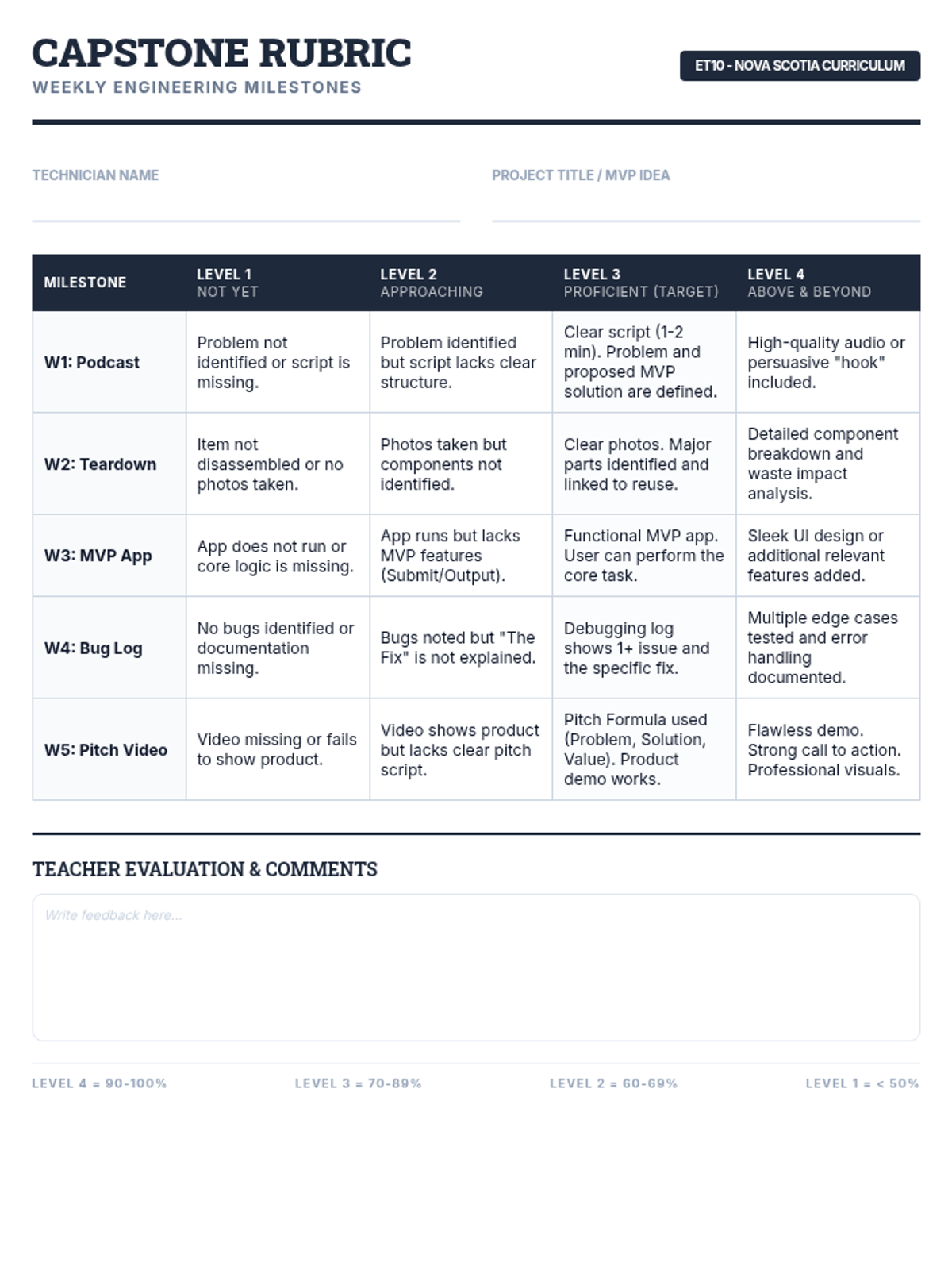
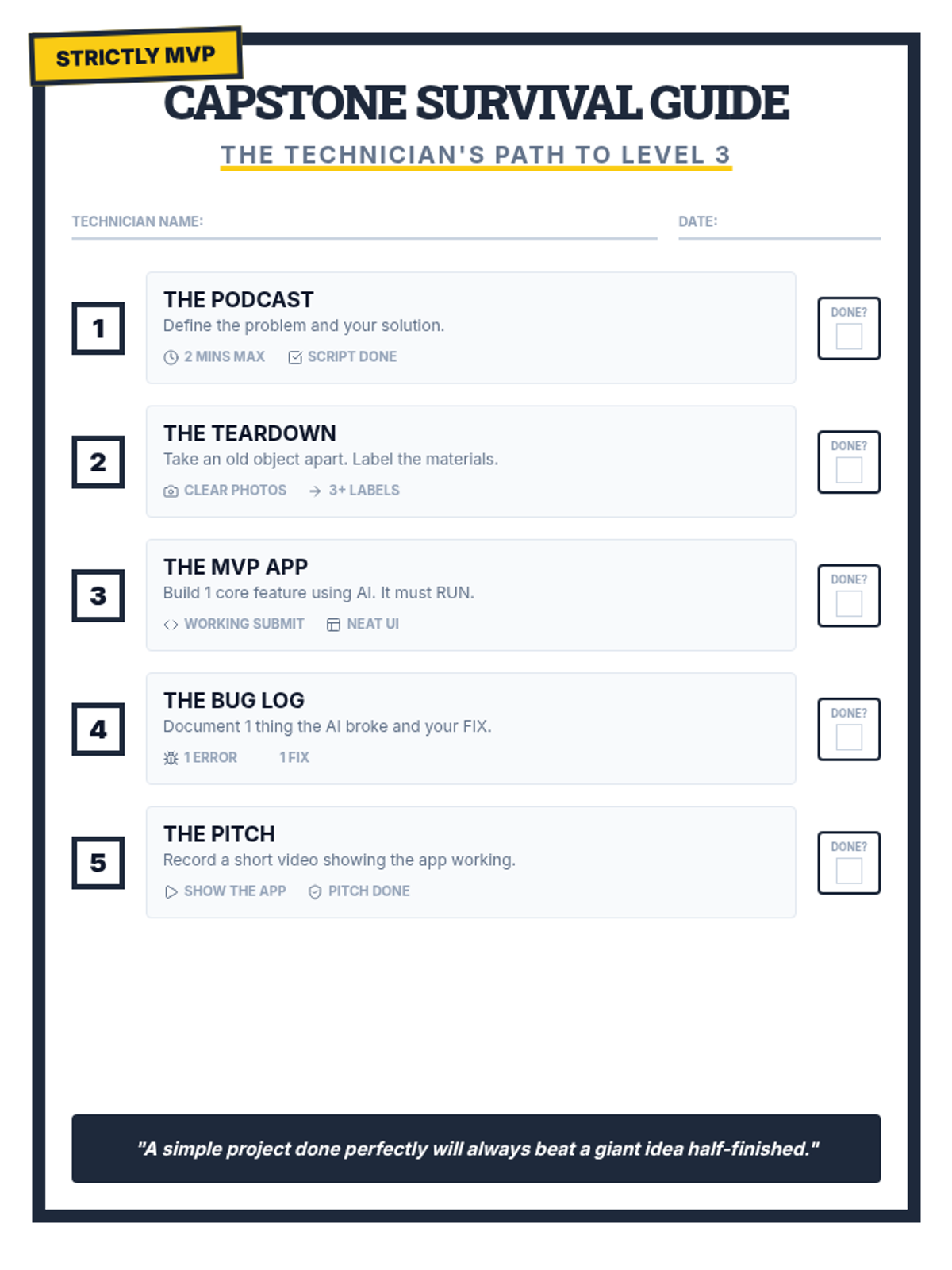
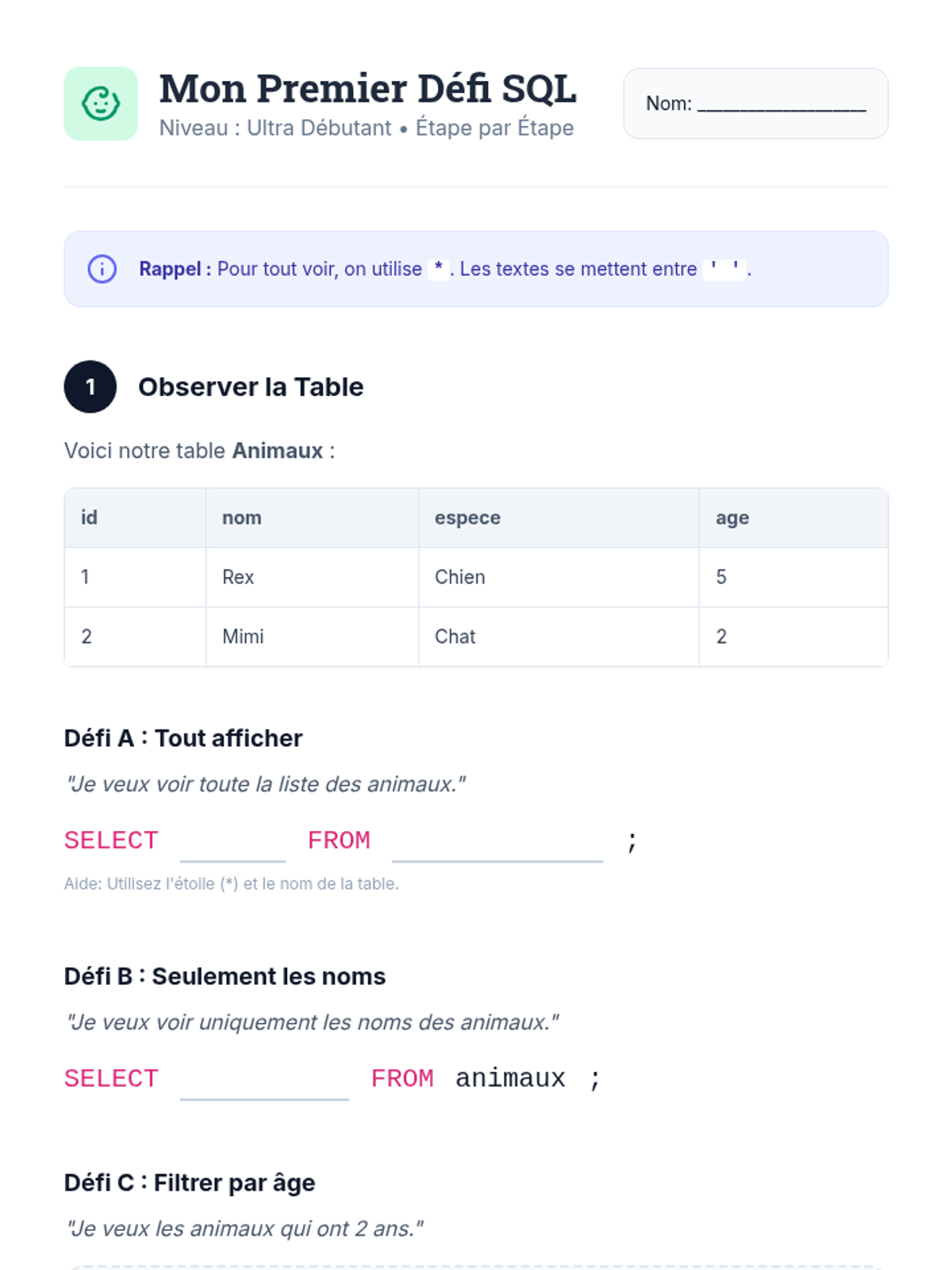
