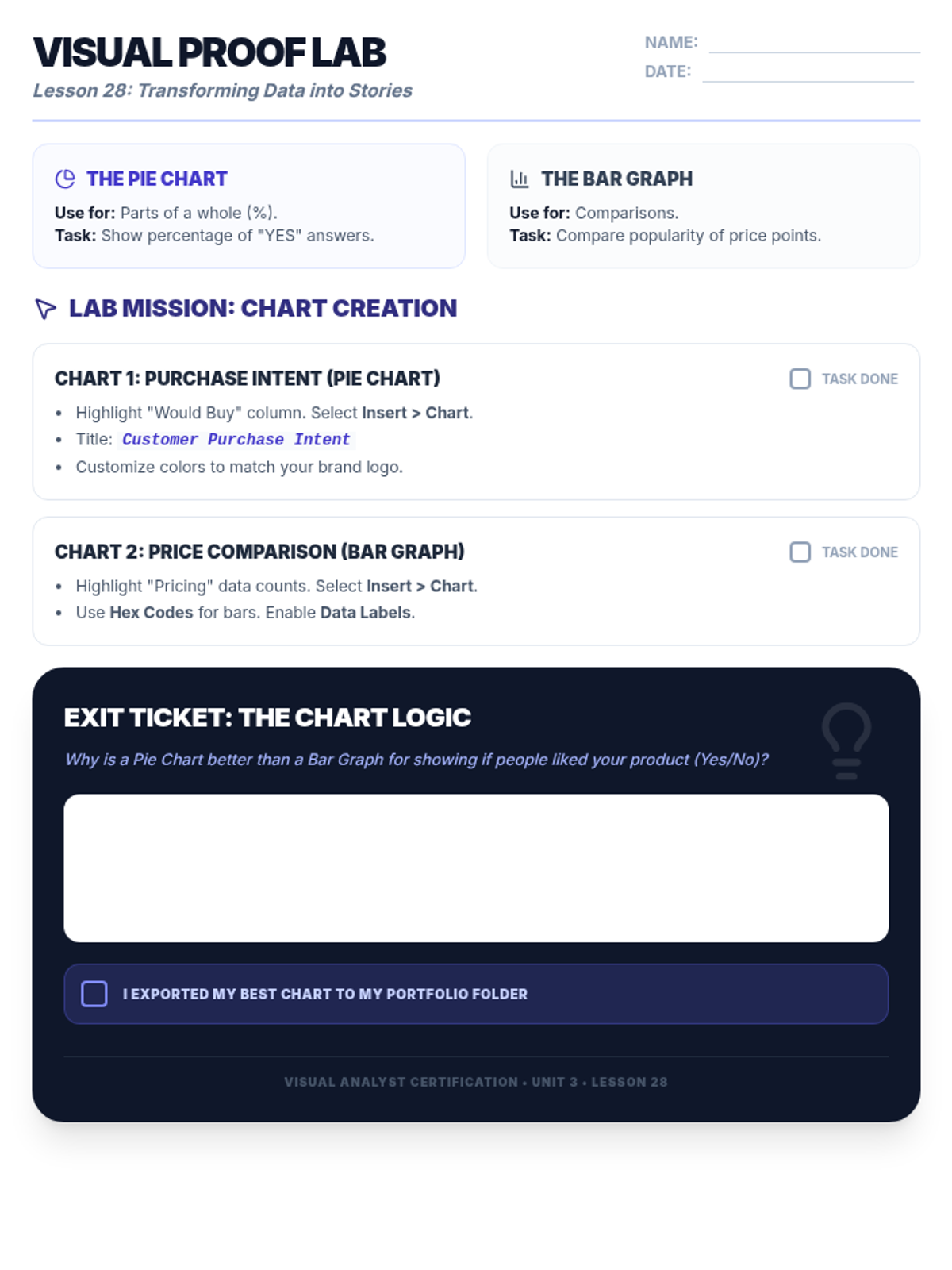
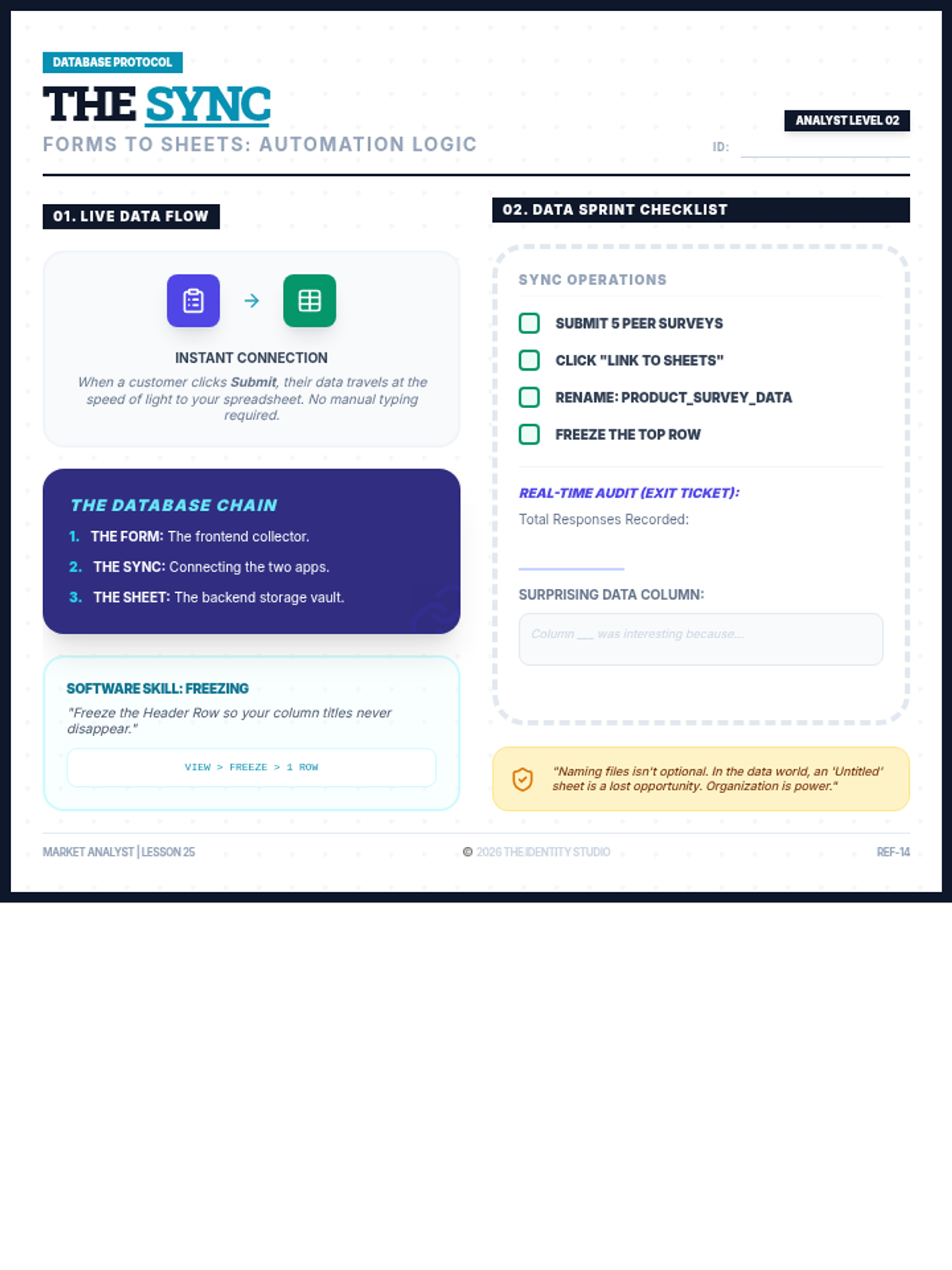
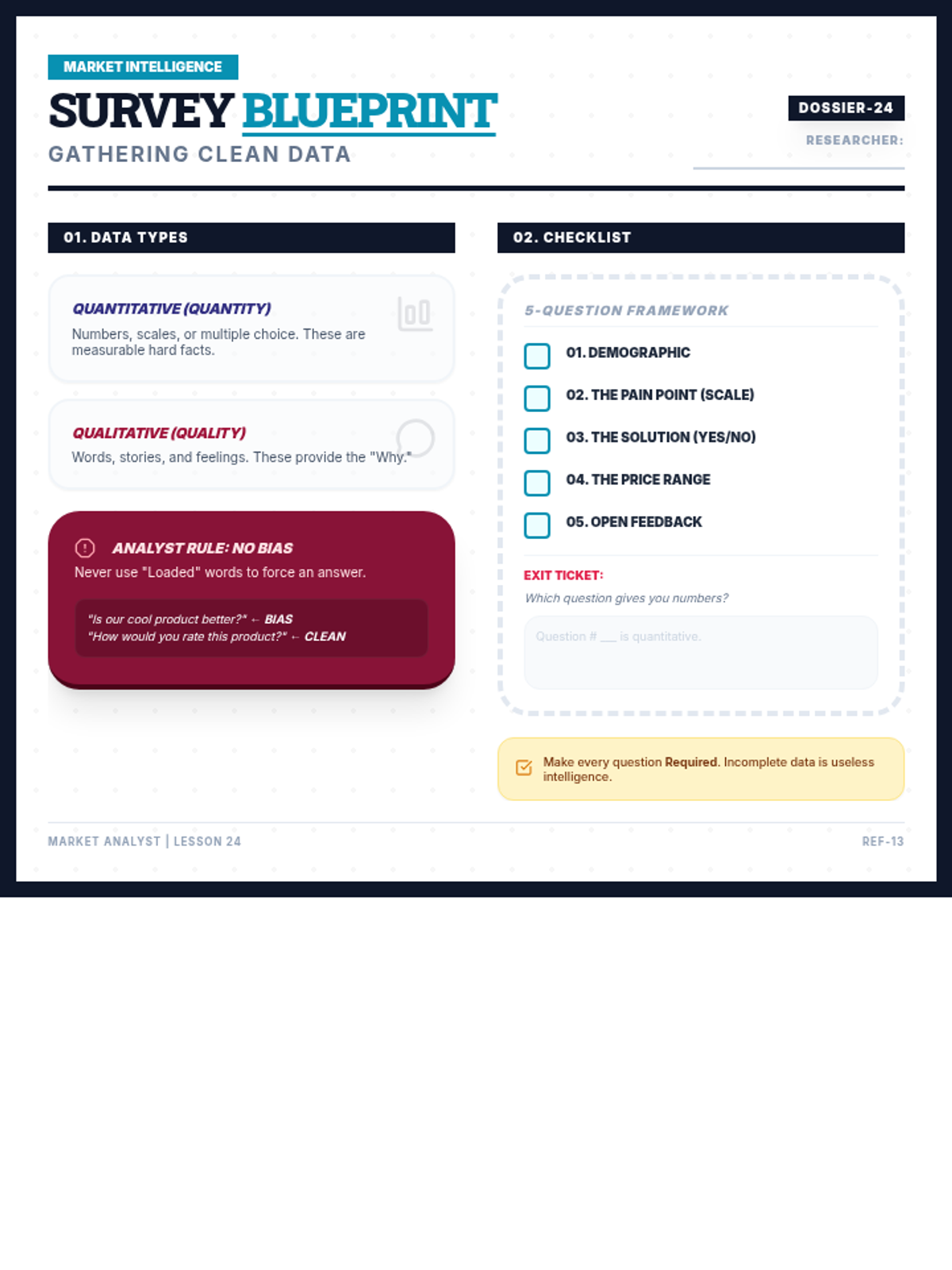
