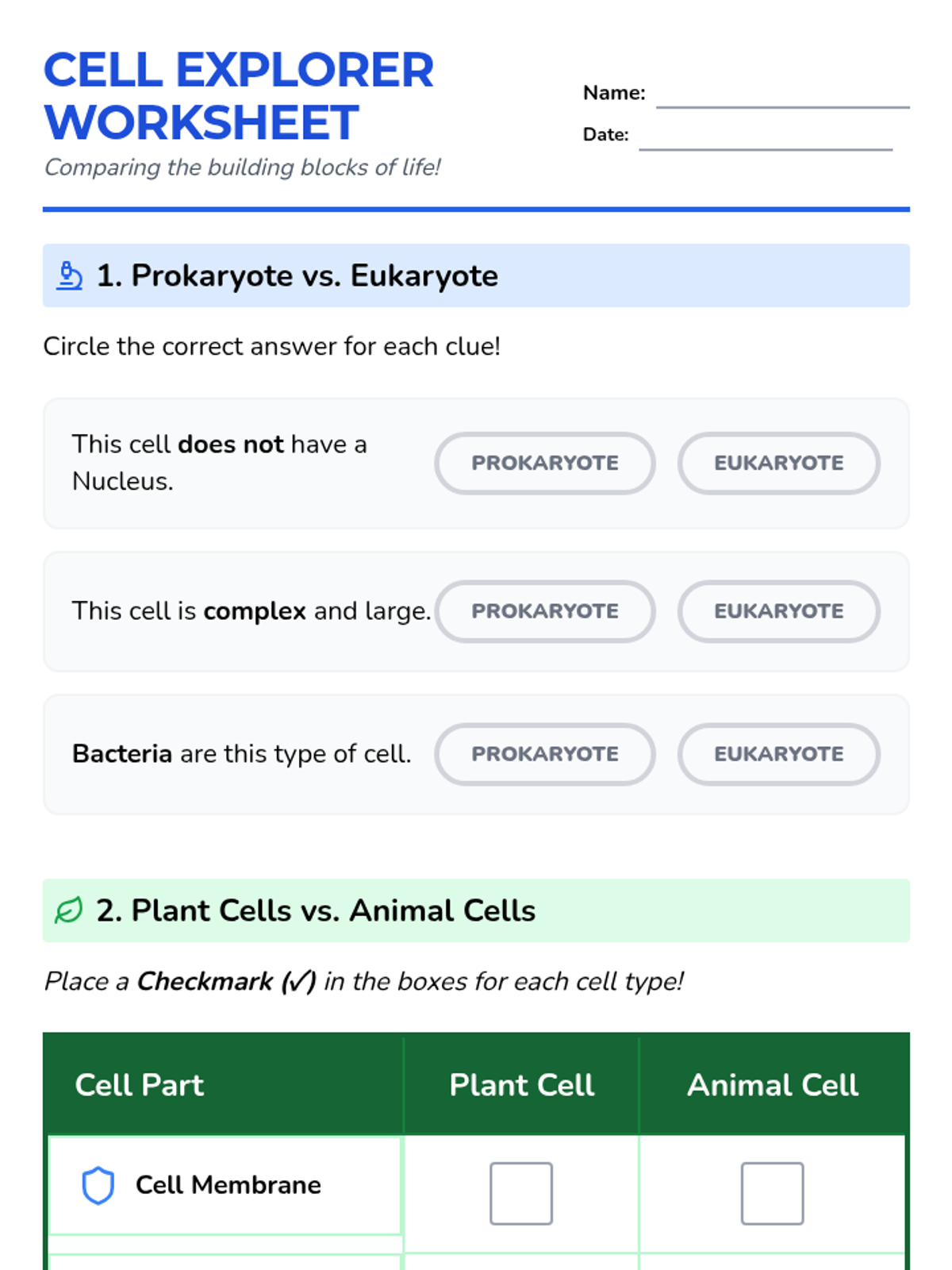
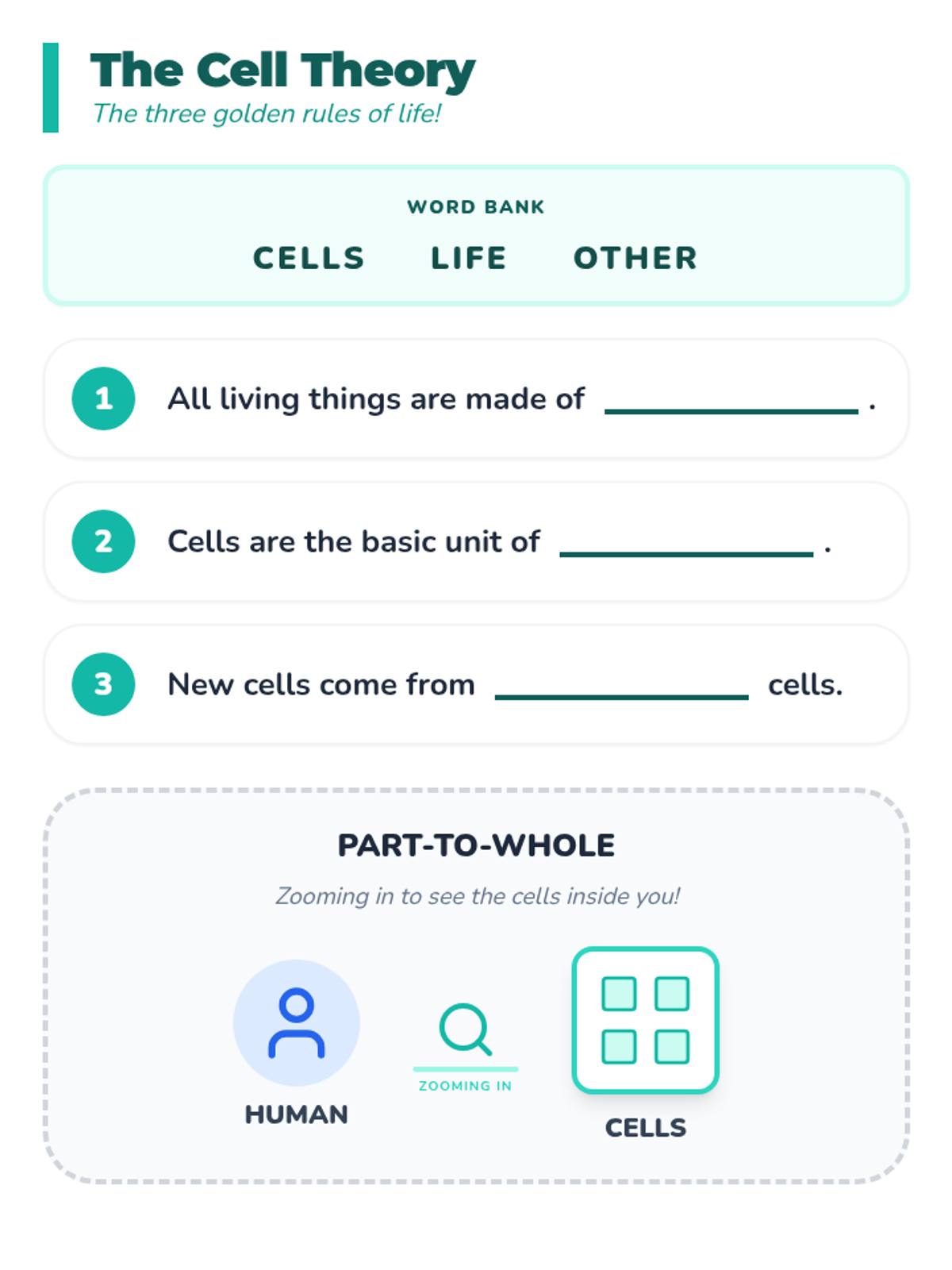