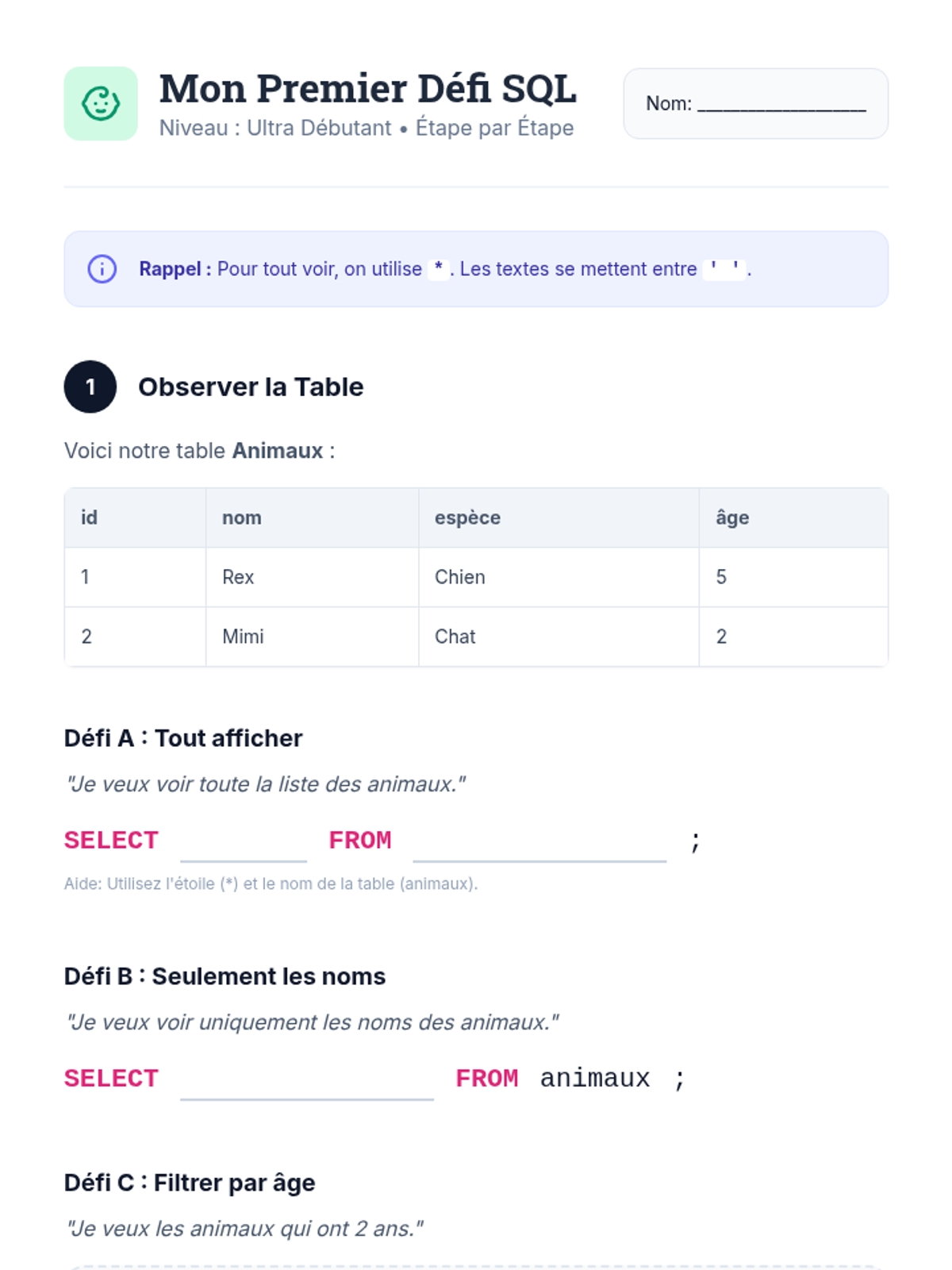
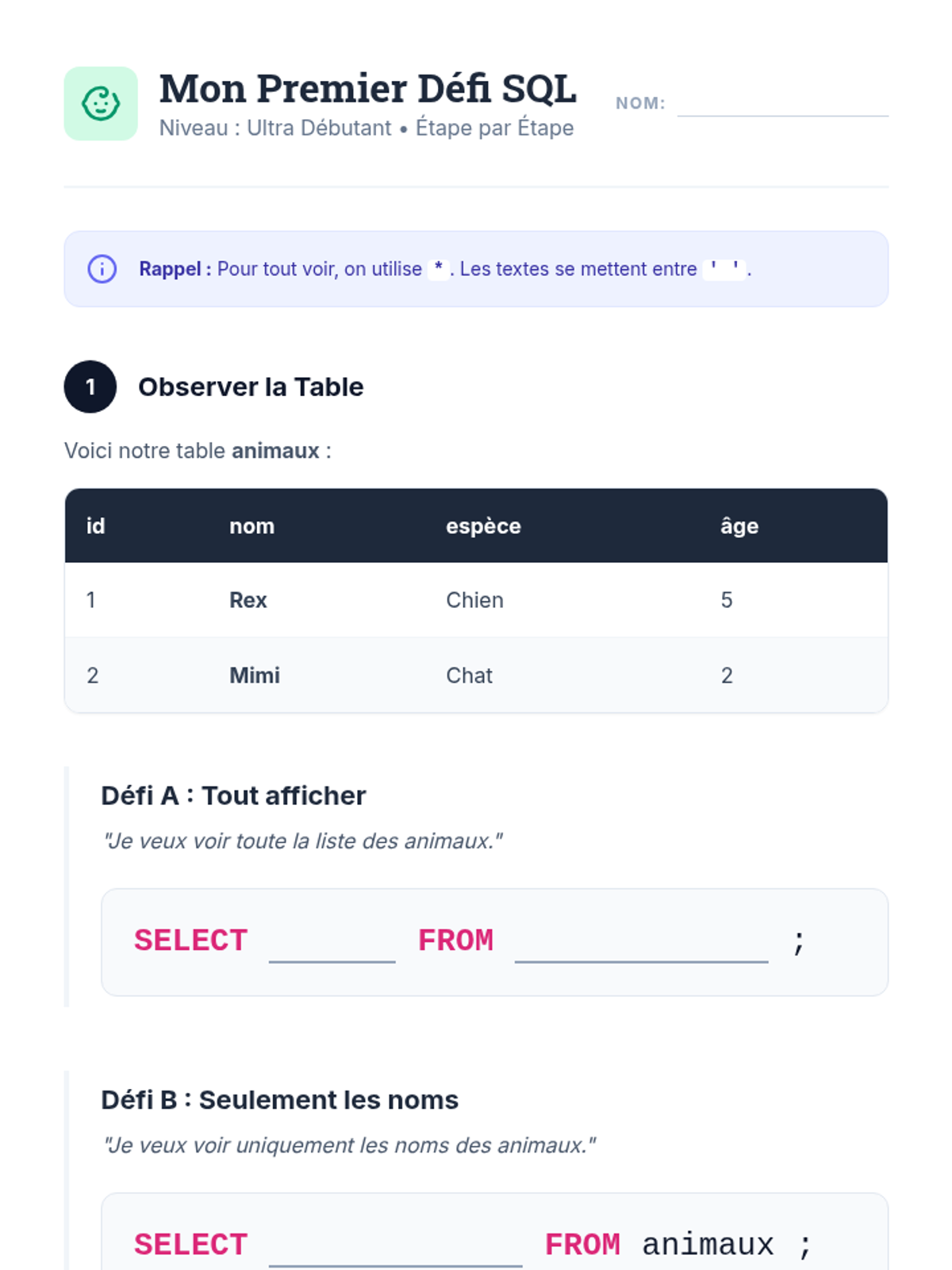
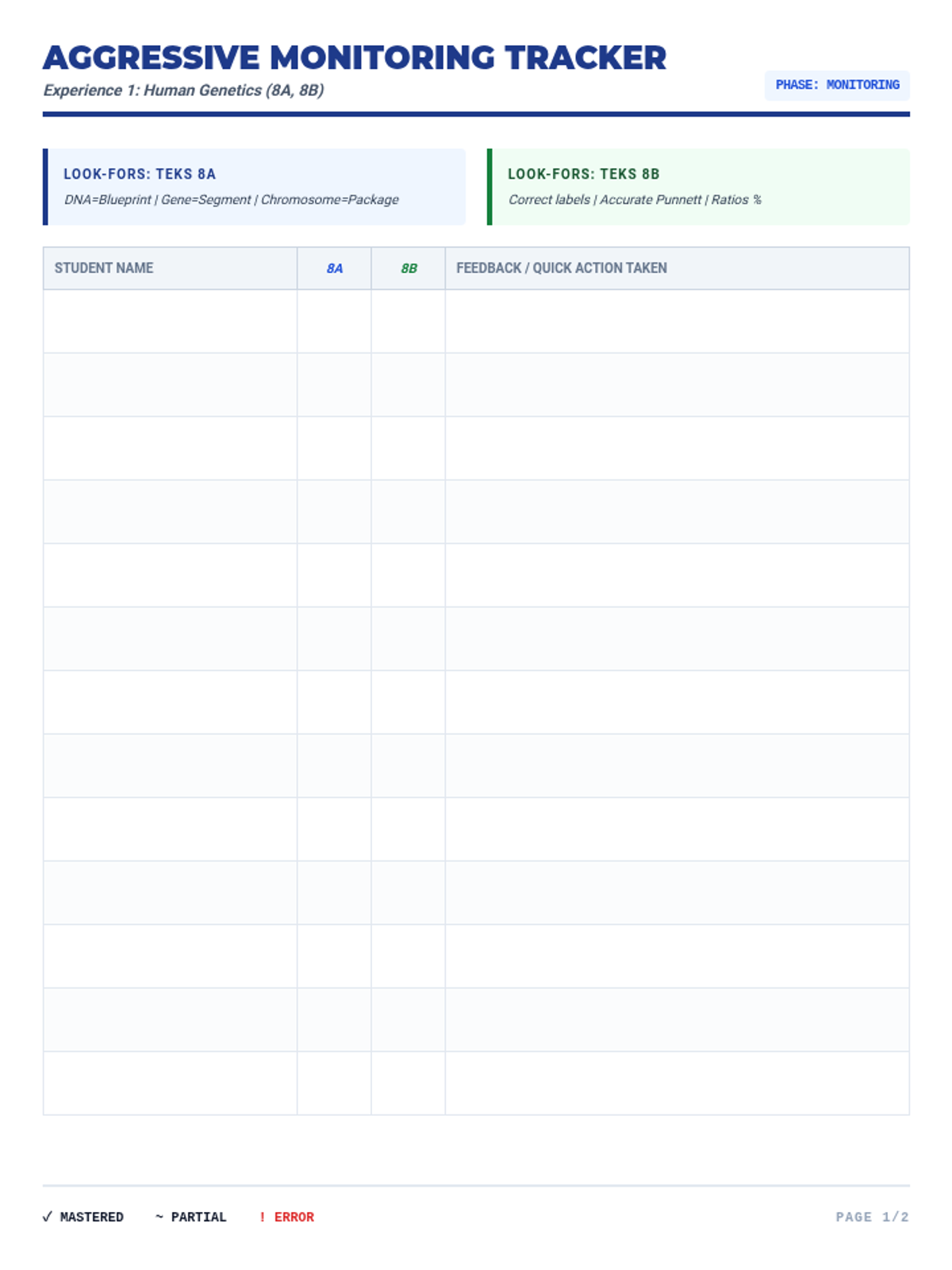