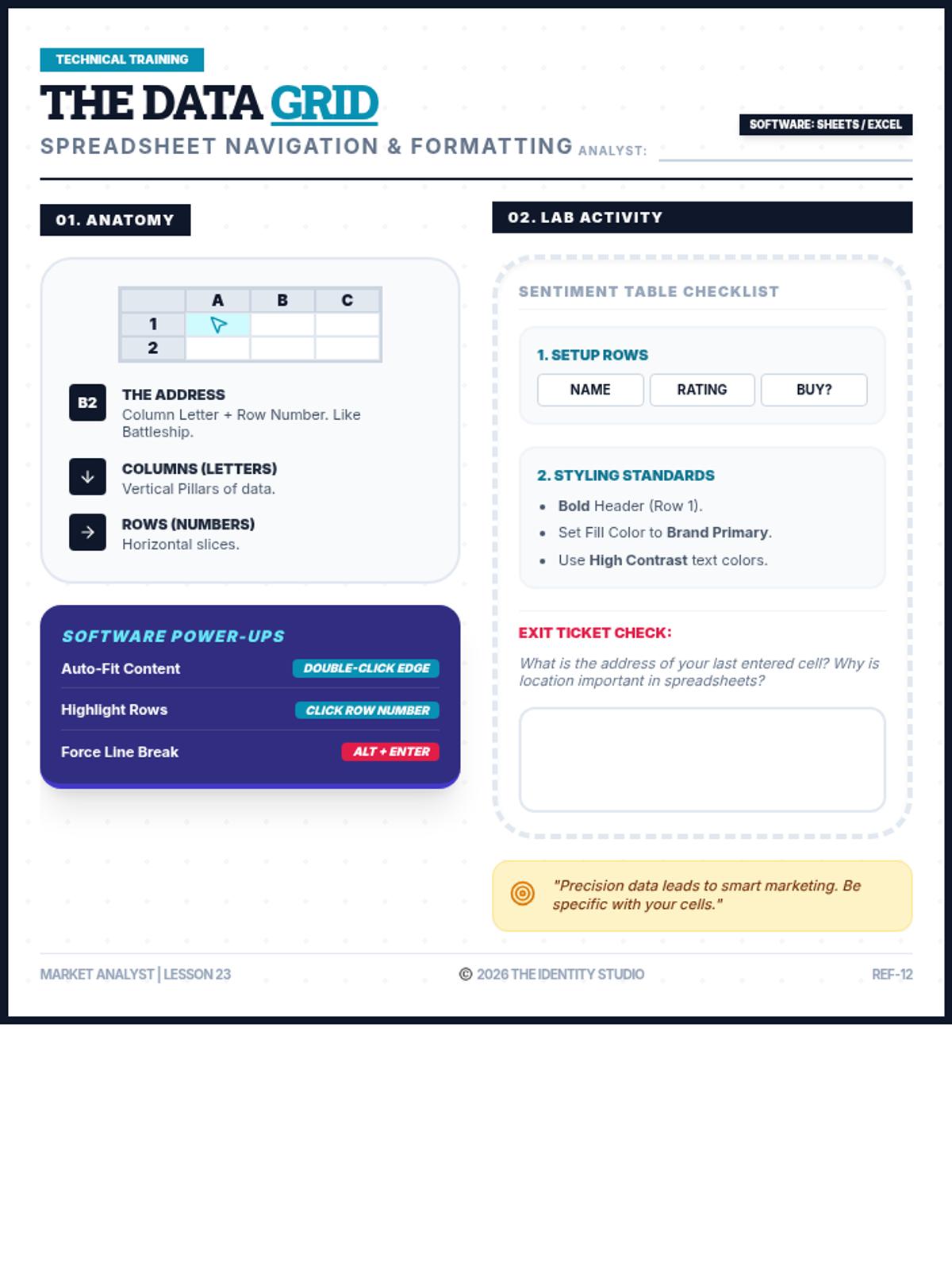
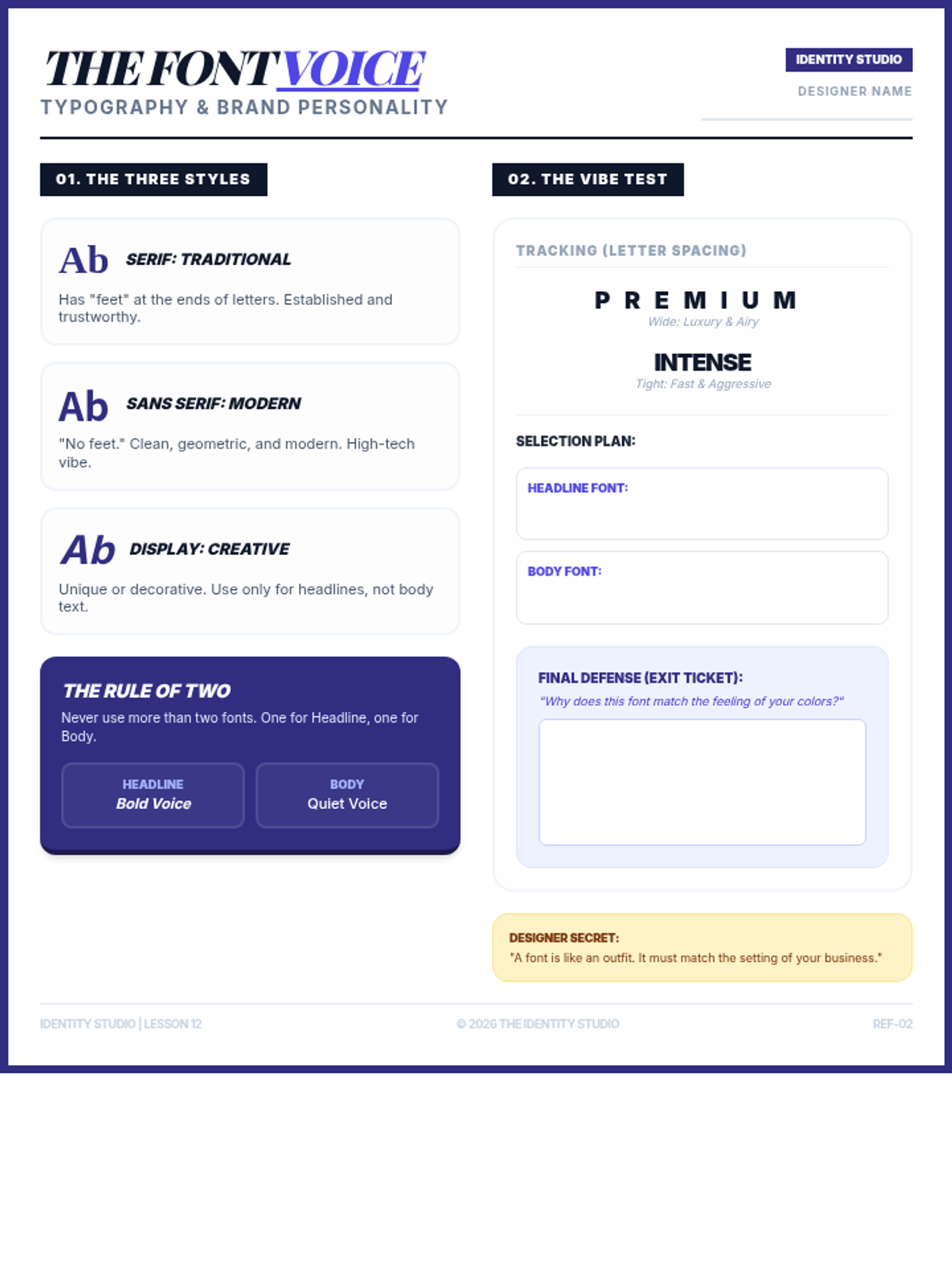
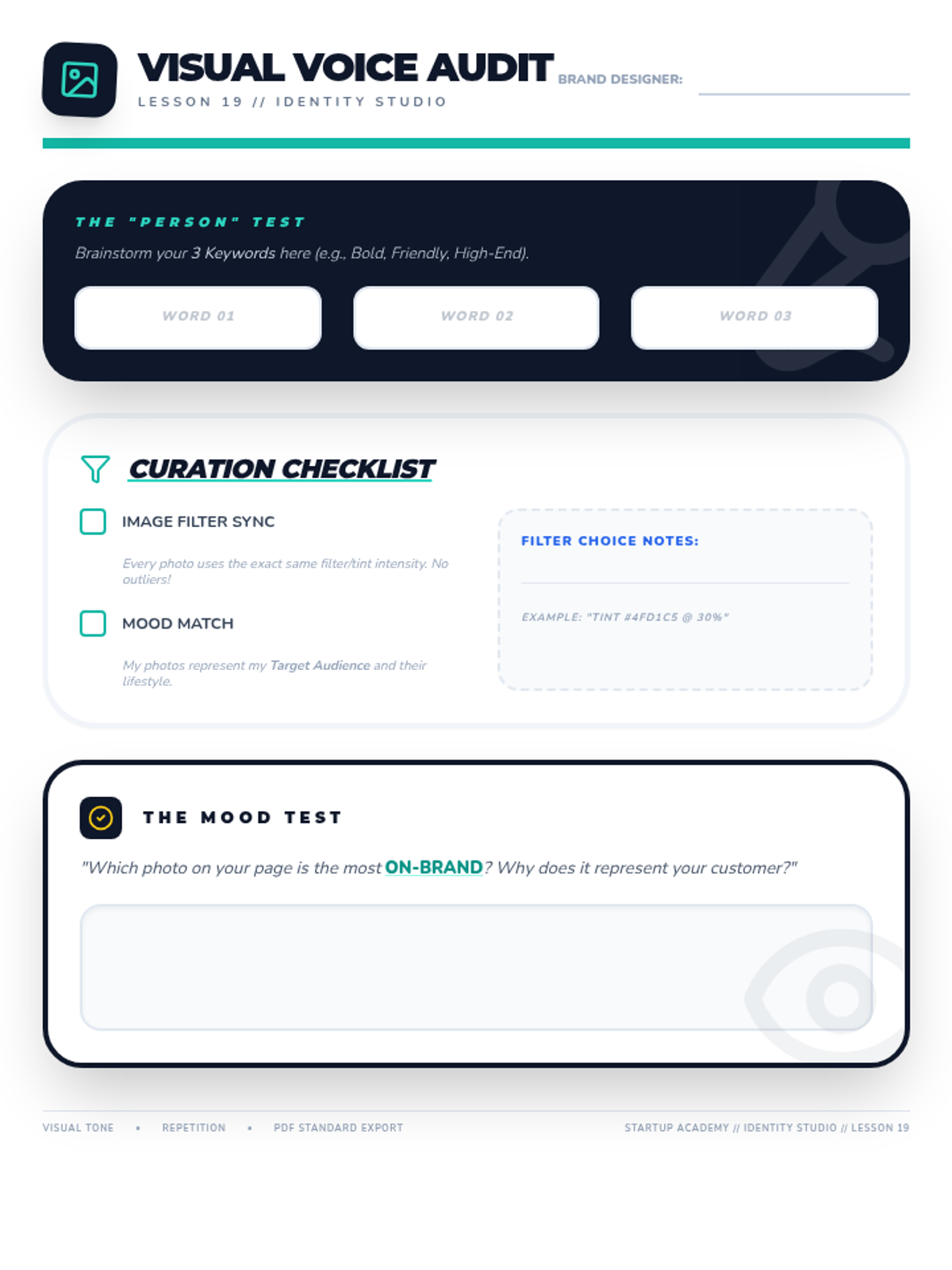