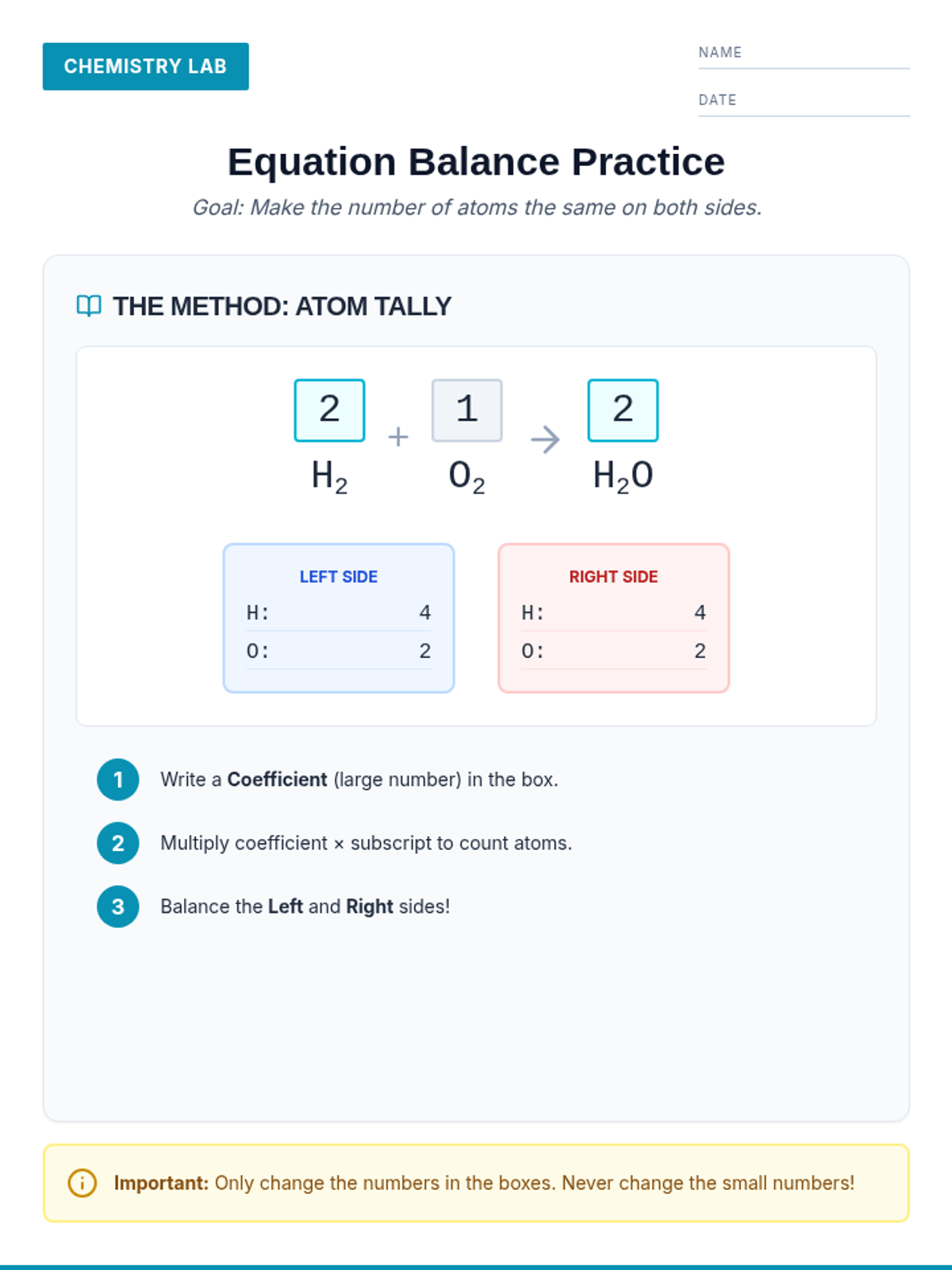
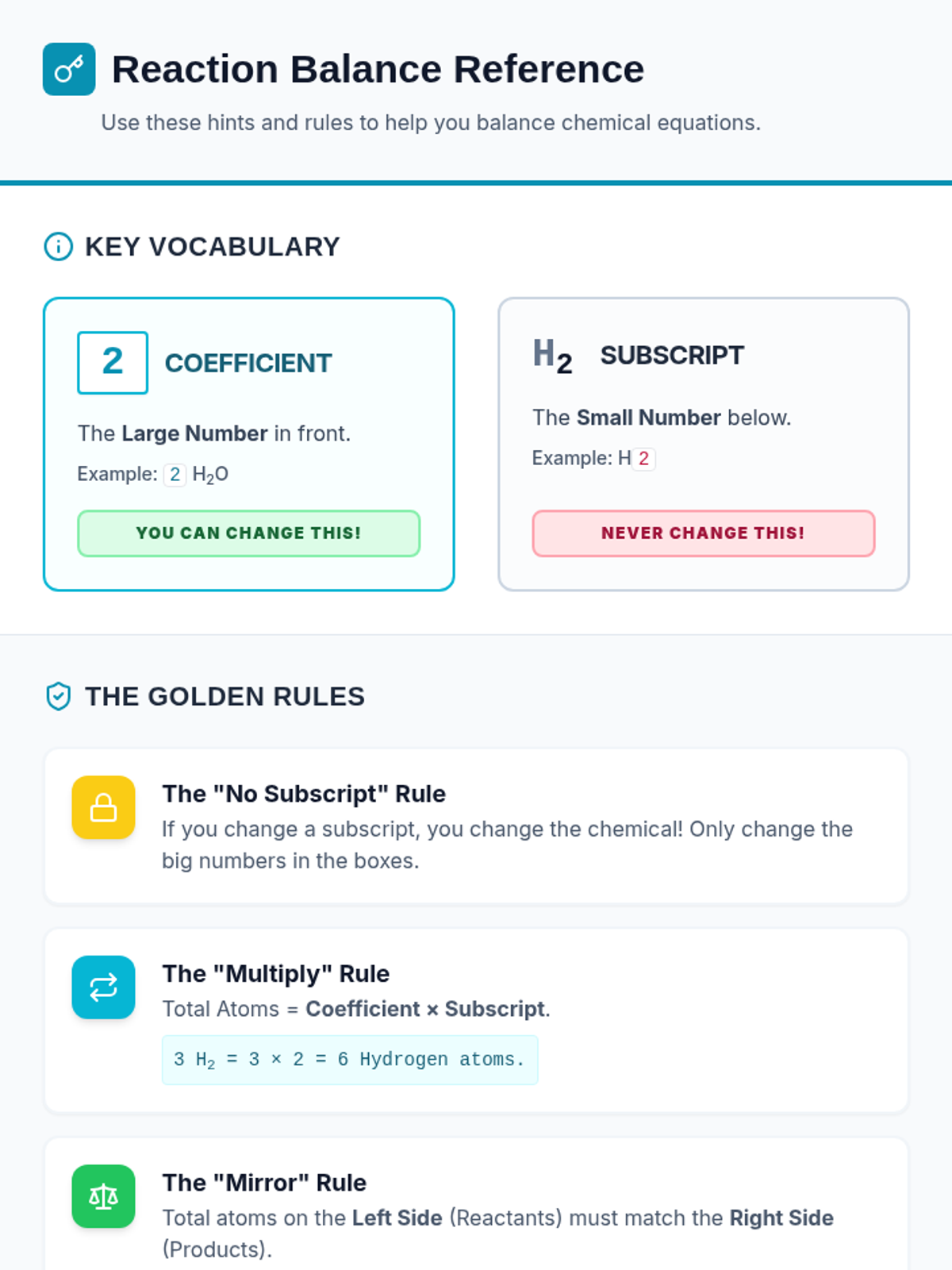