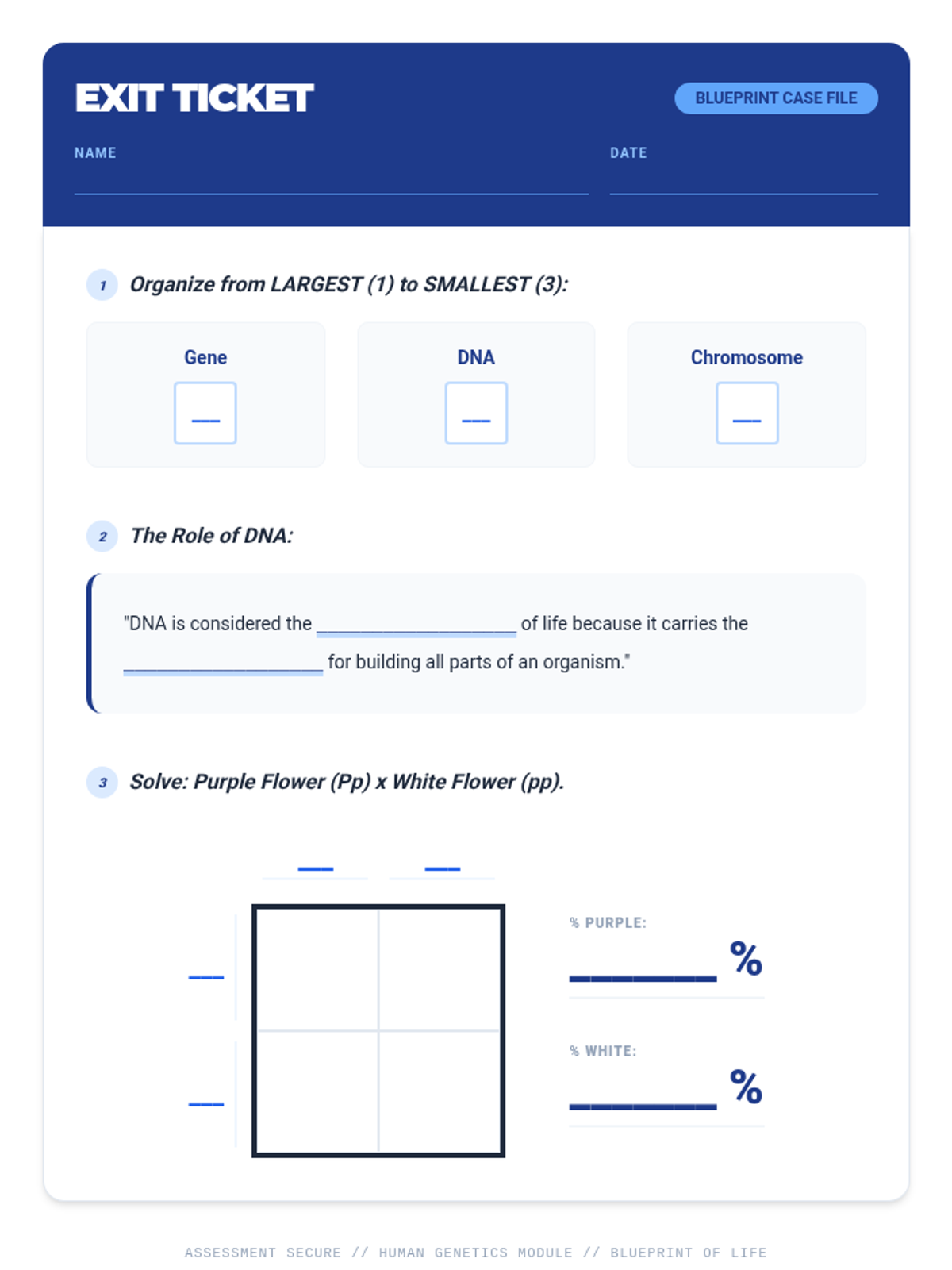
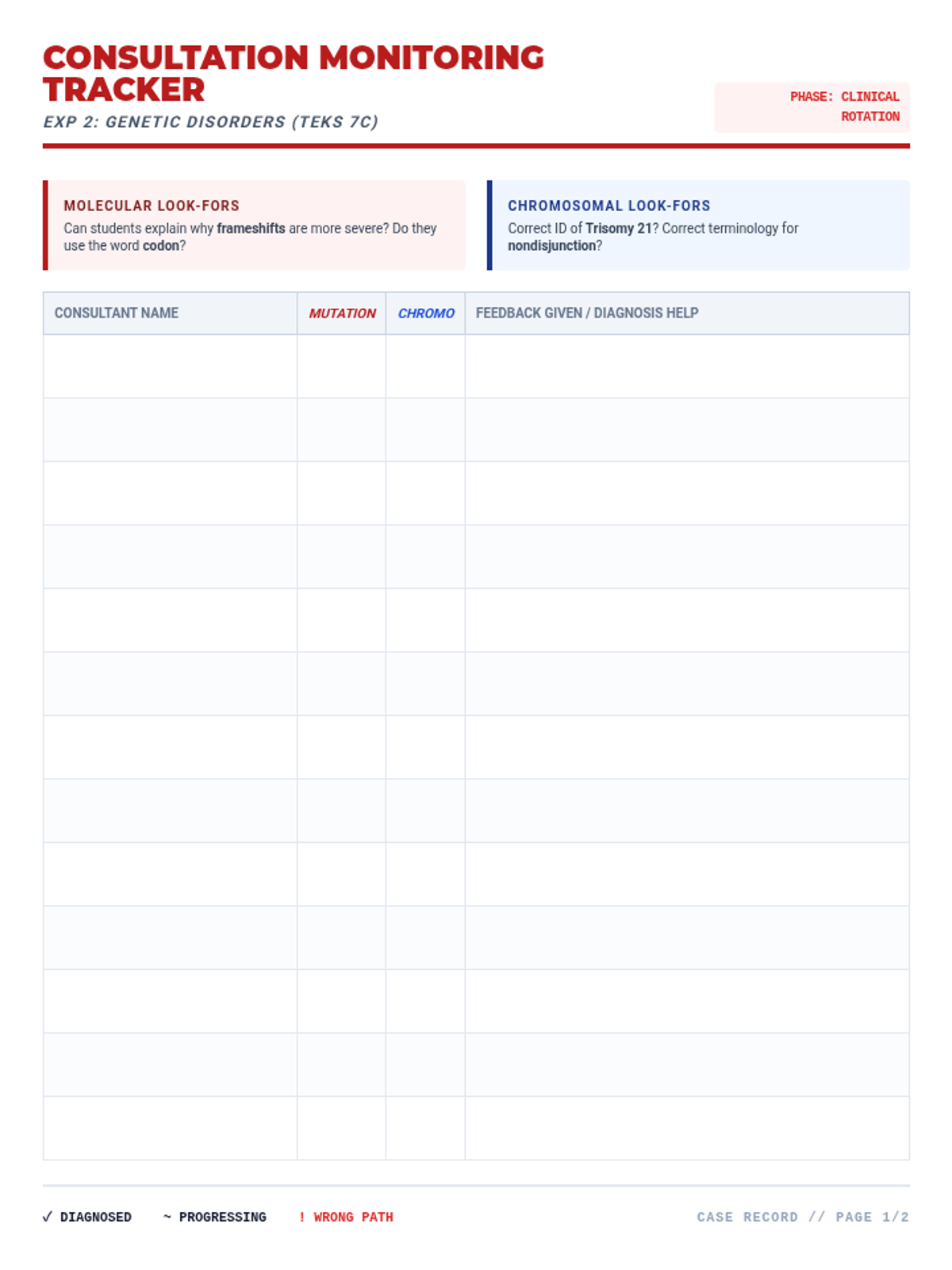
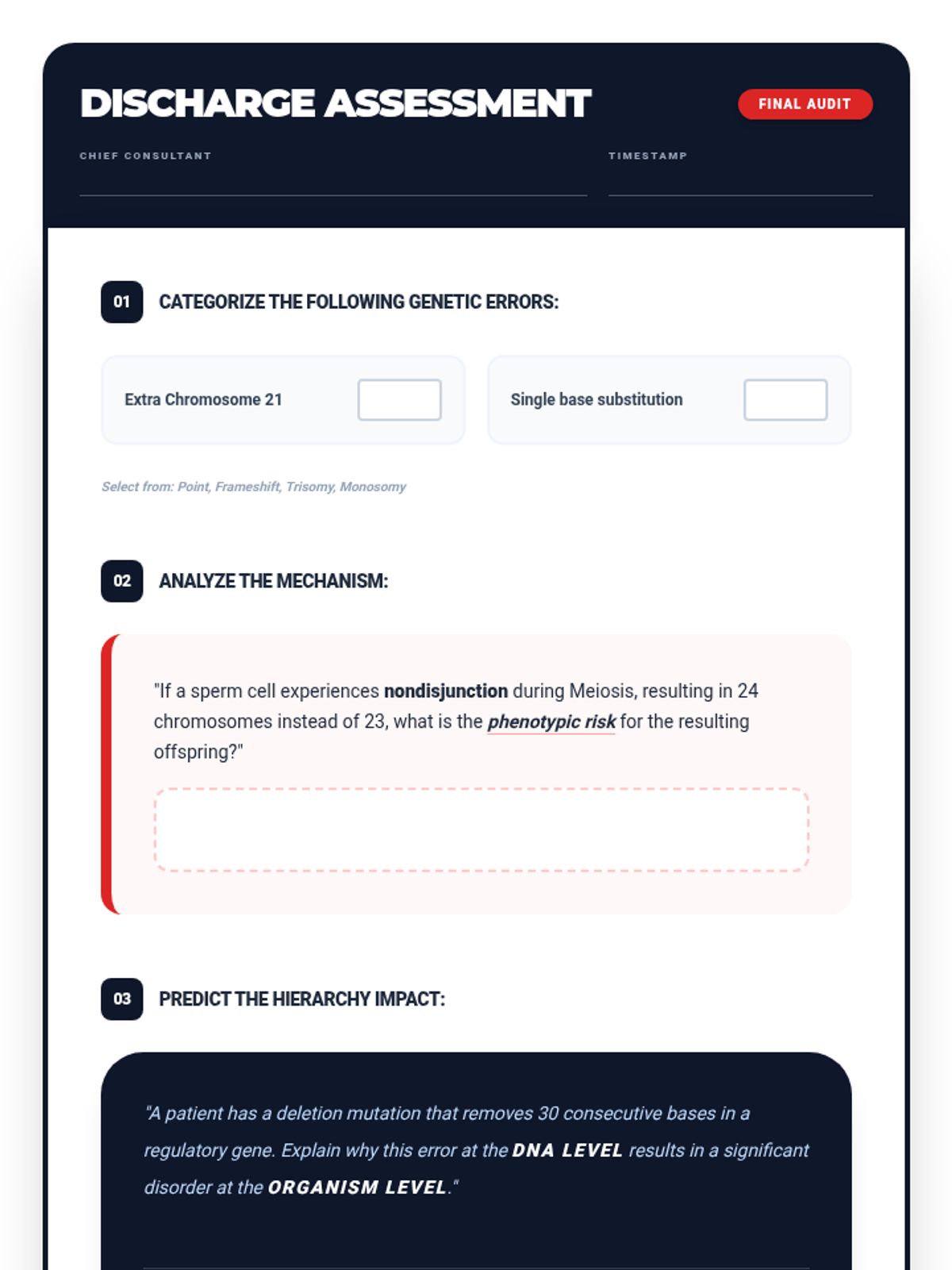