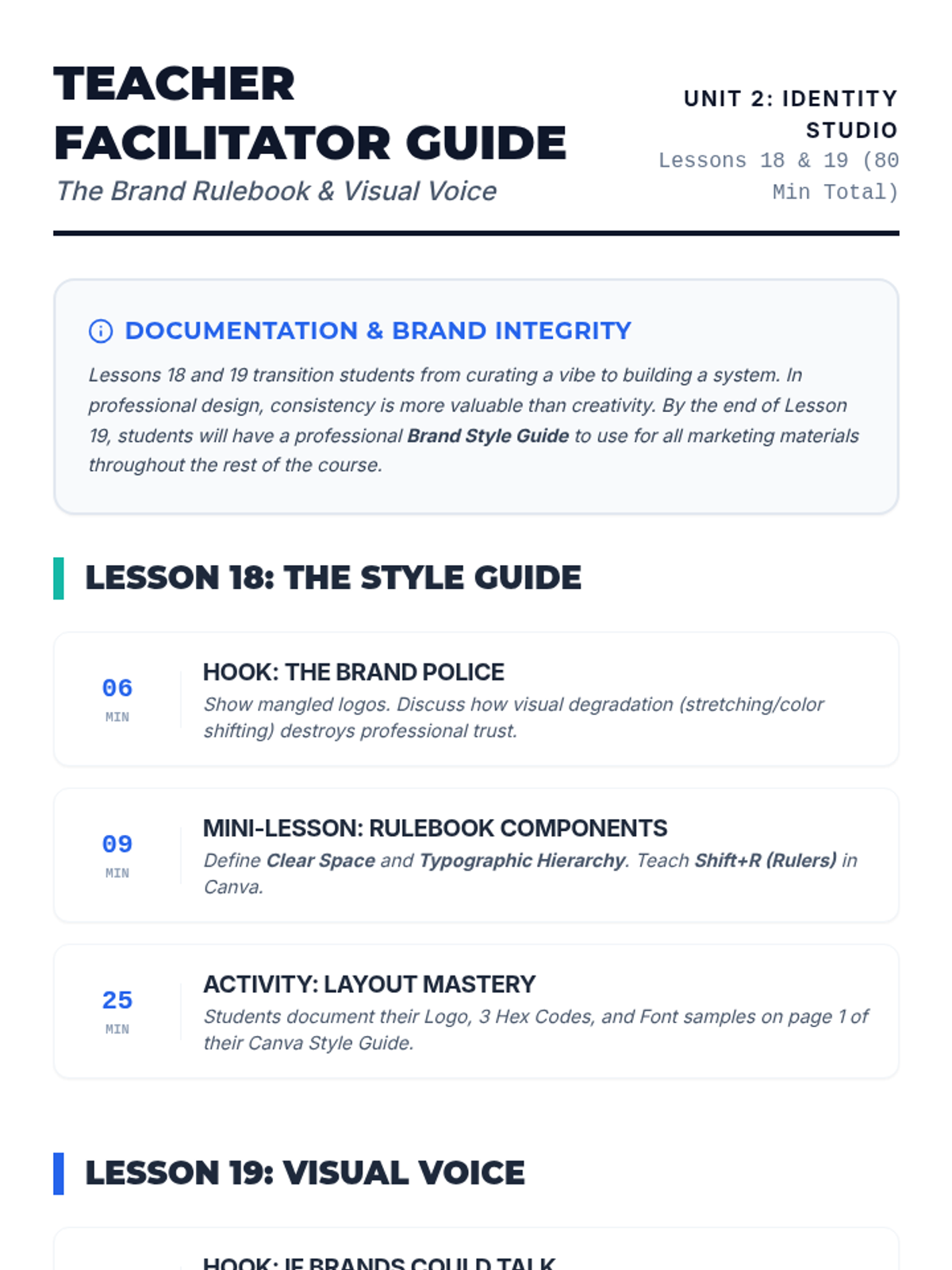
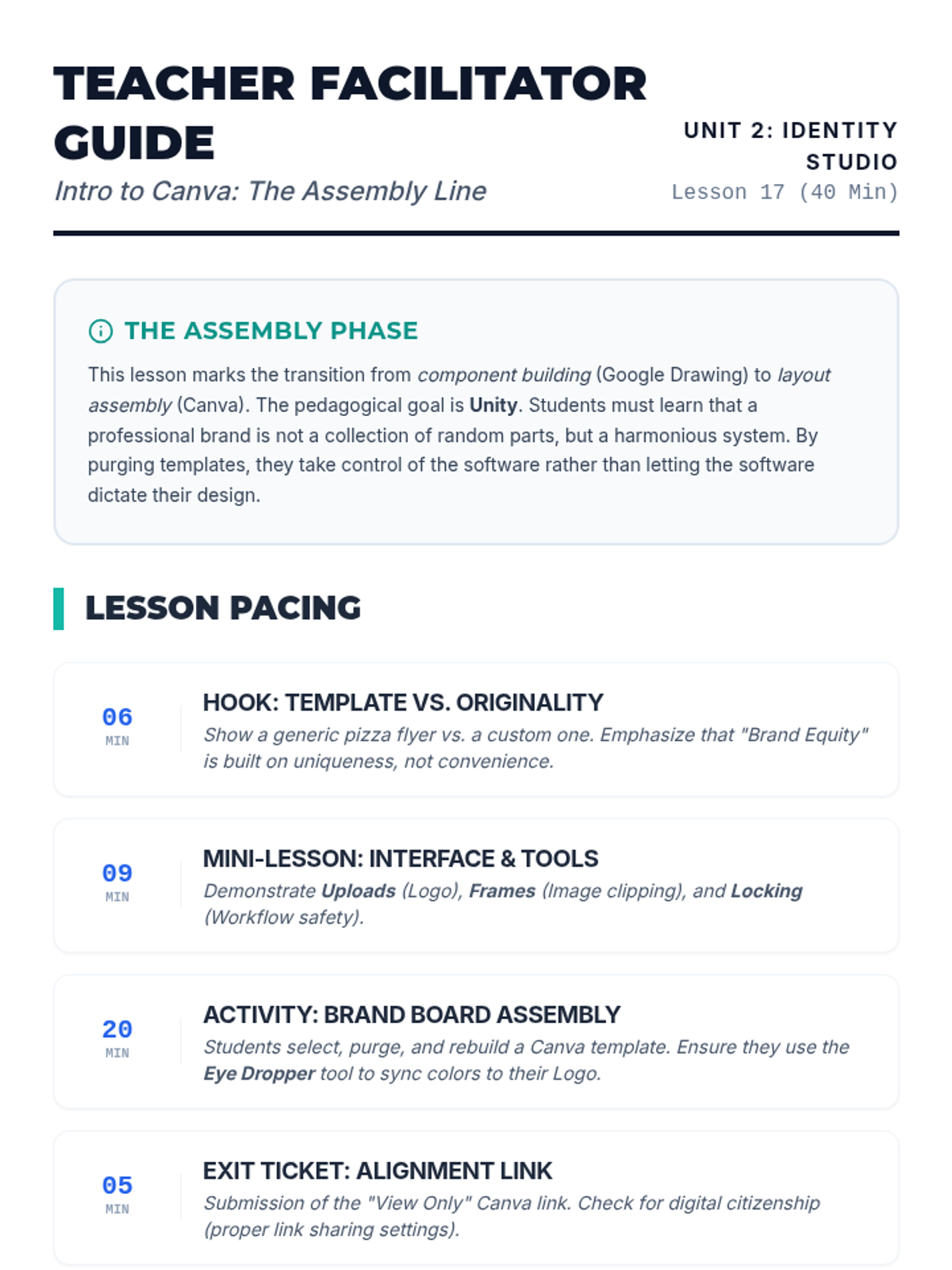
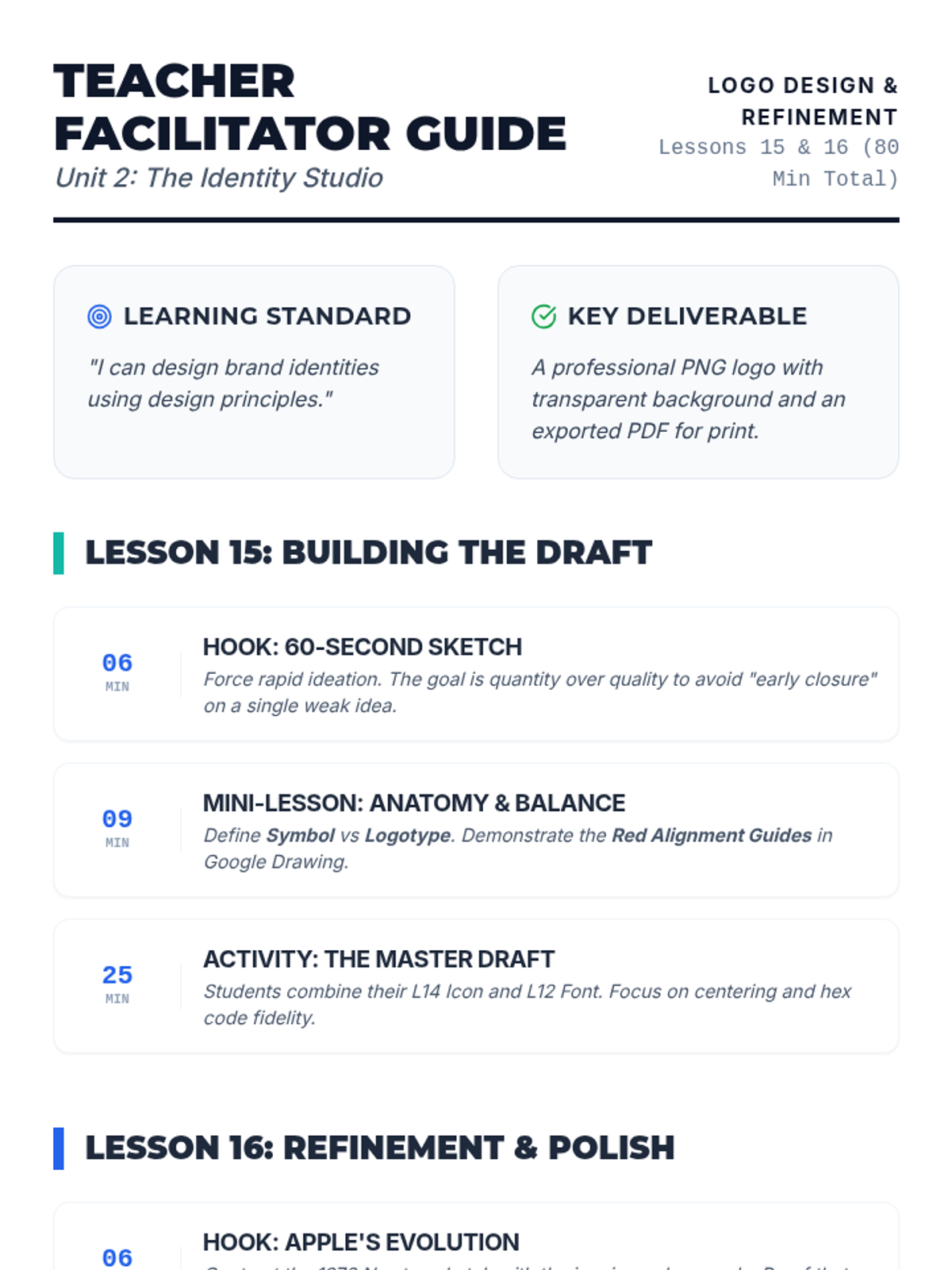