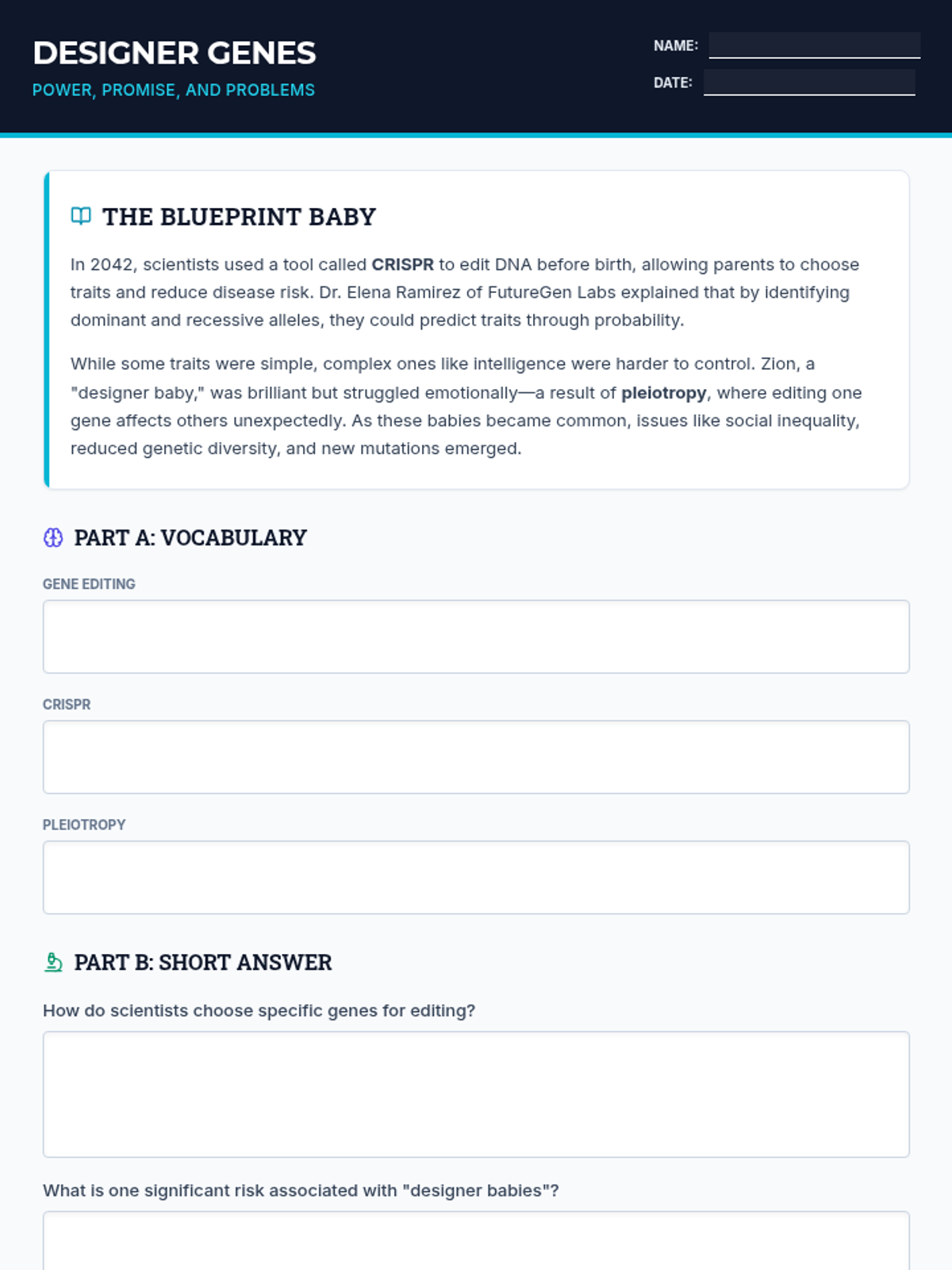