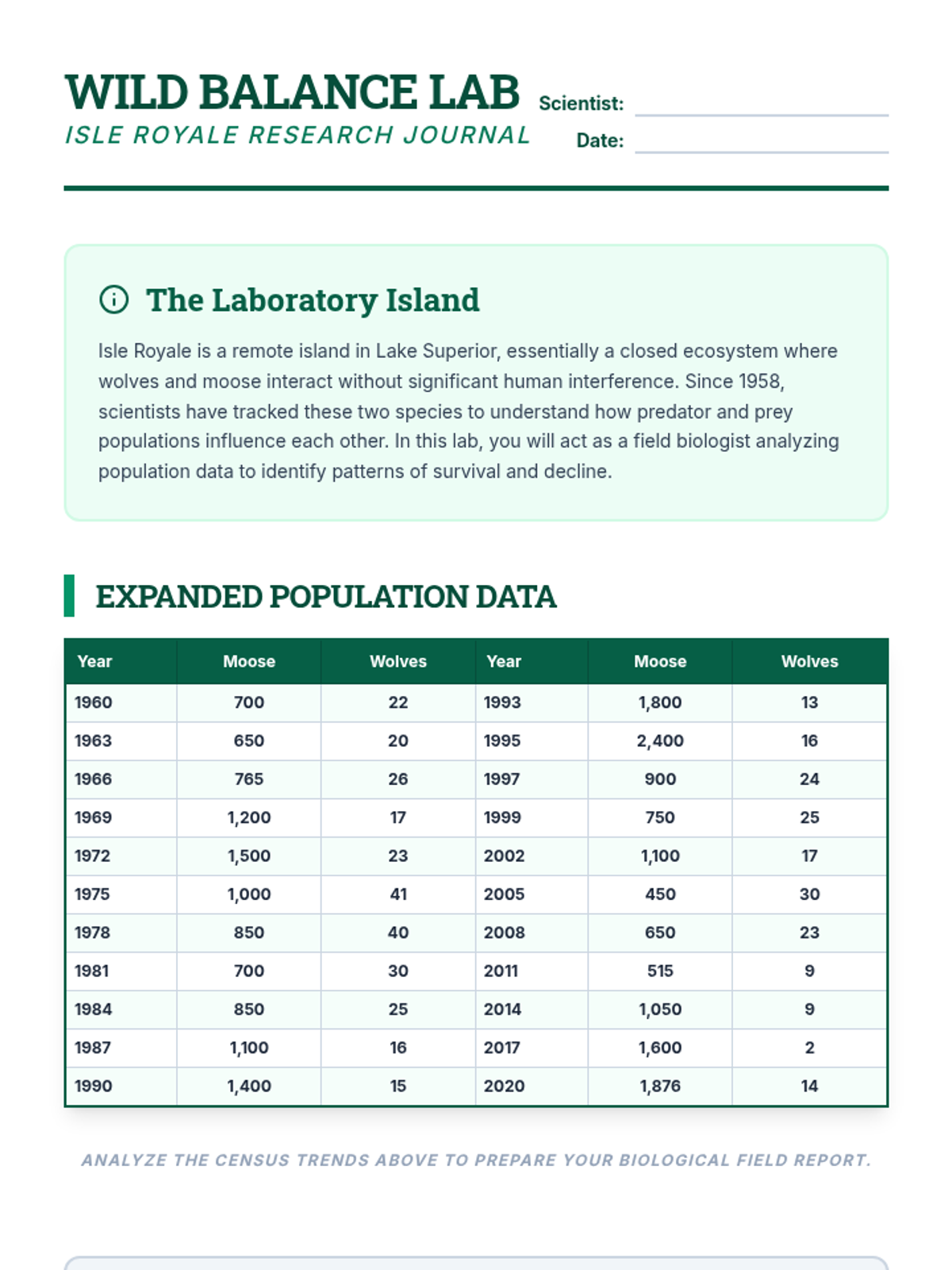
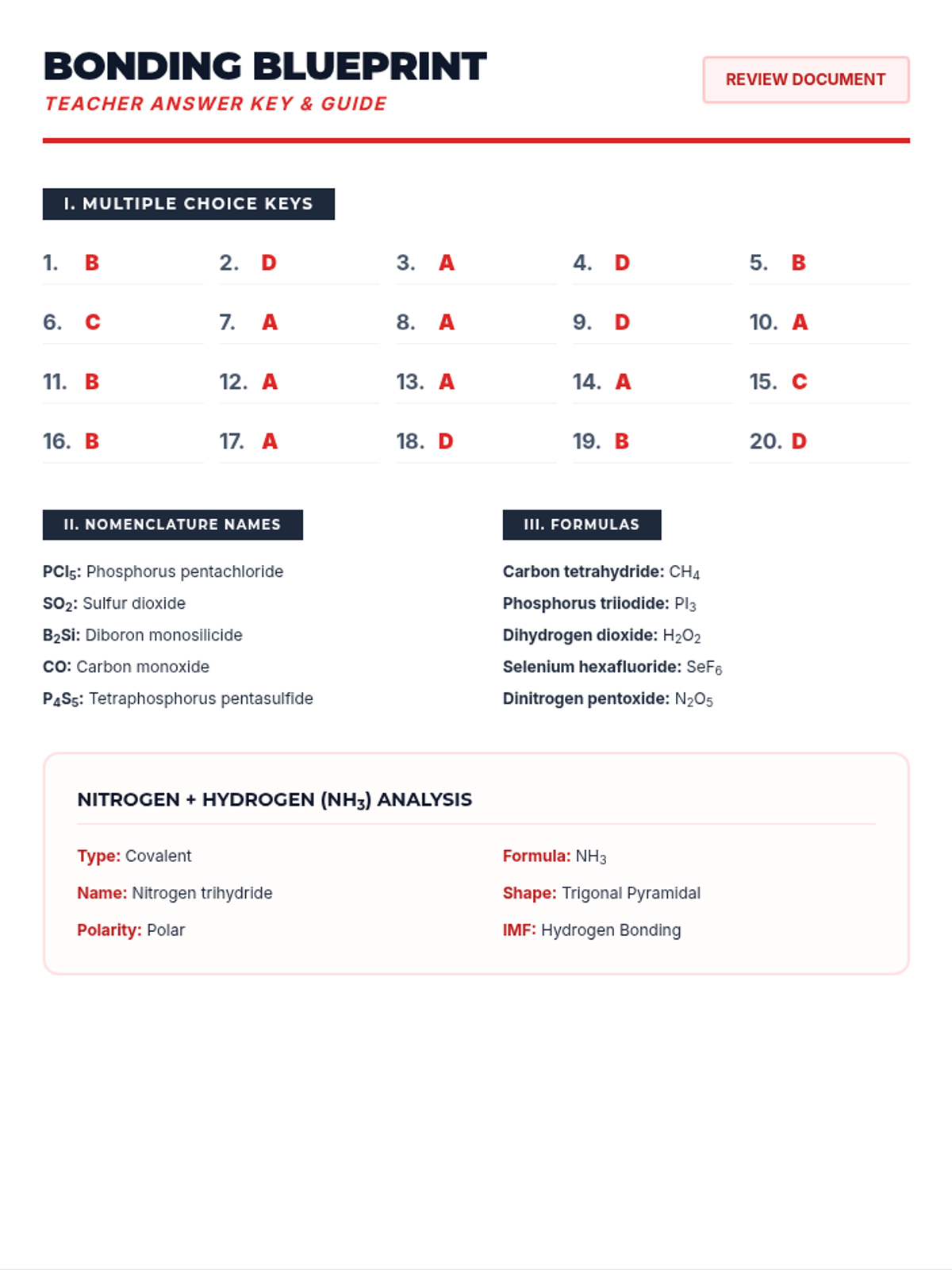
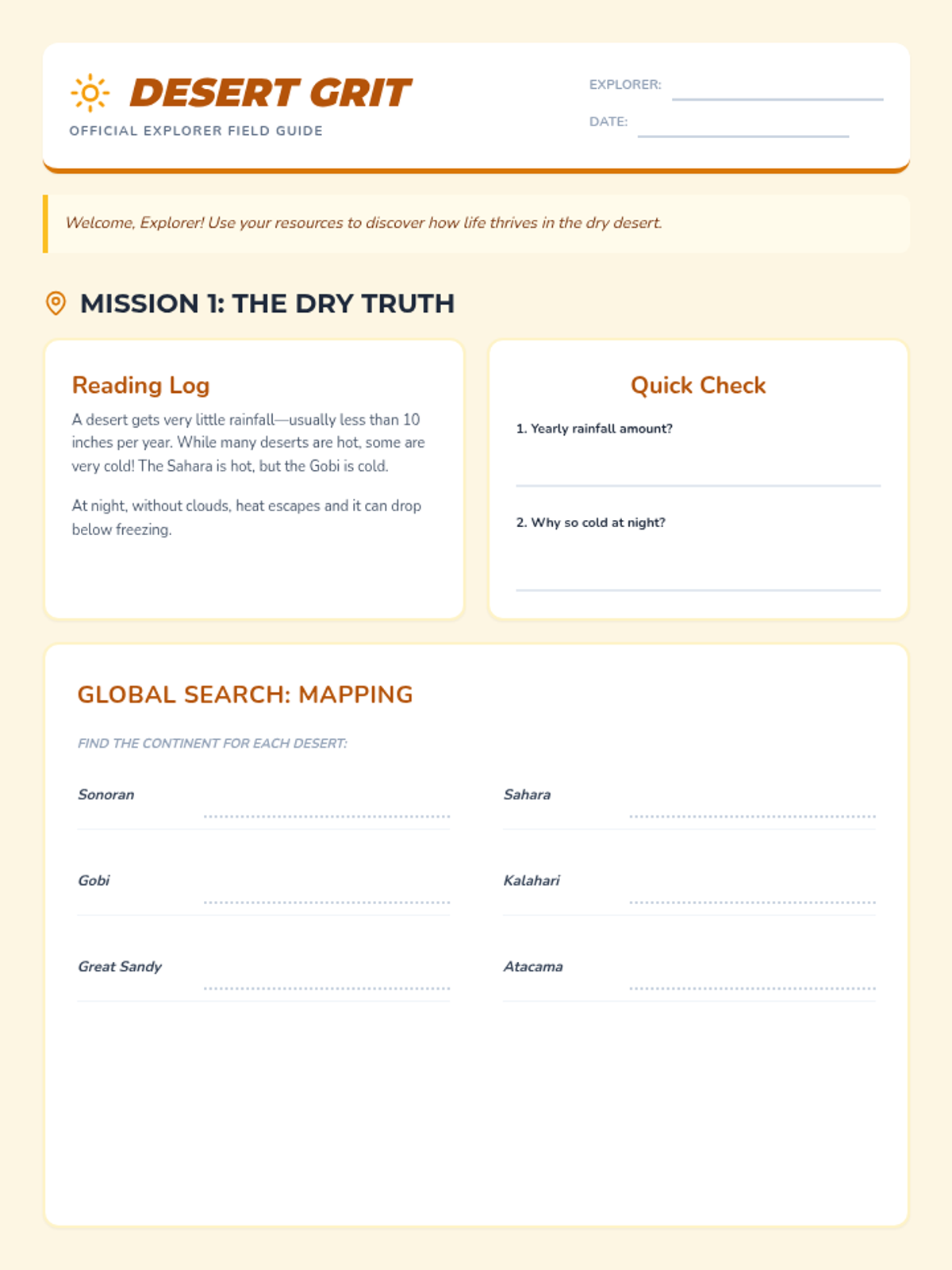