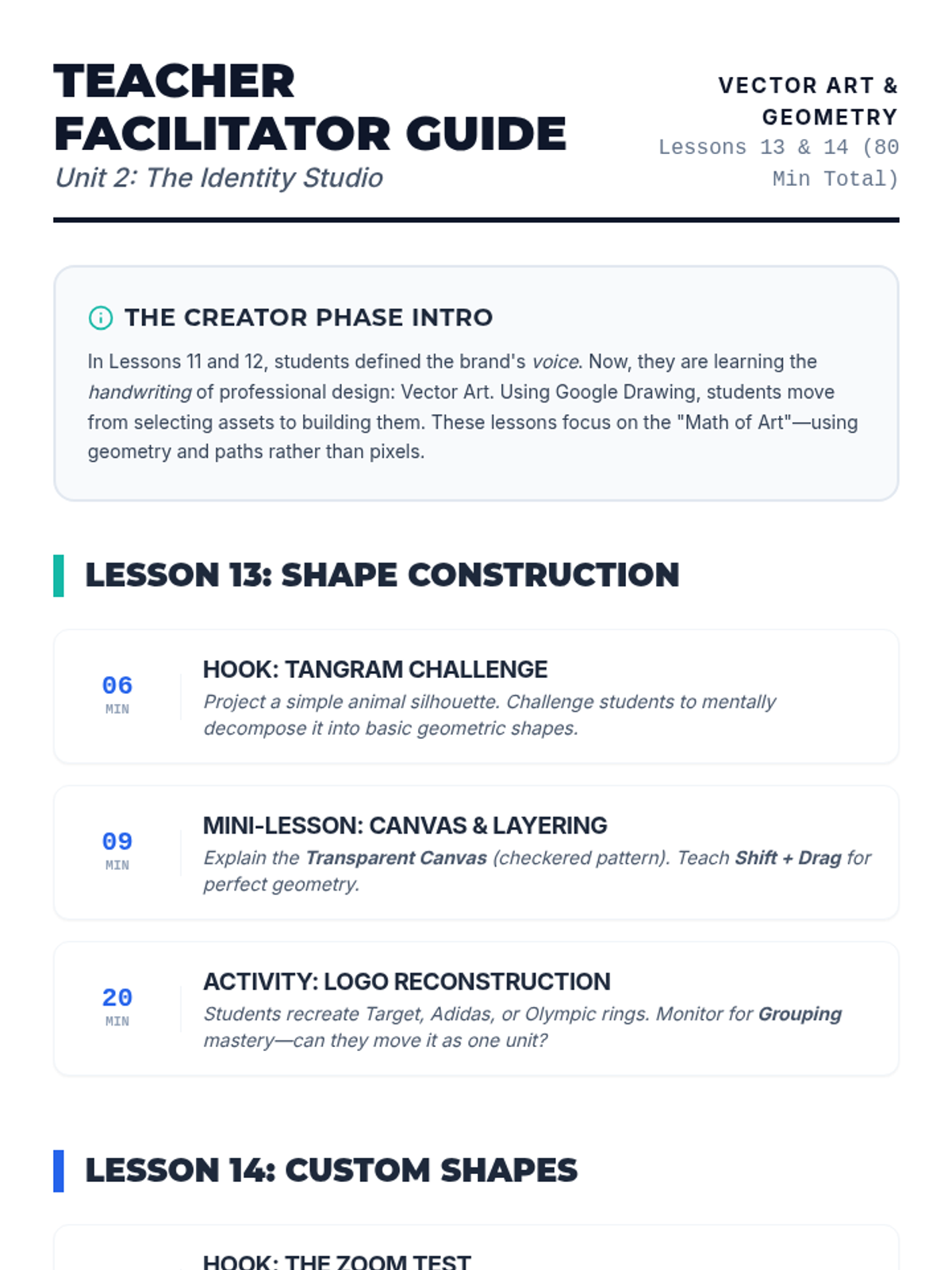
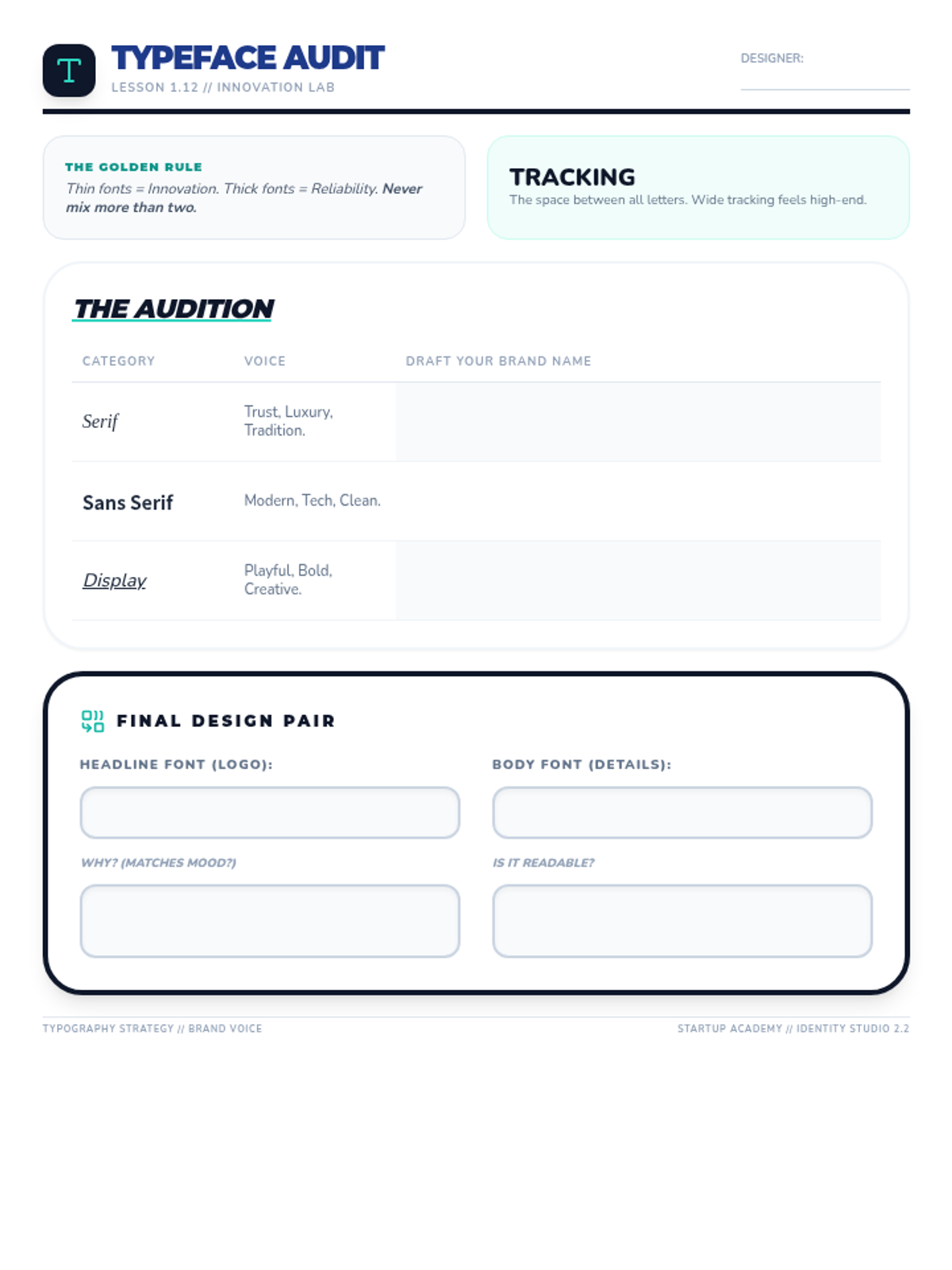