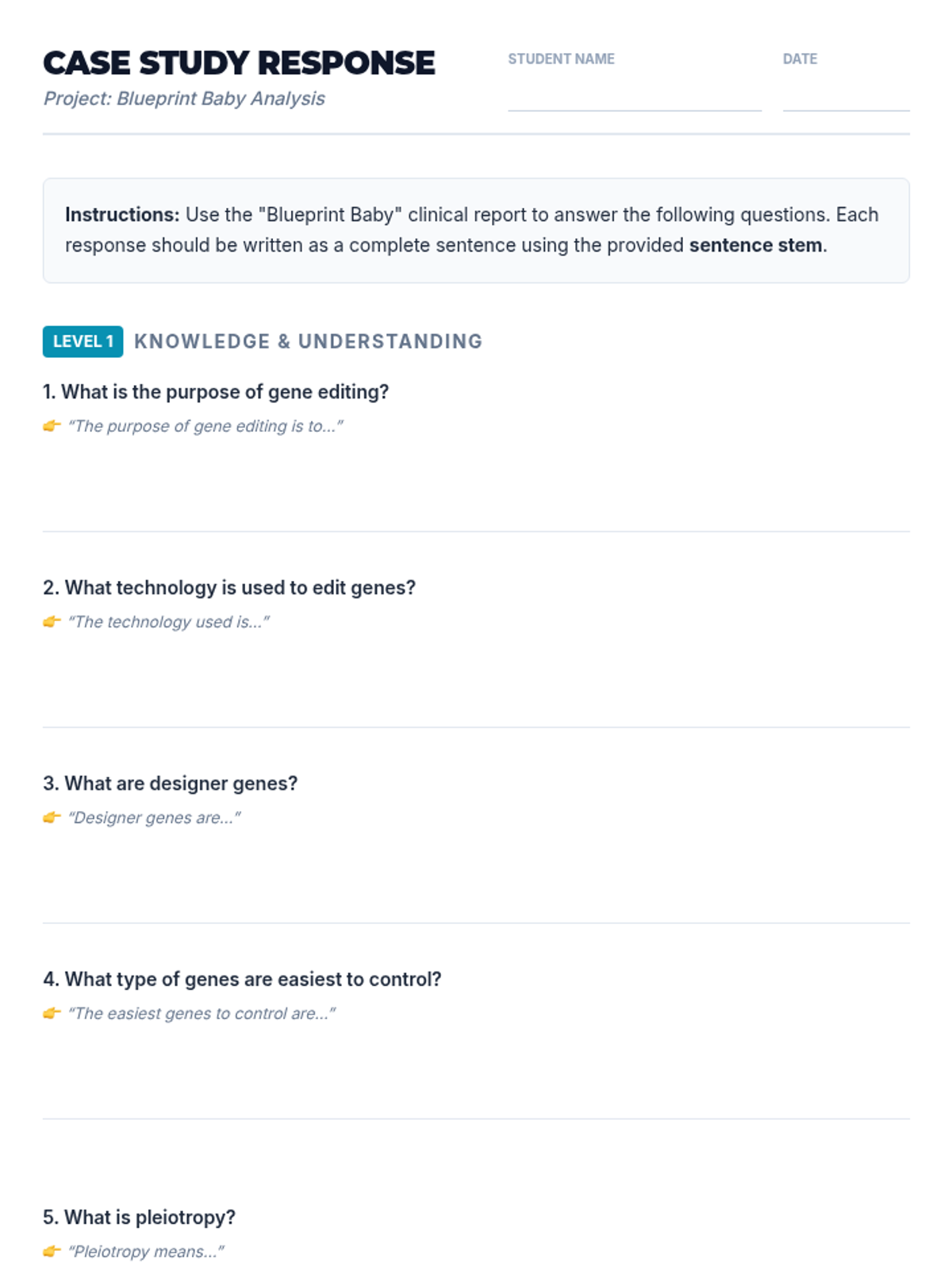
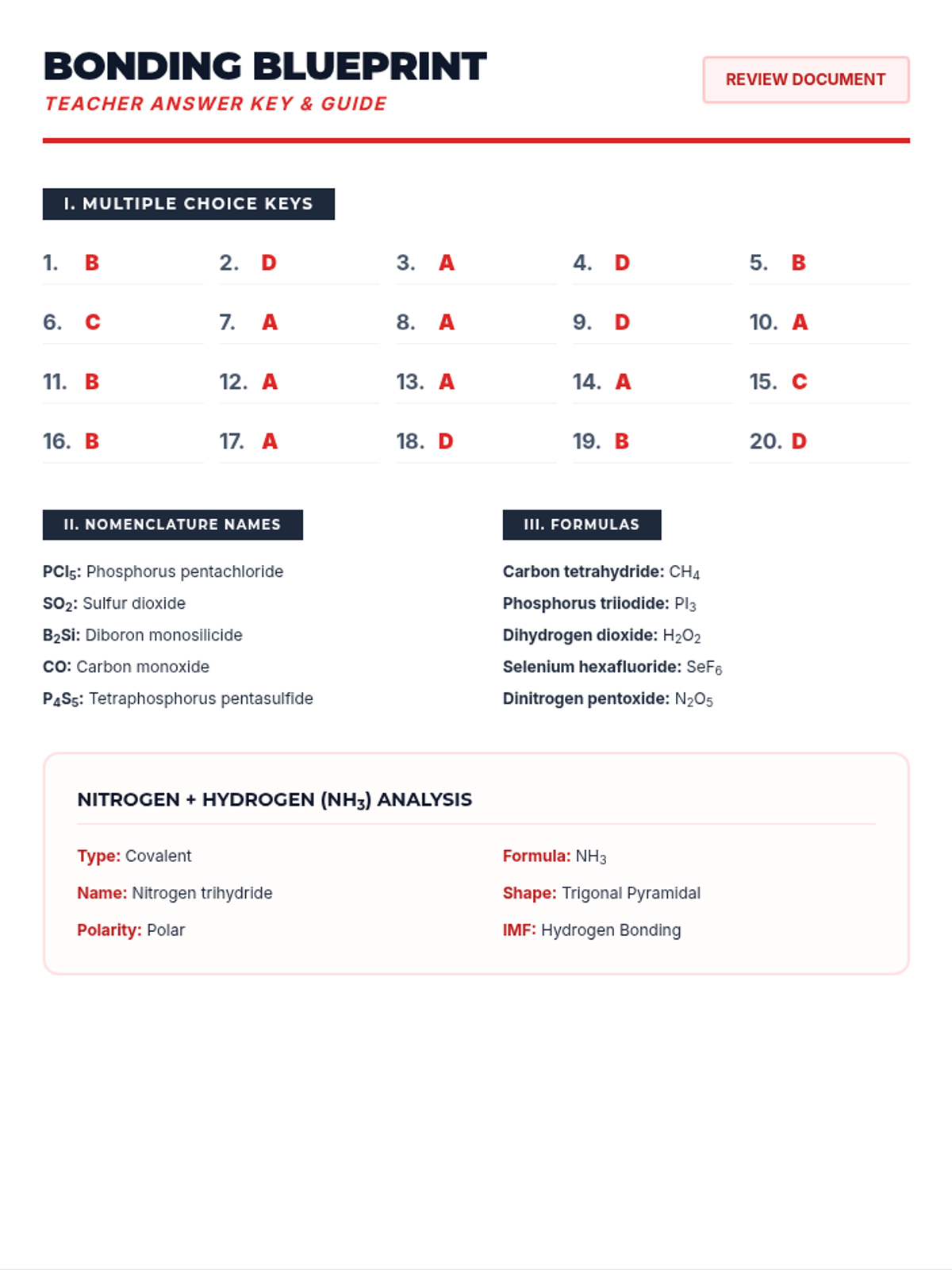