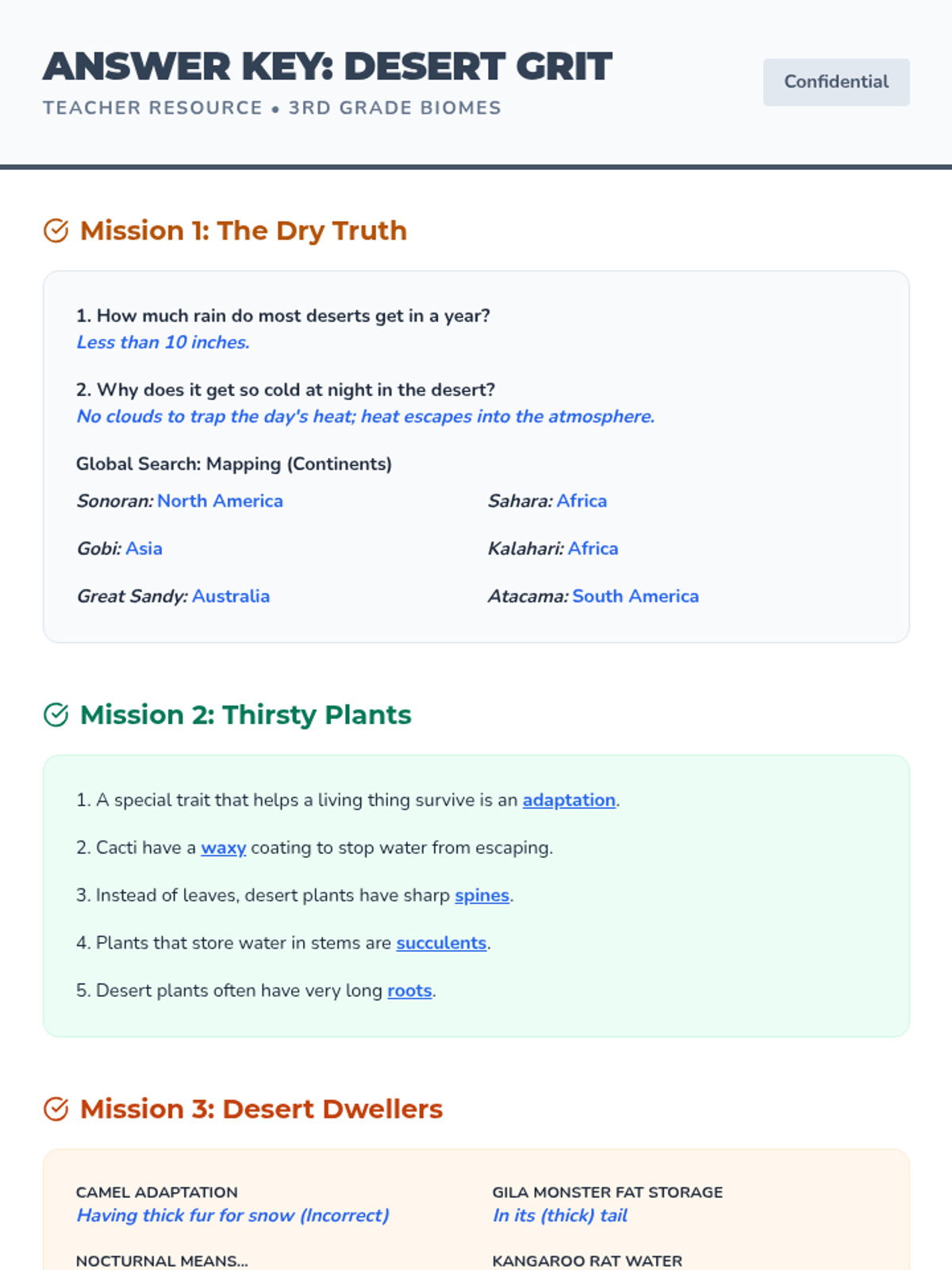
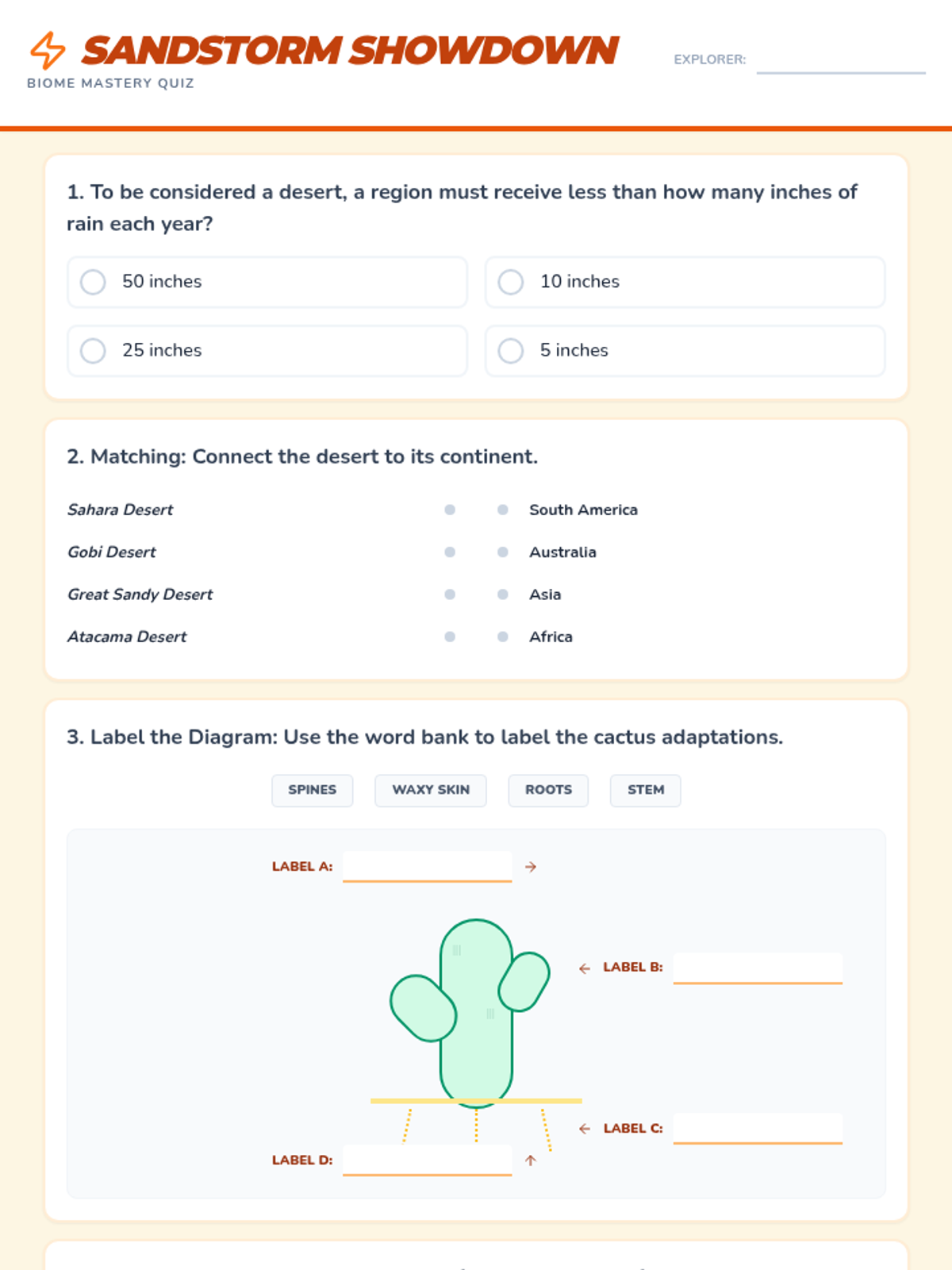
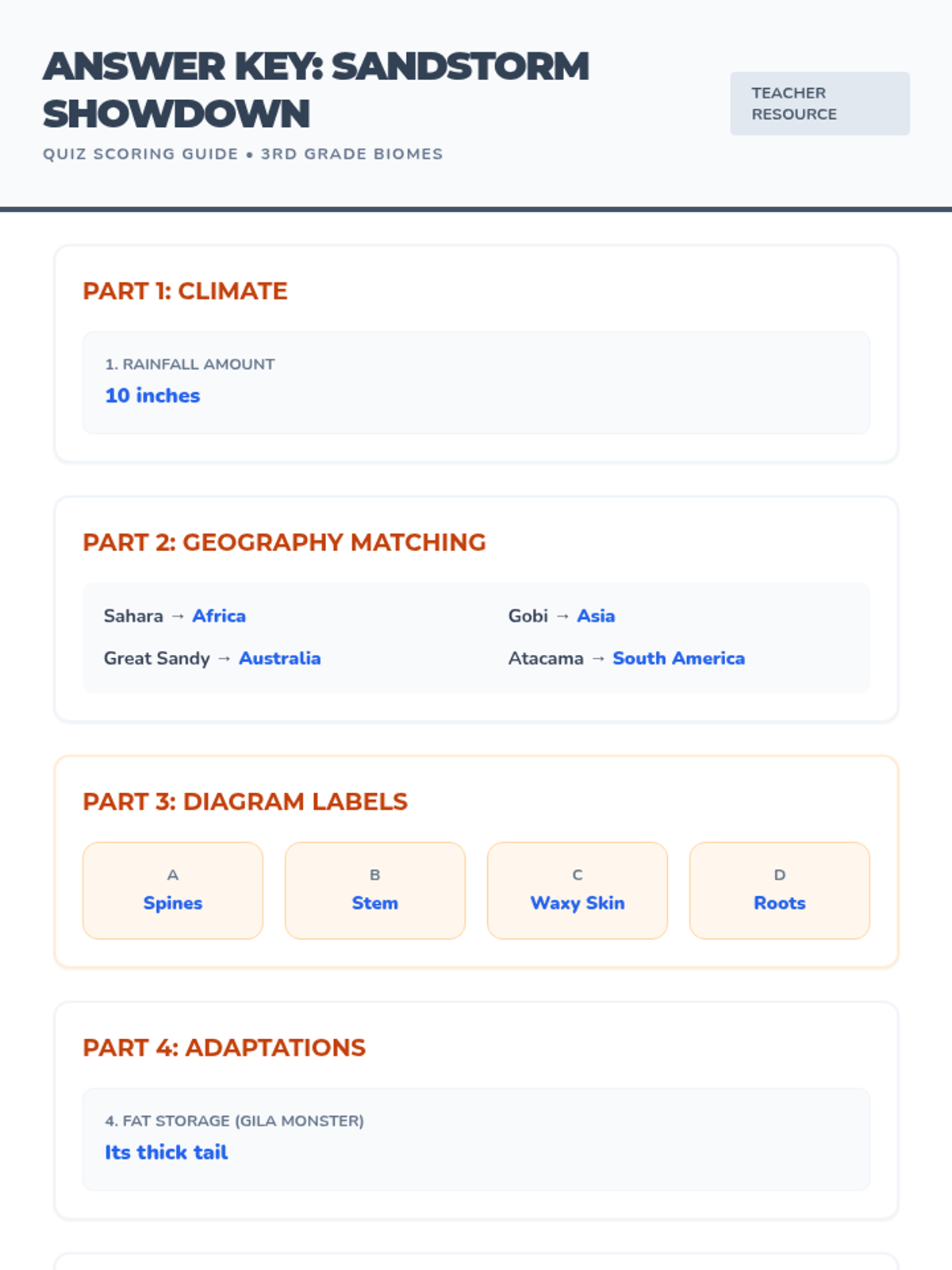