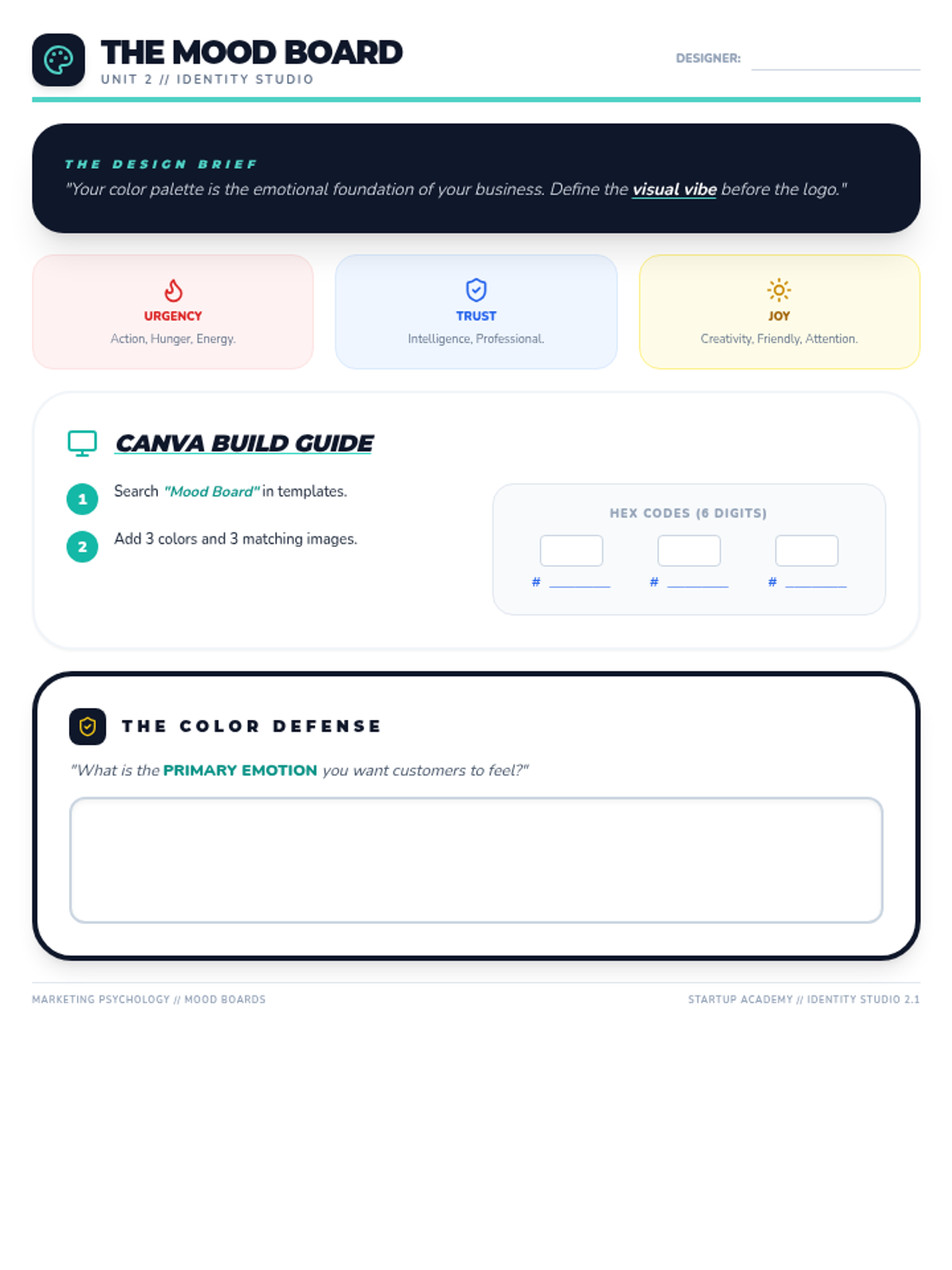
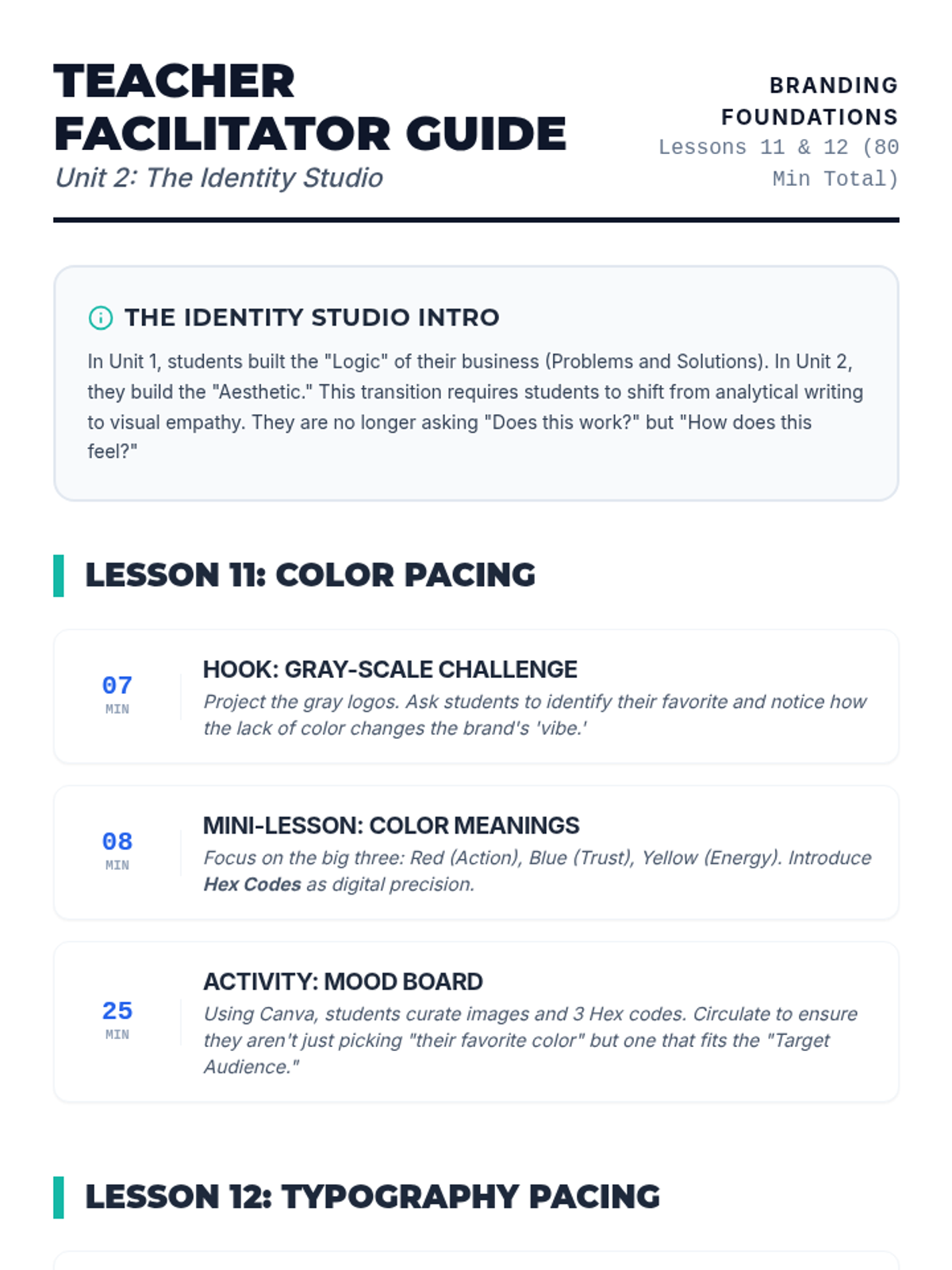
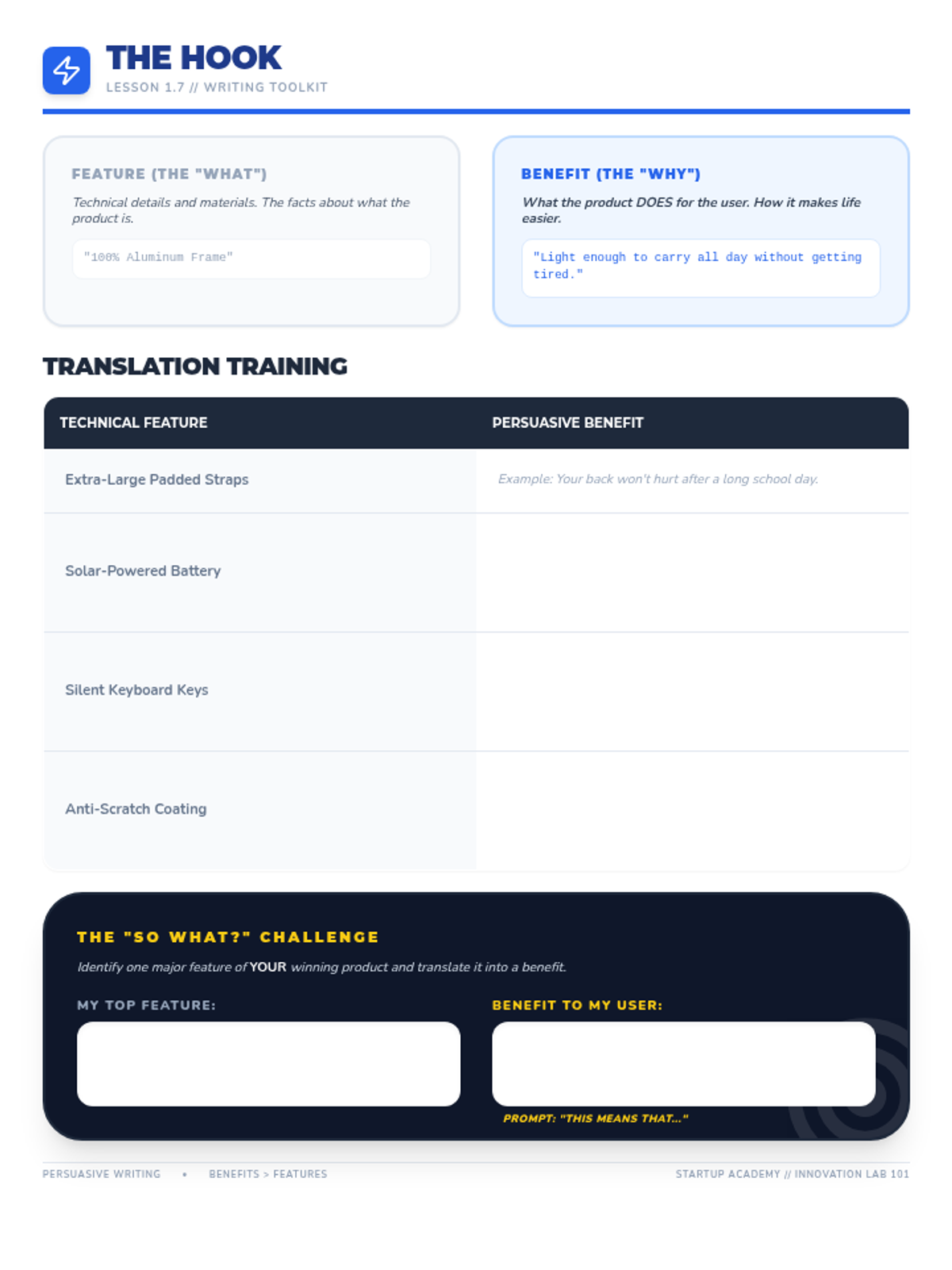