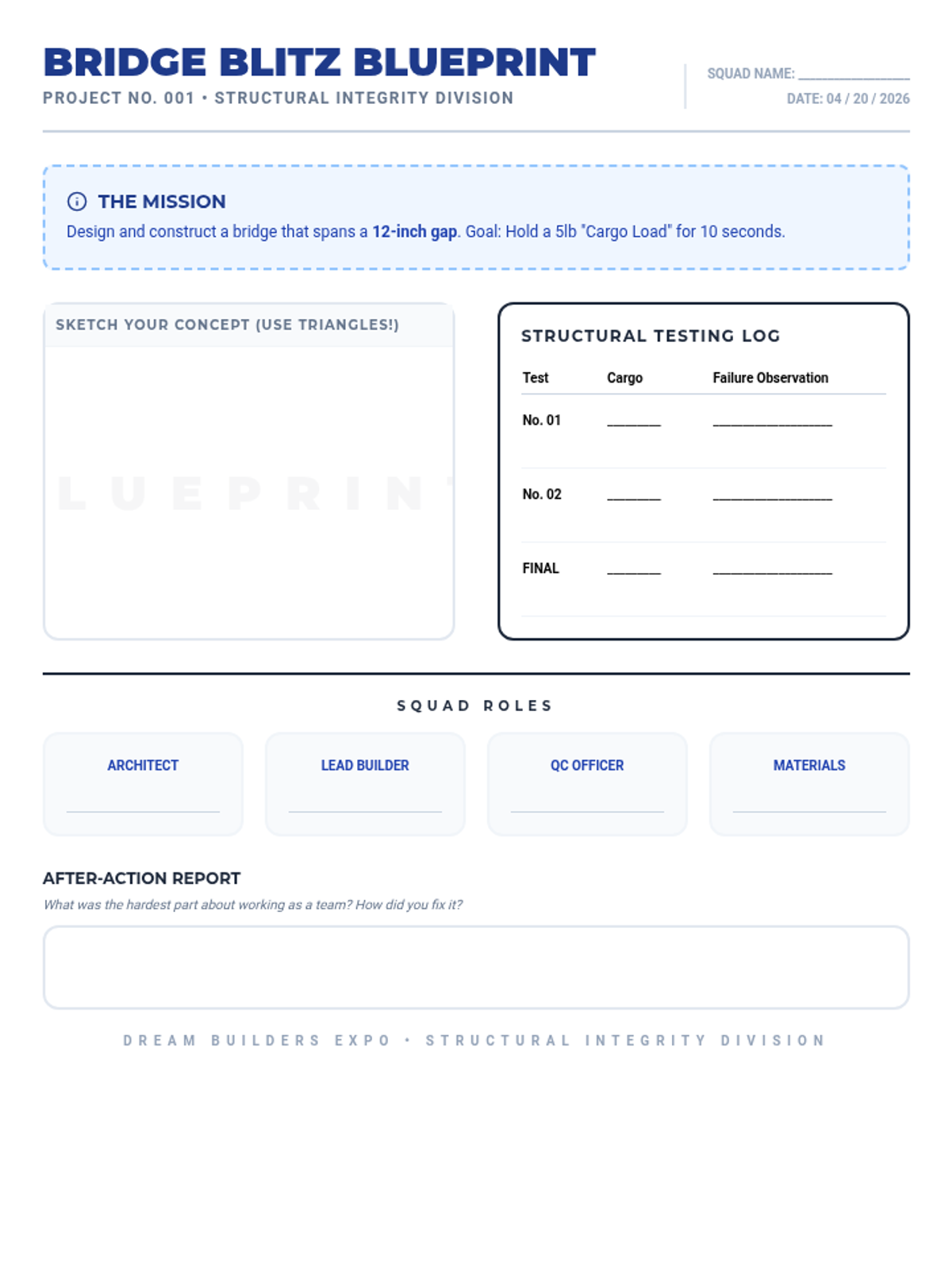
A high-energy, station-based lesson where students investigate the thermodynamics of phase changes. Students analyze intermolecular forces to explain why melting is endothermic and freezing is exothermic, using evidence from heating curves and molecular models.