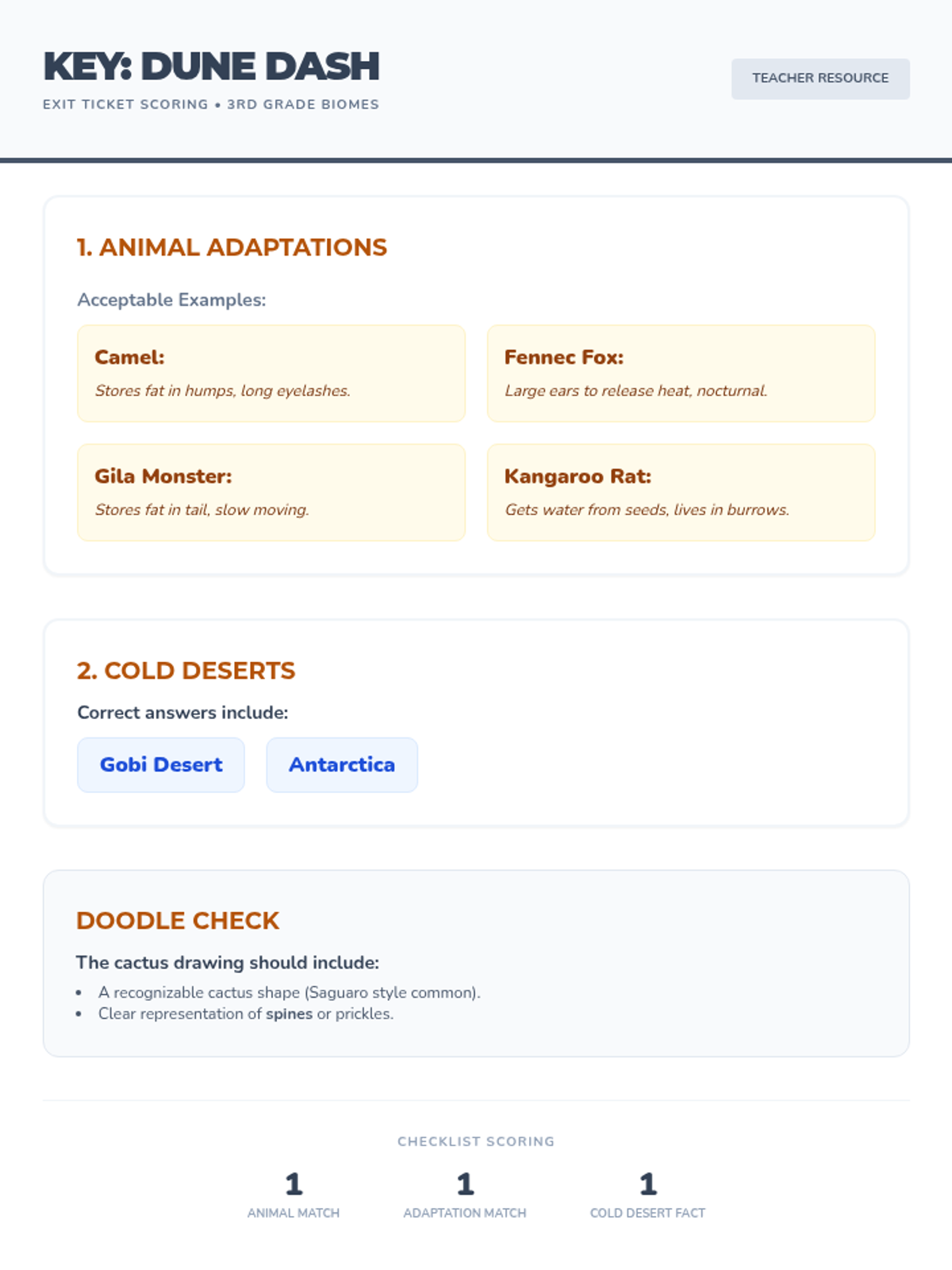
This 12th-grade chemistry sequence explores the physical changes that occur when solutes are added to solvents through the lens of colligative properties. Students investigate vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure, applying mathematical models like Raoult's Law and the Van't Hoff factor to real-world engineering and biological scenarios.