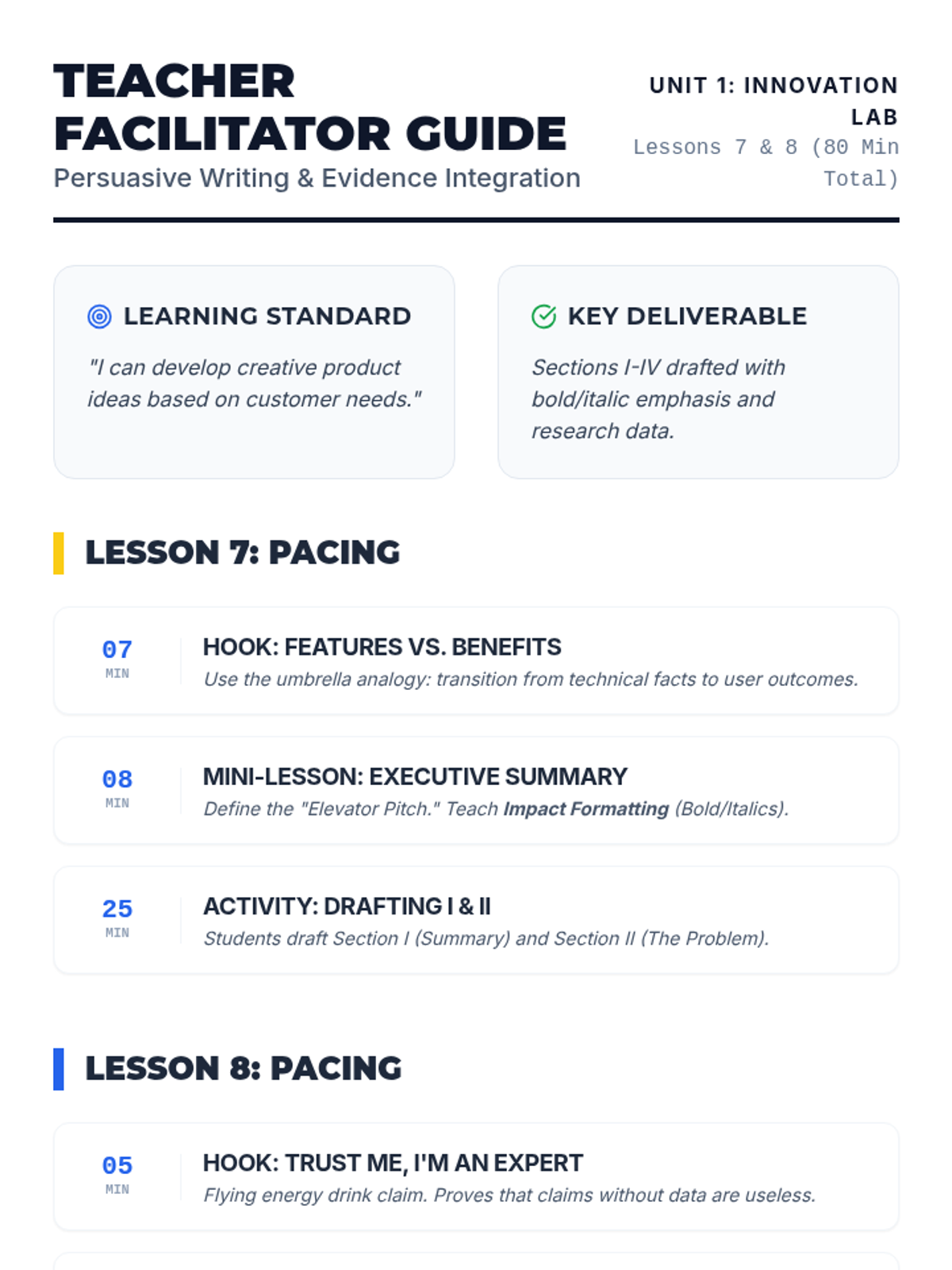
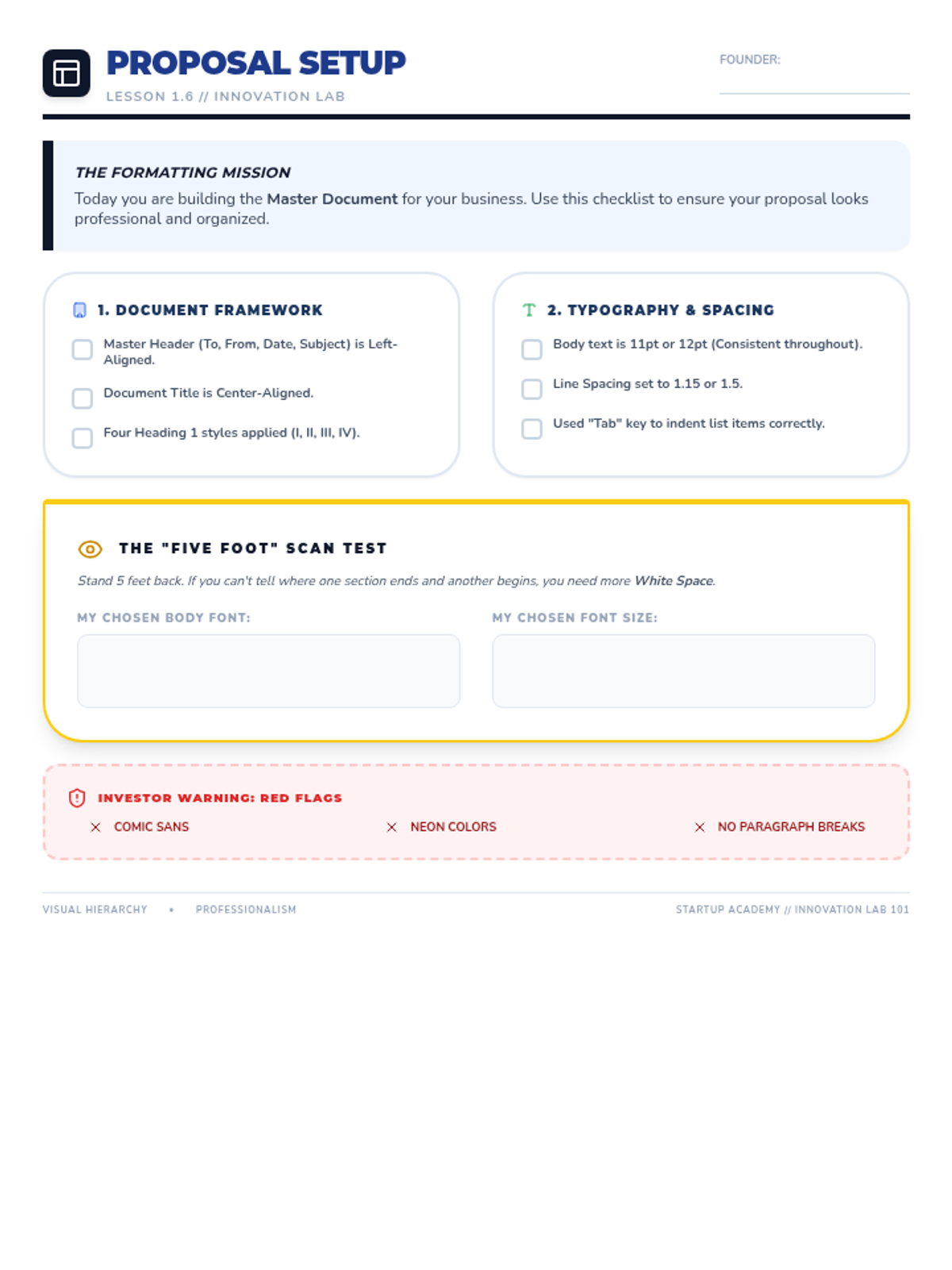
Students explore the concept of buffers—solutions that resist changes in pH. They compare how tap water vs. a buffered solution reacts to the addition of acid. This lesson bridges the gap between industrial chemistry and the homeostasis required in the human body (blood pH).